Abstract
The aim of this work was to develop a comparative study between Bacillus paralicheniformis TB197 and B. subtilis ATCC 21332 strains in terms of growth, cyclic lipopeptide production, nematicidal activity, and active lipopeptide characteristics. Crude lipopeptide extracts (CLEs) from their fermentation broths were obtained, and their nematicidal activity (NA) was estimated as the mean lethal dose (LD50), employing Caenorhabditis elegans. Using a bioguided approach, CLE components were fractionated by semipreparative thin layer chromatography, and active lipopeptides were characterized by mass spectrometry. Both strains produced similar concentrations of CLEs (p ≥ 0.05) (0.99 ± 0.11 and 1.14 ± 0.15 mg/mL by TB197 and ATCC 21332, respectively). The estimated LD50 values of CLEs from the TB197 and ATCC 21332 strains were 3.88 and 8.15 mg/mL, respectively, showing that the NA of the TB197 strain CLE was 2.1-fold higher (p ≤ 0.05). Mass spectrometry revealed that strain TB197 synthesizes several families of lipopeptides, namely, fengycin A (C14–C17), fengycin B (C16–C17), surfactin (C15–C17), and lichenysin (C12, C13, C14, and C16), from which fengycins and lichenysins possess the highest NA (100 and 60% mortality in C. elegans larvae, respectively), while the ATCC 21332 strain produces mainly surfactin (C13–C17) (NA 63% mortality). The main differences found in this study were that the TB197 strain has a higher tolerance to inhibition by the product, and the lipopeptides they synthesize have a higher nematicidal activity due to the diversity of families compared to ATCC 21332. Likewise, it was shown that more polar lipopeptides (fengycins) are more effective at causing mortality in C. elegans larvae.
Key points
• The nematicidal activity of lipopeptides from TB197 is higher than from ATCC 21332
• TB197 produces surfactin, lichenysin, and fengycin, while ATCC 21332 mainly produces surfactin
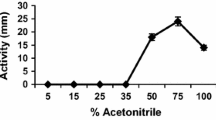
• The most polar lipopeptides (fengycins) cause more mortality in C. elegans L2




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abd-Elgawad MMM, Askary TH (2018) Fungal and bacterial nematicides in integrated nematode management strategies. Egypt J Biol Pest Control 28:1–24. https://doi.org/10.1186/s41938-018-0080-x
Awad M, EL-Shahed YI, Aziz R, Roji Sarmidi M, El-Enshasy A (2012) Antibiotics as Microbial Secondary Metabolites: Production and Application. J Teknol 59:101–111
Bertranda RL (2019) Lag phase is a dynamic, organized, adaptive, and evolvable period that prepares bacteria for cell division. J Bacteriol 201:1–21. https://doi.org/10.1128/JB.00697-18
Bui TT, Suga K, Umakoshi H (2016) Roles of sterol derivatives in regulating the properties of phospholipid bilayer systems. Langmuir 32:6176–6184. https://doi.org/10.1021/acs.langmuir.5b04343
Chen J, Li QX, Song B (2020) Chemical nematicides: recent research progress and outlook. J Agric Food Chem 68:12175–12188
Chooklin CS, Petmeaun S, Maneerat S, Saimmai A (2014) Isolation and characterization of a biosurfactant from Deinococcus caeni PO5 using jackfruit seed powder as a substrate. Ann Microbiol 64:1007–1020. https://doi.org/10.1007/s13213-013-0738-2
Coronel-León J, de Grau G, Grau-Campistany A, Farfan M, Rabanal F, Manresa A, Marqués AM (2015) Biosurfactant production by AL 1.1, a Bacillus licheniformis strain isolated from Antarctica: production, chemical characterization and properties. Ann Microbiol 65:2065–2078. https://doi.org/10.1007/s13213-015-1045-x
