Abstract
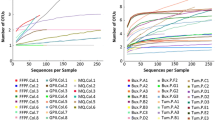
The microbial communities associated with insects play critical roles in many physiological functions such as digestion, nutrition, and defense. Meanwhile, with the development of sequencing technology, more and more studies begin to focus on broader biodiversity of insects and the corresponding mechanisms of insect microbial symbiosis, which need longer time collecting in the field. However, few studies have evaluated the effect of insect microbiome sample preservation approaches especially in different time durations or have assessed whether these approaches are appropriate for both next-generation sequencing (NGS) and third-generation sequencing (TGS) technologies. Here, we used Tessaratoma papillosa (Hemiptera: Tessaratomidae), an important litchi pest, as the model insect and adopted two sequencing technologies to evaluate the effect of four different preservation approaches (cetyltrimethylammonium bromide (CTAB), ethanol, air dried, and RNAlater). We found the samples treated by air dried method, which entomologists adopted for morphological observation and classical taxonomy, would get worse soon. RNAlater as the most expensive approaches for insect microbiome sample preservation did not suit for field works longer than 1 month. We recommended CTAB and ethanol as better preservatives in longer time field work for their effectiveness and low cost. Comparing with the full-length 16S rRNA gene sequenced by TGS, the V4 region of 16S rRNA gene sequenced by NGS has a lower resolution trait and may misestimate the composition of microbial communities. Our results provided recommendations for suitable preservation approaches applied to insect microbiome studies based on two sequencing technologies, which can help researchers properly preserve samples in field works.





Similar content being viewed by others
Data Availability
These sequence data have been submitted to the GenBank databases under accession number PRJNA676180.
References
Zilber-Rosenberg I, Rosenberg E (2008) Role of microorganisms in the evolution of animals and plants: the hologenome theory of evolution. FEMS Microbiol Rev 32:723–735. https://doi.org/10.1111/j.1574-6976.2008.00123.x
Lemanceau P, Blouin M, Muller D, Moenne-Loccoz Y (2017) Let the core microbiota be functional. Trends Plant Sci 22:583–595. https://doi.org/10.1016/j.tplants.2017.04.008
Simon JC, Marchesi JR, Mougel C, Selosse MA (2019) Host–microbiota interactions: from holobiont theory to analysis. Microbiome 7:5. https://doi.org/10.1186/s40168-019-0619-4
Van Borm S, Buschinger A, Boomsma JJ, Billen J (2002) Tetraponera ants have gut symbionts related to nitrogen-fixing root-nodule bacteria. P Roy Soc B-Biol Sci 269:2023–2027. https://doi.org/10.1098/rspb.2002.2101
Colman DR, Toolson EC, Takacs-Vesbach CD (2012) Do diet and taxonomy influence insect gut bacterial communities? Mol Ecol 21:5124–5137. https://doi.org/10.1111/j.1365-294X.2012.05752.x
Sudakaran S, Retz F, Kikuchi Y, Kost C, Kaltenpoth M (2015) Evolutionary transition in symbiotic syndromes enabled diversification of phytophagous insects on an imbalanced diet. Isme J 9:2587–2604. https://doi.org/10.1038/ismej.2015.75
Florez LV, Scherlach K, Gaube P, Ross C, Sitte E, Hermes C, Rodrigues A, Hertweck C, Kaltenpoth M (2017) Antibiotic-producing symbionts dynamically transition between plant pathogenicity and insect-defensive mutualism. Nat Commun 8:15172. https://doi.org/10.1038/ncomms15172
