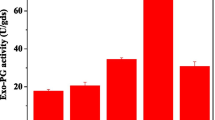
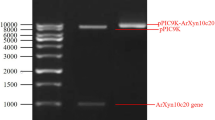
Abstract
Sugar beet pulp is an agricultural processing residue that is a rich source of the cell wall polysaccharide arabinan. Functional oligosaccharides, specifically feruloylated arabino-oligosaccharides (FAOs), can be isolated from sugar beet pulp through selective action by endo-arabinanase (glycoside hydrolase family 43). This study aimed to develop yeast (Pichia pastoris) as an efficient, eukaryotic platform to produce a thermophilic endo-1,5-α-l-arabinanase (TS-ABN) for extracting FAOs from sugar beet pulp. Recombinant TS-ABN was secreted into yeast culture medium at a yield of ~ 80 mg/L, and the protein exhibited specific enzyme activity, pH and temperature optimum, and thermostability comparable to those of the native enzyme. Treatment of sugar beet pulp with Pichia-secreted TS-ABN released FAOs recovered by hydrophobic chromatography at 1.52% (w/w). The isolated FAOs averaged seven arabinose residues per ferulic acid, and treatment of T84 human colon epithelial cells significantly increased expression of two key tight junction-related proteins—zonula occludens-1 and occludin—in a dose-dependent manner. This research establishes a biochemical platform for utilizing sugar beet pulp to produce value-added bioproducts with potential nutraceutical applications.








Similar content being viewed by others
References
Ahmad M, Hirz M, Pichler H, Schwab H (2014) Protein expression in Pichia pastoris: recent achievements and perspectives for heterologous protein production. Appl Microbiol Biotechnol 98(12):5301–5317. https://doi.org/10.1007/s00253-014-5732-5
Anonymity (2016) Agricultural Statistics 2016, United States Department of Agriculture. https://www.nass.usda.gov/Publications/Ag_Statistics/2016/Complete%20Ag%20Stats%202016.pdf
Asadi M (2007) Beet sugar handbook. Wiley, Hoboken
Babbar N, Dejonghe W, Gatti M, Sforza S, Elst K (2016) Pectic oligosaccharides from agricultural by-products: production, characterization and health benefits. Crit Rev Biotechnol 36(4):594–606. https://doi.org/10.3109/07388551.2014.996732
Barathiraja S, Gangadhara PAV, Umapathi V, Dechamma HJ, Reddy GR (2018) Expression and purification of biologically active bovine interferon lambda3 (IL28B) in Pichia pastoris. Protein Expr Purif 145:14–18. https://doi.org/10.1016/j.pep.2017.12.007
Blumenkrantz N, Asboe-Hansen G (1973) New methods for quantitative determination of uronic acids. Anal Biochem 54:484–489. https://doi.org/10.1016/0003-2697(73)90377-1
Brunton NP, Gormley TR, Murray B (2007) Use of the alditol acetate derivatisation for the analysis of reducing sugars in potato tubers. Food Chem 104(1):398–402. https://doi.org/10.1016/j.foodchem.2007.01.045
Cantarel BL, Coutinho PM, Rancurel C, Bernard T, Lombard V, Henrissat B (2009) The Carbohydrate-Active EnZymes database (CAZy): an expert resource for glycogenomics. Nucleic Acids Res 37:233–238. https://doi.org/10.1093/nar/gkn663
Colquhoun I, Ralet MC, Thibault JF, Faulds CB (1994) Feruloylated oligosaccharides form cell wall polysaccharides from sugar beet pulp by NMR spectroscopy. Carbohydr Res 263:243–256. https://doi.org/10.1104/pp.103.035311
Doner LW, Irwin PL (1992) Assay of reducing end-groups in oligosaccharide homologues with 2,2'-bicinchoninate. Anal Biochem 202(1):50–53. https://doi.org/10.1016/0003-2697(92)90204-k