Deseure J, Obeid J, Willison JC, Magnin J (2021) Heliyon reliable determination of the growth and hydrogen production parameters of the photosynthetic bacterium Rhodobacter capsulatus in fed batch culture using a combination of the Gompertz function and the Luedeking-Piret model. Heliyon 7:e07394. https://doi.org/10.1016/j.heliyon.2021.e07394
Ding L, Guo W, Chen X (2019) Exogenous addition of alkanoic acids enhanced production of antifungal lipopeptides in Bacillus amyloliquefaciens Pc3. Appl Microbiol Biotechnol 103:5367–5377. https://doi.org/10.1007/s00253-019-09792-1
Dlamini B, Rangarajan V, Clarke KG (2020) A simple thin layer chromatography based method for the quantitative analysis of biosurfactant surfactin vis-a-vis the presence of lipid and protein impurities in the processing liquid. Biocatal Agric Biotechnol 25:101587. https://doi.org/10.1016/j.bcab.2020.101587
Surendhiran D, Vijay M, Sivaprakash B, Sirajunnisa A (2015) Kinetic modeling of microalgal growth and lipid synthesis for biodiesel production. 3 Biotech 5:663–669. https://doi.org/10.1007/s13205-014-0264-3
Engelbrecht G, Horak I, Jansen van Rensburg PJ, Claassens S (2018) Bacillus-based bionematicides: development, modes of action and commercialisation. Biocontrol Sci Technol 28:629–653. https://doi.org/10.1080/09583157.2018.1469000
Falardeau J, Wise C, Novitsky L, Avis TJ (2013) Ecological and mechanistic insights into the direct and indirect antimicrobial properties of Bacillus subtilis lipopeptides on plant pathogens. J Chem Ecol 39:869–878. https://doi.org/10.1007/s10886-013-0319-7
Franklin KA, Sommers N, Aslan CE, López BR, Bronstein JL, Bustamante E, Búrquez A, Medellín RA, Marazzi B (2021) Plant biotic onteractions in the Sonoran Desert: Current knowlege and future research perspectives. Int J Plant Sci 177:217–234 . https://doi.org/10.1086/684261
Fourie H, Spaull VW, Jones RK, Daneel MS, De Waele D (2017) Nematology in South Africa: A View from the 21st Century. Springer International Publishing, Durban
Gao H, Qi G, Yin R, Zhang H, Li C, Zhao X (2016) Bacillus cereus strain S2 shows high nematicidal activity against Meloidogyne incognita by producing sphingosine. Sci Rep 6:1–11 . https://doi.org/10.1038/srep28756
Ghribi D, Ellouze-Chaabouni S (2011) Enhancement of Bacillus subtilis lipopeptide biosurfactants production through optimization of medium composition and adequate control of aeration. Biotechnol Res Int 2011:1–6. https://doi.org/10.4061/2011/653654
Gu XB, Zheng ZM, Yu HQ, Wang J, Liang FL, Liu RL (2005) Optimization of medium constituents for a novel lipopeptide production by Bacillus subtilis MO-01 by a response surface method. Process Biochem 40:3196–3201 . https://doi.org/10.1016/j.procbio.2005.02.011
Gutiérrez-Chávez C, Benaud N, Ferrari BC (2021) The ecological roles of microbial lipopeptides: where are we going? Comput Struct Biotechnol J 19:1400–1413
Hamill PG, Stevenson A, McMullan PE, Williams JP, Lewis ADR, Sudharsan S, Stevenson KE, Farnsworth KD, Khroustalyova G, Takemoto JY, Quinn JP, Rapoport A, Hallsworth JE (2020) Microbial lag phase can be indicative of, or independent from, cellular stress. Sci Rep 10:1–20. https://doi.org/10.1038/s41598-020-62552-4
Hamley IW, Dehsorkhi A, Jauregi P, Seitsonen J, Ruokolainen J, Coutte F, Chataigné G, Jacques P (2013) Self-assembly of three bacterially-derived bioactive lipopeptides. Soft Matter 9:9572–9578. https://doi.org/10.1039/c3sm51514a
