Abstract
Background
Complications from liver cirrhosis are a leading cause of death in children with cystic fibrosis. Identifying children at risk for developing liver cirrhosis and halting its progression are critical to reducing liver-associated mortality.
Objective
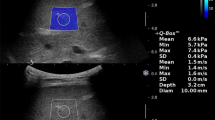
Quantitative US imaging, such as shear-wave elastography (SWE), might improve the detection of liver fibrosis in children with cystic fibrosis (CF) over gray-scale US alone. We incorporated SWE in our pediatric CF liver disease screening program and evaluated its performance using magnetic resonance (MR) elastography.
Materials and methods
Ninety-four children and adolescents with CF underwent 178 SWE exams, aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma-glutamyl transferase (GGT) and platelet measurements. Of these, 27 children underwent 34 MR elastography exams. We evaluated SWE performance using 6-MHz and 9-MHZ point SWE, and 9-MHz two-dimensional (2-D) SWE.
Results
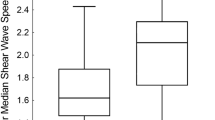
The 6-MHz point SWE was the only method that correlated with MR elastography (r=0.52; 95% confidence interval [CI] 0.20–0.74; P=0.003). SWE of 1.45 m/s distinguished normal from abnormal MR elastography (79% sensitivity, 100% specificity, 100% positive predictive value [PPV], 55% negative predictive value [NPV], area under the receiver operating characteristic [AUROC] curve 0.94). SWE of 1.84 m/s separated mild–moderate (3.00–4.77 kPa) from severe (>4.77 kPa) MR elastography (88% sensitivity, 86% specificity, 78% PPV, 93% NPV, AUROC 0.79). Elevations of AST, ALT, GGT and thrombocytopenia were associated with higher SWE. AST-to-platelet ratio index of 0.42, fibrosis-4 of 0.29, and GGT-to-platelet ratio of 1.43 all had >95% NPV for SWE >1.84 m/s.
Conclusion
Given its correlation with MR elastography, SWE might be a clinically useful predictor of liver fibrosis. We identified imaging criteria delineating the use of SWE to identify increased liver stiffness in children with CF. With multicenter validation, these data might be used to improve the detection and monitoring of liver fibrosis in children with CF.




Similar content being viewed by others
References
FitzSimmons SC (1993) The changing epidemiology of cystic fibrosis. J Pediatr 122:1–9
Cheng K, Ashby D, Smyth RL (2017) Ursodeoxycholic acid for cystic fibrosis-related liver disease. Cochrane Database Syst Rev 9:CD000222
Sokol RJ, Durie PR (1999) Recommendations for management of liver and biliary tract disease in cystic fibrosis. Cystic Fibrosis Foundation Hepatobiliary Disease Consensus Group. J Pediatr Gastroenterol Nutr 28:S1–S13
Debray D, Kelly D, Houwen R et al (2011) Best practice guidance for the diagnosis and management of cystic fibrosis-associated liver disease. J Cyst Fibros 10:S29–S36
Mueller-Abt PR, Frawley KJ, Greer RM, Lewindon PJ (2008) Comparison of ultrasound and biopsy findings in children with cystic fibrosis related liver disease. J Cyst Fibros 7:215–221
Sellers ZM, Lee LW, Barth RA, Milla C (2019) New algorithm for the integration of ultrasound into cystic fibrosis liver disease screening. J Pediatr Gastroenterol Nutr 69:404–410
Lewindon PJ, Puertolas-Lopez MV, Ramm LE et al (2019) Accuracy of transient elastography data combined with APRI in detection and staging of liver disease in pediatric patients with cystic fibrosis. Clin Gastroenterol Hepatol 17:2561–2569.e5
Canas T, Macia A, Munoz-Codoceo RA et al (2015) Hepatic and splenic acoustic radiation force impulse shear wave velocity elastography in children with liver disease associated with cystic fibrosis. Biomed Res Int 2015:517369
Yin M, Glaser KJ, Talwalkar JA et al (2016) Hepatic MR elastography: clinical performance in a series of 1,377 consecutive examinations. Radiology 278:114–124
Trout AT, Sheridan RM, Serai SD et al (2018) Diagnostic performance of MR elastography for liver fibrosis in children and young adults with a spectrum of liver diseases. Radiology 287:824–832
Leung DH, Narkewicz MR (2017) Cystic fibrosis-related cirrhosis. J Cyst Fibros 16:S50–S61
Bedossa P, Poynard T (1996) An algorithm for the grading of activity in chronic hepatitis C. The METAVIR Cooperative Study Group. Hepatology 24:289–293
Yin M, Talwalkar JA, Glaser KJ et al (2007) Assessment of hepatic fibrosis with magnetic resonance elastography. Clin Gastroenterol Hepatol 5:1207–1213
Barr RG, Ferraioli G, Palmeri ML et al (2015) Elastography assessment of liver fibrosis: Society of Radiologists in Ultrasound consensus conference statement. Radiology 276:845–861
Colantonio DA, Kyriakopoulou L, Chan MK et al (2012) Closing the gaps in pediatric laboratory reference intervals: a CALIPER database of 40 biochemical markers in a healthy and multiethnic population of children. Clin Chem 58:854–868
Estey MP, Cohen AH, Colantonio DA et al (2013) CLSI-based transference of the CALIPER database of pediatric reference intervals from Abbott to Beckman, Ortho, Roche and Siemens clinical chemistry assays: direct validation using reference samples from the CALIPER cohort. Clin Biochem 46:1197–1219