Evans E, Messerschmidt U (2017) Review: Sugar beets as a substitute for grain for lactating dairy cattle. J Anim Sci Biotechnol 8:25. https://doi.org/10.1186/s40104-017-0154-8
Finkenstadt VL (2014) A review on the complete utilization of the sugarbeet. Sugar Tech 16:339–346. https://doi.org/10.1007/s12355-013-0285-y
Ge XM, Green S, Zhang N, Sivakumar G, Xu J (2012) Eastern gamagrass as an alternative cellulosic feedstock for bioethanol production. Process Biochem 47:335–339. https://doi.org/10.1016/j.procbio.2011.11.008
Hartsock A, Nelson WJ (2008) Adherens and tight junctions: structure, function and connections to the actin cytoskeleton. Biochim Biophys Acta 1778(3):660–669. https://doi.org/10.1016/j.bbamem.2007.07.012
Holck J, Lorentzen A, Vigsnaes LK, Licht TR, Mikkelsen JD, Meyer AS (2011) Feruloylated and nonferuloylated arabino-oligosaccharides from sugar beet pectin selectively stimulate the growth of Bifidobacterium spp. in human fecal in vitro fermentations. J Agric Food Chem 59(12):6511–6519. https://doi.org/10.1021/jf200996h
Jitonnom J, Hannongbua S (2018) Theoretical study of the arabinan hydrolysis by an inverting GH43 arabinanase. Mol Simul 44(8):631–637. https://doi.org/10.1080/08927022.2017.1422212
Kosinska A, Andlauer W (2013) Modulation of tight junction integrity by food components. Food Res Int 54:951–960. https://doi.org/10.1016/j.foodres.2012.12.038
Kroger S, Vahjen W, Zentek J (2017) Influence of lignocellulose and low or high levels of sugar beet pulp on nutrient digestibility and the fecal microbiota in dogs. J Anim Sci 95(4):1598–1605. https://doi.org/10.2527/jas.2016.0873
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Leijdekkers AG, Aguirre M, Venema K, Bosch G, Gruppen H, Schols HA (2014) In vitro fermentability of sugar beet pulp derived oligosaccharides using human and pig fecal inocula. J Agric Food Chem 62(5):1079–1087. https://doi.org/10.1021/jf4049676
Levigne SV, Ralet MC, Quemener BC, Pollet BN, Lapierre C, Thibault JF (2004) Isolation from sugar beet cell walls of arabinan oligosaccharides esterified by two ferulic acid monomers. Plant Physiol 134(3):1173–1180. https://doi.org/10.1104/pp.103.035311
Lineweaver H, Burk D (1934) The determinantion of enzyme dissociation constants. J Am Chem Soc 56(3):658–666. https://doi.org/10.1021/ja01318a036
Ma X, Liu L, Li Q, Liu Y, Yi L, Ma L, Zhai C (2017) High-level expression of a bacterial laccase, CueO from Escherichia coli K12 in Pichia pastoris GS115 and its application on the decolorization of synthetic dyes. Enzym Microb Technol 103:34–41. https://doi.org/10.1016/j.enzmictec.2017.04.004
McKie VA, Black GW, Millward-Sadler SJ, Hazlewood GP, Laurie JI, Gilbert HJ (1997) Arabinanase A from Pseudomonas fluorescens subsp. cellulosa exhibits both an endo- and an exo- mode of action. Biochem J 323(2):547–555. https://doi.org/10.1042/bj3230547
Mewis K, Lenfant N, Lombard V, Henrissat B (2016) Dividing the large glycoside hydrolase family 43 into subfamilies: a motivation for detailed enzyme characterization. Appl Environ Microbiol 82(6):1686–1692. https://doi.org/10.1128/AEM.03453-15
Micarda V, Renard CMGC, Colquhoun IJ, Thibault JF (1997) End-products of enzymic saccharification of beet pulp, with a special attention to feruloylated oligosaccharides. Carbohydr Res 32:283–292. https://doi.org/10.1016/S0144-8617(96)00172-5