Helder J, Vervoort M, Megen H van, Katarzyna R-M, Quist C, Bakker GS and J (2015) Phytopathogenic Nematodes. Springer Berlin Heidelberg, London
Heryani H, Putra MD (2017) Kinetic study and modeling of biosurfactant production using Bacillus sp. Electron J Biotechnol 27:49–54. https://doi.org/10.1016/j.ejbt.2017.03.005
Horak I, Engelbrecht G, van Rensburg PJJ, Claassens S (2019) Microbial metabolomics: essential definitions and the importance of cultivation conditions for utilizing Bacillus species as bionematicides. J Appl Microbiol 127:326–343
Hussain T, Khan AA (2020) Bacillus subtilis HussainT-AMU and its Antifungal activity against Potato Black scurf caused by Rhizoctonia solani on seed tubers. Biocatal Agric Biotechnol 23:2–19 . https://doi.org/10.1016/j.bcab.2019.101443
Jaishankar J, Srivastava P (2017) Molecular basis of stationary phase survival and applications. Front Microbiol 8:1–12. https://doi.org/10.3389/fmicb.2017.02000
Jemil N, Manresa A, Rabanal F, Ben Ayed H, Hmidet N, Nasri M (2017) Structural characterization and identification of cyclic lipopeptides produced by Bacillus methylotrophicus DCS1 strain. J Chromatogr B Anal Technol Biomed Life Sci 1060:374–386. https://doi.org/10.1016/j.jchromb.2017.06.013
Jones JT, Haegeman A, Danchin EGJ, Gaur HS, Helder J, Jones MGK, Kikuchi T, Manzanilla-López R, Palomares-Rius JE, Wesemael WML, Perry RN (2013) Top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol 14:946–961. https://doi.org/10.1111/mpp.12057
Kang BR, Park JS, Jung WJ (2020) Antifungal evaluation of fengycin isoforms isolated from Bacillus amyloliquefaciens PPL against Fusarium oxysporum f. sp. lycopersici. Microb Pathog 149:104509. https://doi.org/10.1016/j.micpath.2020.104509
Kaspar F, Neubauer P, Gimpel M (2019) Bioactive secondary metabolites from Bacillus subtilis: a comprehensive review. J Nat Prod 82:2038–2053. https://doi.org/10.1021/acs.jnatprod.9b00110
Kim K, Lee Y, Ha A, Kim JI, Park AR, Yu NH, Son H, Choi GJ, Park HW, Lee CW, Lee T, Lee YW, Kim JC (2017) Chemosensitization of fusarium graminearum to chemical fungicides using cyclic lipopeptides produced by bacillus amyloliquefaciens strain JCK-12. Front Plant Sci 8:1–16. https://doi.org/10.3389/fpls.2017.02010
Li J, Zou C, Xu J, Ji X, Niu X, Yang J, Huang X, Zhang KQ (2015) Molecular mechanisms of nematode-nematophagous microbe interactions: basis for biological control of plant-parasitic nematodes. Annu Rev Phytopathol 53:67–95. https://doi.org/10.1146/annurev-phyto-080614-120336
Li YM, Haddad NIA, Yang SZ, Mu BZ (2008) Variants of lipopeptides produced by Bacillus licheniformis HSN221 in different medium components evaluated by a rapid method ESI-MS. Int J Pept Res Ther 14:229–235. https://doi.org/10.1007/s10989-008-9137-0
Lima FSO, Mattos VS, Silva ES, Carvalho MAS, Teixeira RA, Silva JC, Correa VR (2018) Nematodes Affecting Potato and Sustainable Practices for Their Management. In: Soybean - The Basis of Yield, Biomass and Productivity. Dianópolis, pp 95–110. https://doi.org/10.5772/67030
Medeot DB, Fernandez M, Morales GM, Jofré E (2020) Fengycins from bacillus amyloliquefaciens MEP218 exhibit antibacterial activity by producing alterations on the cell surface of the pathogens Xanthomonas axonopodis pv. vesicatoria and Pseudomonas aeruginosa PA01. Front Microbiol 10:1–12. https://doi.org/10.3389/fmicb.2019.03107
Meyers CLF, Meyers DJ (2008) Thin-Layer Chromatography. In: Current Protocols in Nucleic Acid Chemistry, 1st edn. Wiley Interscience, Baltimore, pp 1–13