Liu N, Li HJ, Chevrette MG, Zhang L, Cao L, Zhou HK, Zhou XG, Zhou ZH, Pope PB, Currie CR, Huang YP, Wang Q (2019) Functional metagenomics reveals abundant polysaccharide-degrading gene clusters and cellobiose utilization pathways within gut microbiota of a wood-feeding higher termite. Isme J 13:104–117. https://doi.org/10.1038/s41396-018-0255-1
Sudakaran S, Kost C, Kaltenpoth M (2017) Symbiont acquisition and replacement as a source of ecological innovation. Trends Microbiol 25:375–390. https://doi.org/10.1016/j.tim.2017.02.014
Mao M, Bennett GM (2020) Symbiont replacements reset the co-evolutionary relationship between insects and their heritable bacteria. Isme J 14:1384–1395. https://doi.org/10.1038/s41396-020-0616-4
Yao ZC, Ma QK, Cai ZH, Raza MF, Bai S, Wang YC, Zhang P, Ma HQ, Zhang HY (2019) Similar shift patterns in gut bacterial and fungal communities across the life stages of Bactrocera minax larvae from two field populations. Front Microbiol 10:2262. https://doi.org/10.3389/fmicb.2019.02262
Shukla SP, Plata C, Reichelt M, Steiger S, Heckel DG, Kaltenpoth M, Vilcinskas A, Vogel H (2018) Microbiome-assisted carrion preservation aids larval development in a burying beetle. P Natl Acad Sci Usa 115:11274–11279. https://doi.org/10.1073/pnas.1812808115
Vogel H, Shukla SP, Engl T, Weiss B, Fischer R, Steiger S, Heckel DG, Kaltenpoth M, Vilcinskas A (2017) The digestive and defensive basis of carcass utilization by the burying beetle and its microbiota. Nat Commun 8:15186. https://doi.org/10.1038/ncomms15186
Walters W, Hyde ER, Berg-Lyons D, Ackermann G, Humphrey G, Parada A, Gilbert JA, Jansson JK, Caporaso JG, Fuhrman JA, Apprill A, Knight R (2016) Improved bacterial 16S rRNA gene (V4 and V4–5) and fungal internal transcribed spacer marker gene primers for microbial community surveys. MSystems 1:e00009. https://doi.org/10.1128/mSystems.00009-15
Buermans HPJ, den Dunnen JT (2014) Next generation sequencing technology: advances and applications. Bba-Mol Basis Dis 1842:1932–1941. https://doi.org/10.1016/j.bbadis.2014.06.015
Fuks G, Elgart M, Amir A, Zeisel A, Turnbaugh PJ, Soen Y, Shental N (2018) Combining 16S rRNA gene variable regions enables high-resolution microbial community profiling. Microbiome 6:17. https://doi.org/10.1186/s40168-017-0396-x
Deutscher AT, Burke CM, Darling AE, Riegler M, Reynolds OL, Chapman TA (2018) Near full-length 16S rRNA gene next-generation sequencing revealed Asaia as a common midgut bacterium of wild and domesticated Queensland fruit fly larvae. Microbiome 6:85. https://doi.org/10.1186/s40168-018-0463-y
Burke CM, Darling AE (2016) A method for high precision sequencing of near full-length 16S rRNA genes on an Illumina MiSeq. Peerj 4:e2492. https://doi.org/10.7717/peerj.2492
van Dijk EL, Jaszczyszyn Y, Naquin D, Thermes C (2018) The third revolution in sequencing technology. Trends Genet 34:666–681. https://doi.org/10.1016/j.tig.2018.05.008
Ameur A, Kloosterman WP, Hestand MS (2019) Single-molecule sequencing: towards clinical applications. Trends Biotechnol 37:72–85. https://doi.org/10.1016/j.tibtech.2018.07.013
Singer E, Bushnell B, Coleman-Derr D, Bowman B, Bowers RM, Levy A, Gies EA, Cheng JF, Copeland A, Klenk HP, Hallam SJ, Hugenholtz P, Tringe SG, Woyke T (2016) High-resolution phylogenetic microbial community profiling. Isme J 10:2020–2032. https://doi.org/10.1038/ismej.2015.249
Crotti E, Balloi A, Hamdi C, Sansonno L, Marzorati M, Gonella E, Favia G, Cherif A, Bandi C, Alma A, Daffonchio D (2012) Microbial symbionts: a resource for the management of insect-related problems. Microb Biotechnol 5:307–317. https://doi.org/10.1111/j.1751-7915.2011.00312.x