Mohnen D (2008) Pectin structure and biosynthesis. Curr Opin Plant Biol 11(3):266–277. https://doi.org/10.1016/j.pbi.2008.03.006
Moon JS, Shin SY, Choi HS, Joo W, Cho SK, Li L, Kang JH, Kim TJ, Han NS (2015) In vitro digestion and fermentation properties of linear sugar-beet arabinan and its oligosaccharides. Carbohydr Polym 131:50–56. https://doi.org/10.1016/j.carbpol.2015.05.022
Ohta T, Yamasaki S, Egashira Y, Sanada H (1994) Antioxidative activity of corn bran hemicellulose fragments. J Agric Food Chem 42(3):653–656. https://doi.org/10.1021/jf00039a010
Oosterveld A, Beldman G, Schols HA, Voragen AGJ (1996) Arabinose and ferulic acid rich pectic polysaccharides extracted from sugar beet pulp. Carbohydr Res 288:143–153. https://doi.org/10.1016/S0008-6215(96)90791-0
Ralet MC, Thibault JF, Faulds CB, Williamson G (1994) Isolation and purification of feruloylated oligosaccharides from cell walls of sugarbeet pulp. Carbohydr Res 263(2):227–241. https://doi.org/10.1016/0008-6215(94)00175-8
Ramon D, vd Veen P, Visser J (1993) Arabinan degrading enzymes from Aspergillus nidulans: induction and purification. FEMS Microbiol Lett 113(1):15–22. https://doi.org/10.1111/j.1574-6968.1993.tb06481.x
Santos CR, Polo CC, Costa MC, Nascimento AF, Meza AN, Cota J, Hoffmam ZB, Honorato RV, Oliveira PS, Goldman GH, Gilbert HJ, Prade RA, Ruller R, Squina FM, Wong DW, Murakami MT (2014) Mechanistic strategies for catalysis adopted by evolutionary distinct family 43 arabinanases. J Biol Chem 289(11):7362–7373. https://doi.org/10.1074/jbc.M113.537167
Sato N, Takano Y, Mizuno M, Nozaki K, Umemura S, Matsuzawa T, Amano Y, Makishima S (2013) Production of feruloylated arabino-oligosaccharides (FA-AOs) from beet fiber by hydrothermal treatment. J Supercrit Fluids 79:84–91. https://doi.org/10.1016/j.supflu.2013.01.012
Saulnier L, Thibault JF (1999) Ferulic acid and diferulic acids as components of sugar-beet pectins and maize bran heteroxylans. J Sci Food Agric 79(3):396–402. https://doi.org/10.1002/(SICI)1097-0010(19990301)79:3<396::AID-JSFA262>3.0.CO;2-B
Shi H, Ding H, Huang Y, Wang L, Zhang Y, Li X, Wang F (2014) Expression and characterization of a GH43 endo-arabinanase from Thermotoga thermarum. BMC Biotechnol 14:35. https://doi.org/10.1186/1472-6750-14-35
Squina FM, Prade RA, Wang H, Murakami MT (2009) Expression, purification, crystallization and preliminary crystallographic analysis of an endo-1,5-alpha-L-arabinanase from hyperthermophilic Thermotoga petrophila. Acta Crystallogr Sect F Struct Biol Cryst Commun 65(9):902–905. https://doi.org/10.1107/S1744309109029844
Squina FM, Santos CR, Ribeiro DA, Cota J, de Oliveira RR, Ruller R, Mort A, Murakami MT, Prade RA (2010) Substrate cleavage pattern, biophysical characterization and low-resolution structure of a novel hyperthermostable arabinanase from Thermotoga petrophila. Biochem Biophys Res Commun 399(4):505–511. https://doi.org/10.1016/j.bbrc.2010.07.097
Takao M, Akiyama K, Sakai T (2002a) Purification and characterization of thermostable endo-1,5-alpha-L-arabinase from a strain of Bacillus thermodenitrificans. Appl Environ Microbiol 68(4):1639–1646. https://doi.org/10.1128/AEM.68.4.1639-1646.2002
Takao M, Yamaguchi A, Yoshikawa K, Terashita T, Sakai T (2002b) Molecular cloning of the gene encoding thermostable endo-1,5-alpha-L-arabinase of Bacillus thermodenitrificans TS-3 and its expression in Bacillus subtilis. Biosci Biotechnol Biochem 66(2):430–433. https://doi.org/10.1271/bbb.66.430