Mihalache G, Balaes T, Gostin I, Stefan M, Coutte F, Krier F (2018) Lipopeptides produced by Bacillus subtilis as new biocontrol products against fusariosis in ornamental plants. Environ Sci Pollut Res 25:29784–29793. https://doi.org/10.1007/s11356-017-9162-7
Mongkolthanaruk W (2012) Classification of Bacillus beneficial substances related to plants, humans and animals. J Microbiol Biotechnol 22:1597–1604. https://doi.org/10.4014/jmb.1204.04013
Nadeem H, Niazi P, Asif M, Kaskavalci G, Ahmad F (2021) Bacterial strains integrated with surfactin molecules of Bacillus subtilis MTCC441 enrich nematocidal activity against Meloidogyne incognita. Plant Biol 23:1027–1036 . https://doi.org/10.1111/plb.13301
Nanjundan J, Ramasamy R, Uthandi S, Ponnusamy M (2019) Antimicrobial activity and spectroscopic characterization of surfactin class of lipopeptides from Bacillus amyloliquefaciens SR1. Microb Pathog 128:374–380. https://doi.org/10.1016/j.micpath.2019.01.037
Nazareth TC, Zanutto CP, Tripathi L, Juma A, Maass D, de Souza AAU, de Arruda Guelli Ulson de Souza SM, Banat IM (2020) The use of low-cost brewery waste product for the production of surfactin as a natural microbial biocide. Biotechnol Reports 28:1–10. https://doi.org/10.1016/j.btre.2020.e00537
Ongena M, Jacques P (2008) Bacillus lipopeptides: versatile weapons for plant disease biocontrol. Trends Microbiol 16:115–125. https://doi.org/10.1016/j.tim.2007.12.009
Penha RO, Vandenberghe LPS, Faulds C, Soccol VT, Soccol CR (2020) Bacillus lipopeptides as powerful pest control agents for a more sustainable and healthy agriculture: recent studies and innovations. Planta 251:1–15 . https://doi.org/10.1007/s00425-020-03357-7
Porta-de-la-Riva M, Fontrodona L, Villanueva A, Cerón J (2012) Basic Caenorhabditis elegans Methods: Synchronization and Observation. Jove 64:1–9 . https://doi.org/10.3791/4019
Ramalakshmi A, Sharmila R, Iniyakumar M, Gomathi V (2020) Nematicidal activity of native Bacillus thuringiensis against the root knot nematode, Meloidogyne incognita (Kofoid and White). Egypt J Biol Pest Control 30:1–16 . https://doi.org/10.1186/s41938-020-00293-2
Rangarajan V, Clarke KG (2016) Towards bacterial lipopeptide products for specific applications — a review of appropriate downstream processing schemes. Process Biochem 51:2176–2185. https://doi.org/10.1016/j.procbio.2016.08.026
Rosas-Garcia N (2009) Biopesticide production from Bacillus thuringiensis: an environmentally friendly alternative. Recent Pat Biotechnol 3:28–36. https://doi.org/10.2174/187220809787172632
Rowe L, Peller J, Mammoser C, Davidson K, Gunter A (2018) Stability of non-proteinogenic amino acids to UV and gamma irradiation. Int J Astrobiol 18:2–10. https://doi.org/10.1017/S1473550418000381
Ruiz M, Bodhicharla R, Svensk E, Devkota R, Busayavalasa K, Palmgren H, Ståhlman M, Boren J, Pilon M (2018) Membrane fluidity is regulated by the C. Elegans transmembrane protein FLD-1 and its human homologs TLCD1/2. Elife 7:1–25. https://doi.org/10.7554/eLife.40686
Satya Eswari J, Anand M, Venkateswarlu C (2016) Optimum culture medium composition for lipopeptide production by Bacillus subtilis using response surface model-based ant colony optimization. Sadhana 41:55–65. https://doi.org/10.1007/s12046-015-0451-x
Sivapathasekaran C, Mukherjee S, Sen R (2010) Biosurfactant production and growth kinetics of bacteria in a designer marine medium: improved physiochemical properties. Biotechnol J 5:1060–1068. https://doi.org/10.1002/biot.201000175