Sterling RK, Lissen E, Clumeck N et al (2006) Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 43:1317–1325
Chou R, Wasson N (2013) Blood tests to diagnose fibrosis or cirrhosis in patients with chronic hepatitis C virus infection: a systematic review. Ann Intern Med 158:807–820
Lemoine M, Thursz M, Mallet V, Shimakawa Y (2017) Diagnostic accuracy of the gamma-glutamyl transpeptidase to platelet ratio (GPR) using transient elastography as a reference. Gut 66:195–196
Goksuluk D, Korkmaz S, Zarasiz G, Karaagaoglu AE (2016) easyROC: an interactive web-tool for ROC curve analysis using R language environment. R J 8:213–230
Diedenhofen B, Musch J (2015) Cocor: a comprehensive solution for the statistical comparison of correlations. PLoS One 10:e0121945
Colombo C, Battezzati PM, Crosignani A et al (2002) Liver disease in cystic fibrosis: a prospective study on incidence, risk factors, and outcome. Hepatology 36:1374–1382
Stonebraker JR, Ooi CY, Pace RG et al (2016) Features of severe liver disease with portal hypertension in patients with cystic fibrosis. Clin Gastroenterol Hepatol 14:1207–1215.e3
Boëlle PY, Debray D, Guillot L et al (2019) Cystic fibrosis liver disease: outcomes and risk factors in a large cohort of French patients. Hepatology 69:1648–1656
Bodewes FA, van der Doef HP, Houwen RH, Verkade HJ (2015) Increase of serum gamma-glutamyltransferase associated with development of cirrhotic cystic fibrosis liver disease. J Pediatr Gastroenterol Nutr 61:113–118
Ling SC, Ye W, Leung DH et al (2019) Liver ultrasound patterns in children with cystic fibrosis correlate with non-invasive tests of liver disease. J Pediatr Gastroenterol Nutr 69:351–357
Manco M, Zupone CL, Alghisi F et al (2012) Pilot study on the use of acoustic radiation force impulse imaging in the staging of cystic fibrosis associated liver disease. J Cyst Fibros 11:427–432
Friedrich-Rust M, Wunder K, Kriener S et al (2009) Liver fibrosis in viral hepatitis: noninvasive assessment with acoustic radiation force impulse imaging versus transient elastography. Radiology 252:595–604
Gao S, Peng Y, Guo H et al (2014) Texture analysis and classification of ultrasound liver images. Biomed Mater Eng 24:1209–1216
Poetter-Lang S, Staufer K, Baltzer P et al (2019) The efficacy of MRI in the diagnostic workup of cystic fibrosis-associated liver disease: a clinical observational cohort study. Eur Radiol 29:1048–1058
Hayes D Jr, Krishnamurthy R, Hu HH (2018) Magnetic resonance elastography demonstrates elevated liver stiffness in cystic fibrosis patients. J Cyst Fibros 17:e54–e56
Acknowledgments
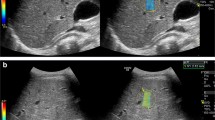
Funding was provided by the Cystic Fibrosis Foundation (SELLER16L0, SELLER19GE0 to Z.M.S), National Institute of Diabetes and Digestive and Kidney Diseases (K08DK124684 to Z.M.S.) and Stanford University (Z.M.S). Dr. Shreyas Vasanawala (Stanford University, Palo Alto, CA) assisted in generating the MR elastography confidence overlay figure.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Online Supplementary Material 1
Scatter plots show the Pearson correlation coefficient of shear-wave elastography (SWE) and magnetic resonance (MR) elastography using the linear 9 L4 probe in point SWE (a) and in 2-D SWE (b) (EPS 182572 kb).
Online Supplementary Material 2
Scatter plots show the median (line) comparing US qualitative patterns with shear-wave elastography (SWE). Significance was determined by comparing nodular to normal, and nodular to echogenic using the Student’s t test. The dotted line represents optimal cut-off as determined by the Youden index (PNG 98 kb).
Online Supplementary Material 3
Scatter plots show the Pearson correlation coefficient of shear-wave elastography (SWE) and magnetic resonance (MR) elastography using the curved 6C1 probe at 6 MHz separated based on body mass index (BMI) percentile (a and b) and age (c and d) (EPS 200324 kb).
Online Supplementary Material 4
Plots depict each patient’s median (dot) and minimum/maximum (whiskers) reported 9-MHz point shear-wave elastography (SWE) (a) and 9-MHz 2-D SWE (b) values, separated by MR elastography score (vertical dotted lines). c Confidence overlay for the image shown in Fig. 3 (EPS 180817 kb).
Online Supplementary Material 6
(DOXC 13.6 kb).
Online Supplementary Material 6
Plots show the Pearson correlation coefficient of shear-wave elastography (SWE) and aspartate aminotransferase (AST)-to-platelet ratio index (APRI) (a), fibrosis-4 (FIB-4) (b), and gamma-glutamyl transferase (GGT)-to-platelet ratio (GPR) (c) (TIF 5303 kb)
Rights and permissions
About this article
Cite this article
Levitte, S., Lee, L.W., Isaacson, J. et al. Clinical use of shear-wave elastography for detecting liver fibrosis in children and adolescents with cystic fibrosis. Pediatr Radiol 51, 1369–1377 (2021). https://doi.org/10.1007/s00247-021-05015-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-021-05015-w