Lee WJ, Brey PT (2013) How microbiomes influence metazoan development: insights from history and Drosophila modeling of gut–microbe interactions. Annu Rev Cell Dev Bi 29:571–592. https://doi.org/10.1146/annurev-cellbio-101512-122333
Raymann K, Moran NA (2018) The role of the gut microbiome in health and disease of adult honey bee workers. Curr Opin Insect Sci 2:97–104. https://doi.org/10.1016/j.cois.2018.02.012
Birer C, Tysklind N, Zinger L, Duplais C (2017) Comparative analysis of DNA extraction methods to study the body surface microbiota of insects: a case study with ant cuticular bacteria. Mol Ecol Resour 17:e34–e45. https://doi.org/10.1111/1755-0998.12688
Ng SH, Stat M, Bunce M, Simmons LW (2018) The influence of diet and environment on the gut microbial community of field crickets. Evol Ecol 8:4704–4720. https://doi.org/10.1002/ece3.3977
Wu GD, Lewis JD, Hoffmann C, Chen YY, Knight R, Bittinger K, Hwang J, Chen J, Berkowsky R, Nessel L, Li H, Bushman FD (2010) Sampling and pyrosequencing methods for characterizing bacterial communities in the human gut using 16S sequence tags. BMC Microbiol 10:206. https://doi.org/10.1186/1471-2180-10-206
Hale VL, Tan CL, Knight R, Amato KR (2015) Effect of preservation method on spider monkey (Ateles geoffroyi) fecal microbiota over 8 weeks. J Microbiol Methods 113:16–26. https://doi.org/10.1016/j.mimet.2015.03.021
Song SJ, Amir A, Metcalf JL, Amato KR, Xu ZZ, Humphrey G, Knight R (2016) Preservation methods differ in fecal microbiome stability, affecting suitability for field studies. Msystems 1:e00021–e00016. https://doi.org/10.1128/mSystems.00021-16
Gorzelak MA, Gill SK, Tasnim N, Ahmadi-Vand Z, Jay M, Gibson DL (2015) Methods for improving human gut microbiome data by reducing variability through sample processing and storage of stool. PLoS One 10:e0134802. https://doi.org/10.1371/journal.pone.0134802
De Cock M, Virgilio M, Vandamme P, Augustinos A, Bourtzis K, Willems A, De Meyer M (2019) Impact of sample preservation and manipulation on insect gut microbiome profiling. A test case with fruit flies (Diptera, Tephritidae). Front Microbiol 10:2833. https://doi.org/10.3389/fmicb.2019.02833
Hammer TJ, Dickerson JC, Fierer N (2015) Evidence-based recommendations on storing and handling specimens for analyses of insect microbiota. Peerj 3:e1190. https://doi.org/10.7717/peerj.1190
Schulte MJ, Martin K, Sauerborn J (2006) Effects of azadirachtin injection in litchi trees (Litchi chinensis Sonn.) on the litchi stink bug (Tessaratoma papillosa Drury) in northern Thailand. J Pest Sci 79:241–250. https://doi.org/10.1007/s10340-006-0142-9
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Huntley J, Fierer N, Owens SM, Betley J, Fraser L, Bauer M, Gormley N, Gilbert JA, Smith G, Knight R (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. Isme J 6:1621–1624. https://doi.org/10.1038/ismej.2012.8
Frank JA, Reich CI, Sharma S, Weisbaum JS, Wilson BA, Olsen GJ (2008) Critical evaluation of two primers commonly used for amplification of bacterial 16S rRNA genes. Appl Environ Microbiol 74:2461–2470. https://doi.org/10.1128/AEM.02272-07
Stackebrandt E, Goodfellow M (1991) Nucleic acid techniques in bacterial systematics. Wiley, Hoboken
Callahan BJ, McMurdie PJ, Rosen MJ, Han AW, Johnson AJA, Holmes SP (2016) DADA2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13:581–583. https://doi.org/10.1038/NMETH.3869
Bolyen E, Rideout JR, Dillon MR, Bokulich N, Abnet CC, Al-Ghalith GA et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Bokulich NA, Kaehler BD, Rideout JR, Dillon M, Bolyen E, Knight R, Huttley GA, Caporaso JG (2018) Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 6:90. https://doi.org/10.1186/s40168-018-0470-z