Tian B, Yang J, Zhang KQ (2007) Bacteria used in the biological control of plant-parasitic nematodes: populations, mechanisms of action, and future prospects. FEMS Microbiol Ecol 61:197–213. https://doi.org/10.1111/j.1574-6941.2007.00349.x
Tranier MS, Pognant-Gros J, de la Cruz Quiroz R, González CNA, Mateille T, Roussos S (2014) Commercial biological control agents targeted against plant-parasitic root-knot nematodes. Brazilian Arch Biol Technol 57:831–841. https://doi.org/10.1590/S1516-8913201402540
Trudgill DL, Blok VC (2001) Apomitic polyphagous root knot nematodes : exceptionally successful and damaging biotrophic root pathogens. Annu Rev Phytopathol 39:53–77
Tunsagool P, Ploypetch S, Jaresitthikunchai J, Roytrakul S, Choowongkomon K, Rattanasrisomporn J (2021) Efficacy of cyclic lipopeptides obtained from Bacillus subtilis to inhibit the growth of Microsporum canis isolated from cats. Heliyon 7:e07980. https://doi.org/10.1016/j.heliyon.2021.e07980
Tyc O, Song C, Dickschat JS, Vos M, Garbeva P (2017) The ecological role of volatile and soluble secondary metabolites Produced by Soil Bacteria. Trends Microbiol 25:280–292
Wang Y, Zhang C, Liang J, Wang L, Gao W, Jiang J, Chang R (2020) Surfactin and fengycin B extracted from Bacillus pumilus W-7 provide protection against potato late blight via distinct and synergistic mechanisms. Appl Microbiol Biotechnol 104:7467–7481. https://doi.org/10.1007/s00253-020-10773-y
Xiang N, Lawrence KS, Kloepper JW, Donald PA, McInroy JA, Lawrence GW (2017) Biological control of Meloidogyne incognita by spore-forming plant growth-promoting rhizobacteria on cotton. Plant Dis 101:774–784. https://doi.org/10.1094/PDIS-09-16-1369-RE
Yakimov MM, Abraham WR, Meyer H, Giuliano L, Golyshin PN (1999) Structural characterization of lichenysin A components by fast atom bombardment tandem mass spectrometry. Biochim Biophys Acta Mol Cell Biol Lipids 1438:273–280. https://doi.org/10.1016/S1388-1981(99)00058-X
Yang H, Li X, Li X, Yu H, Shen Z (2015) Identification of lipopeptide isoforms by MALDI-TOF-MS/MS based on the simultaneous purification of iturin, fengycin, and surfactin by RP-HPLC. Anal Bioanal Chem 407:2529–2542. https://doi.org/10.1007/s00216-015-8486-8
Zhao H, Shao D, Jiang C, Shi J, Li Q, Huang Q, Rajoka MSR, Yang H, Jin M (2017) Biological activity of lipopeptides from Bacillus. Appl Microbiol Biotechnol 101:5951–5960. https://doi.org/10.1007/s00253-017-8396-0
Zheng Z, Zheng J, Zhang Z, Peng D, Sun M (2016) Nematicidal spore-forming Bacilli share similar virulence factors and mechanisms. Sci Rep 6:1–10 . https://doi.org/10.1038/srep31341
Acknowledgements
Author Estefany Chavarria acknowledges the scholarship (number 867046) granted by the National Council on Science and Technology (CONACYT) for PhD studies. We are grateful to Innovak Global for the financial support and access to their R&D laboratories.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. EC performed all the experiments, analyzed the data, and wrote the original manuscript. EC and MG analyzed the mass spectra. AA supervised the research and reviewed the manuscript. All the authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chavarria-Quicaño, E., De la Torre-González, F., González-Riojas, M. et al. Nematicidal lipopeptides from Bacillus paralicheniformis and Bacillus subtilis: A comparative study. Appl Microbiol Biotechnol 107, 1537–1549 (2023). https://doi.org/10.1007/s00253-023-12391-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12391-w