Vigsnaes LK, Holck J, Meyer AS, Licht TR (2011) In vitro fermentation of sugar beet arabino-oligosaccharides by fecal microbiota obtained from patients with ulcerative colitis to selectively stimulate the growth of Bifidobacterium spp. and Lactobacillus spp. Appl Environ Microbiol 77(23):8336–8344. https://doi.org/10.1128/AEM.05895-11
Wang H, Li Z, Liu H, Li S, Qiu H, Li K, Luo X, Song Y, Wang N, He H, Zhou H, Ma W, Zhang T (2017) Heterologous expression in Pichia pastoris and characterization of a novel GH11 xylanase from saline-alkali soil with excellent tolerance to high pH, high salt concentrations and ethanol. Protein Expr Purif 139:71–77. https://doi.org/10.1016/j.pep.2017.06.003
Wittchen ES, Haskins J, Stevenson BR (1999) Protein interactions at the tight junction - actin has multiple binding partners, and ZO-1 forms independent complexes with ZO-2 and ZO-3. J Biol Chem 274(49):35179–35185. https://doi.org/10.1074/jbc.274.49.35179
Yamaguchi A, Tada T, Wada K, Nakaniwa T, Kitatani T, Sogabe Y, Takao M, Sakai T, Nishimura K (2005) Structural basis for thermostability of endo-1,5-alpha-L-arabinanase from Bacillus thermodenitrificans TS-3. J Biochem 137(5):587–592. https://doi.org/10.1093/jb/mvi078
Yapo BM, Robert C, Etienne I, Wathelet B, Paquot M (2007) Effect of extraction conditions on the yield, purity and surface properties of sugar beet pulp pectin extracts. Food Chem 100:1356–1364. https://doi.org/10.1016/j.foodchem.2005.12.012
Yeoman CJ, Han Y, Dodd D, Schroeder CM, Mackie RI, Cann IK (2010) Thermostable enzymes as biocatalysts in the biofuel industry. Adv Appl Microbiol 70:1–55. https://doi.org/10.1016/S0065-2164(10)70001-0
Yu KM, Yiu-Nam Lau J, Fok M, Yeung YK, Fok SP, Shek F, Wong WT, Choo QL (2018) Efficient expression and isolation of recombinant human interleukin-11 (rhIL-11) in Pichia pastoris. Protein Expr Purif 146:69–77. https://doi.org/10.1016/j.pep.2018.01.012
Zhang P, Yuan X, Du Y, Li JJ (2018a) Heterologous expression and biochemical characterization of a GHF9 endoglucanase from the termite Reticulitermes speratus in Pichia pastoris. BMC Biotechnol 18(1):35. https://doi.org/10.1186/s12896-018-0432-3
Zhang X, Jiang A, Qi B, Yu H, Xiong Y, Zhou G, Qin M, Dou J, Wang J (2018b) Secretion expression of human neutrophil peptide 1 (HNP1) in Pichia pastoris and its functional analysis against antibiotic-resistant Helicobacter pylori. Appl Microbiol Biotechnol 102(11):4817–4827. https://doi.org/10.1007/s00253-018-8982-9
Funding
This study was funded in part by USDA-NIFA Agriculture and Food Research Initiative Grants (2014-67017-21766 and 2015-67014-22824) and the Arkansas Biosciences Institute at Arkansas State University, the major research component of the Arkansas Tobacco Settlement Proceeds Act of 2000.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1021 kb)
Rights and permissions
About this article
Cite this article
Zhang, N., Wright, T., Wang, X. et al. Production of thermostable endo-1,5-α-l-arabinanase in Pichia pastoris for enzymatically releasing functional oligosaccharides from sugar beet pulp. Appl Microbiol Biotechnol 104, 1595–1607 (2020). https://doi.org/10.1007/s00253-019-10238-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-10238-x