Ginestet C (2011) ggplot2: Elegant Graphics for Data Analysis. J R Stat Soc A Stat 174:245–245. https://doi.org/10.1111/j.1467-985X.2010.00676_9.x
Kelly BJ, Gross R, Bittinger K, Sherrill-Mix S, Lewis JD, Collman RG, Bushman FD, Li HZ (2015) Power and sample-size estimation for microbiome studies using pairwise distances and PERMANOVA. Bioinformatics 31:2461–2468. https://doi.org/10.1093/bioinformatics/btv183
Duron O, Noel V (2016) A wide diversity of Pantoea lineages are engaged in mutualistic symbiosis and cospeciation processes with stinkbugs. Environ Microbiol Rep 8:715–727. https://doi.org/10.1111/1758-2229.12432
Hosokawa T, Ishii Y, Nikoh N, Fujie M, Satoh N, Fukatsu T (2016) Obligate bacterial mutualists evolving from environmental bacteria in natural insect populations. Nat Microbiol 1:15011. https://doi.org/10.1038/NMICROBIOL.2015.11
Kashkouli M, Castelli M, Floriano AM, Bandi C, Epis S, Fathipour Y, Mehrabadi M, Sassera D (2020) Characterization of a novel Pantoea symbiont allows inference of a pattern of convergent genome reduction in bacteria associated with Pentatomidae. Environ Microbiol 23:36–50. https://doi.org/10.1111/1462-2920.15169
Grimaldi D, Engel MS (2005) Evolution of the insects. Cambridge University Press, Cambridge
Bauer E, Kaltenpoth M, Salem H (2020) Minimal fermentative metabolism fuels extracellular symbiont in a leaf beetle. Isme J 14:866–870. https://doi.org/10.1038/s41396-019-0562-1
Binetruy F, Buysse M, Lejarre Q, Barosi R, Villa M, Rahola N, Paupy C, Ayala D, Duron O (2020) Microbial community structure reveals instability of nutritional symbiosis during the evolutionary radiation of Amblyomma ticks. Mol Ecol 29:1016–1029. https://doi.org/10.1111/mec.15373
Lam TYC, Mei R, Wu ZY, Lee PKH, Liu WT, Lee PH (2020) Superior resolution characterisation of microbial diversity in anaerobic digesters using full-length 16S rRNA gene amplicon sequencing. Water Res 178:115815. https://doi.org/10.1016/j.watres.2020.115815
Brennan CA, Garrett WS (2019) Fusobacterium nucleatum – symbiont, opportunist and oncobacterium. Nat Rev Microbiol 17:156–166. https://doi.org/10.1038/s41579-018-0129-6
Flemming HC, Wuertz S (2019) Bacteria and archaea on Earth and their abundance in biofilms. Nat Rev Microbiol 17:247–260. https://doi.org/10.1038/s41579-019-0158-9
Funding
This work was supported by the National Natural Science Foundation of China (Nos. 31222051 and 31772425) and Science and Technology Program of Guangzhou, China.
Author information
Authors and Affiliations
Contributions
Conceptualization: Q.X.; methodology: Z.-W.Y.; formal analysis and investigation: Z.-W.Y., Y.M., J.Z., Z.-H.L., J.-Y.L., and Y.-H.W.; writing—original draft preparation: Z.-W.Y. and Y.M.; writing—review and editing: Q.X. and W.-J.L.; funding acquisition: Q.X. and Y.-H.W.; supervision: Q.X.
Corresponding authors
Ethics declarations
Ethics approval
Not applicable
Consent to participate
Not applicable
Consent for publication
Not applicable
Conflict of interest
The authors declare that they have no conflicts of interest.
Supplementary Information
ESM 1
(PDF 510 kb)
Rights and permissions
About this article
Cite this article
Yang, ZW., Men, Y., Zhang, J. et al. Evaluation of Sample Preservation Approaches for Better Insect Microbiome Research According to Next-Generation and Third-Generation Sequencing. Microb Ecol 82, 971–980 (2021). https://doi.org/10.1007/s00248-021-01727-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-021-01727-6