Abstract
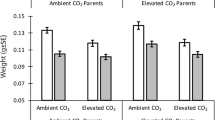
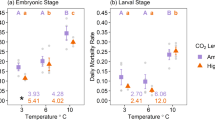
Responses of marine populations to climate conditions reflect the integration of a suite of complex and interrelated physiological and behavioral responses at the individual level. Many of these responses are not immediately reflected in changes to survival, but may impact growth or survival at later life stages. Understanding the broad range of impacts of rising CO2 concentrations on marine fishes is critical to predicting the consequences of ongoing ocean acidification. Walleye pollock (Gadus chalcogrammus) support the largest single-species fishery in the world and provide a critical forage base throughout north Pacific ecosystems. Previous studies of high CO2 effects on early life stages of walleye pollock have suggested a general resiliency in this species, but those studies focused primarily on growth and survival rates. Here, we expand on earlier studies with an independent experiment focused on walleye pollock larval development, swimming behavior, and lipid composition from fertilization to 4 weeks post-hatch at ambient (~ 425 µatm) and elevated (~ 1230 µatm) CO2 levels. Consistent with previous observations, size metrics of walleye pollock were generally insensitive to CO2 treatment. However, 4-week post-hatch larvae had significantly reduced rates of swim bladder inflation. A modest change in the swimming behavior of post-feeding larvae was observed at four, but not at 2 weeks post-hatch. Although there were no differences in overall lipid levels between CO2 treatments, the ratio of energy storage lipids (triacylglycerols) to structural membrane lipids (sterols) was lower among larvae reared at high CO2 levels. Although we observed higher survival to 4 weeks post-hatch among fish reared at high CO2 levels, the observations of reduced swim bladder inflation rates and changes in lipid cycling suggest the presence of sub-lethal effects of acidification that may carry over and manifest in later life stages. These observations support the continued need to evaluate the impacts of ocean acidification on marine fishes across a wide range of traits and life stages with replicated, independent experiments.






Similar content being viewed by others
Data availability
All data from this project will be made publicly available through NOAA’s National Centers for Environmental Information at https://www.nodc.noaa.gov/oads/stewardship/data_assets.html.
References
Ashur MM, Johnston NK, Dixson DL (2017) Impacts of ocean acidification on sensory function in marine organisms. Integr Comp Biol. https://doi.org/10.1093/icb/icx010
Baumann H, Parks EM, Murray CS (2018a) Robust quantification of fish early life history CO2 sensitivities via serial experimentation. Biol Lett. https://doi.org/10.1098/rsbl.20180408
Baumann H, Parks EM, Murray CS (2018b) Starvation rates in larval and juvenile Atlantic silversides (Menidia menidia) are unaffected by high CO2 conditions. Mar Biol 165:75. https://doi.org/10.1007/s00227-018-3335-x
Baumann H, Talmage SC, Gobler CJ (2012) Reduced early life growth and survival in a fish in direct response to increased carbon dioxide. Nat Clim Chang 2:38–41. https://doi.org/10.1038/nclimate1291
Bell MV, Dick JR (1991) Molecular species composition of the major diacyl glycerophospholipids from muscle, liver, retina and brain of cod (Gadus morhua). Lipids 26:565–573. https://doi.org/10.1007/BF02536419
Bignami S, Sponaugle S, Cowen RK (2013) Response to ocean acidification in larvae of a large tropical marine fish, Rachycentron canadum. Glob Chang Biol 19:996–1006. https://doi.org/10.1111/gcb.12133
Boorman GA, Botts S, Bunton TE, Fournie JW, Harshbarger JC, Hawkins WE, Hinton DE, Jokinen MP, Okihiro MS, Wolfe MJ (1997) Diagnostic criteria for degenerative, inflammatory, proliferative nonneoplastic and neoplastic liver lesions in medaka (Oryzias latipes): Consensus of a national toxicology program pathology working group. Toxicol Pathol 25:202–210. https://doi.org/10.1177/019262339702500210
Budge SM, Iverson SJ, Koopman HN (2006) Studying trophic ecology in marine ecosystems using fatty acids: a primer on analysis and interpretation. Mar Mammal Sci 22:759–801. https://doi.org/10.1111/j.1748-7692.2006.00079.x
Caldeira K, Wickett ME (2005) Ocean model predictions of chemistry changes from carbon dioxide emissions to the atmosphere and ocean. J Geophys Res. https://doi.org/10.1029/2004JC002671
Campana SE, Stefánsdóttir RB, Jakobsdóttir K, Sólmundsson J (2020) Shifting fish distributions in warming sub-Arctic oceans. Sci Rep 10:1–14. https://doi.org/10.1038/s41598-020-73444-y
Cattano C, Claudet J, Domenici P, Milazzo M (2018) Living in a high CO2 world: a global meta-analysis shows multiple trait-mediated fish responses to ocean acidification. Ecol Monogr 88:320–335. https://doi.org/10.1002/ecm.1297
Cattano C, Giomi F, Milazzo M (2016) Effects of ocean acidification on embryonic respiration and development of a temperate wrasse living along a natural CO2 gradient. Conserv Physiol. https://doi.org/10.1093/conphys/cov073
Chambers RC, Candelmo AC, Habeck EA, Poach ME, Wieczorek D, Cooper KR, Greenfield CE, Phelan BA (2014) Effects of elevated CO2 in the early life stages of summer flounder, Paralichthys dentatus, and potential consequences of ocean acidification. Biogeosciences 11:1613–1626. https://doi.org/10.5194/bg-11-1613-2014
Checkley DM, Dickson AG, Takahashi M, Radich JA, Eisenkolb N, Asch R (2009) Elevated CO2 enhances otolith growth in young fish. Science 324:1683–1683. https://doi.org/10.1126/science.1169806
Cheung WWL, Frölicher TL (2020) Marine heatwaves exacerbate climate change impacts for fisheries in the northeast Pacific. Sci Rep 10:6678. https://doi.org/10.1038/s41598-020-63650-z
Ciais P, Sabine C, Bala G, Bopp L, Brovkin V, Canadell A, Chhabra A, DeFries R, Galloway J, Heinmann M, Jones C, Le Quéré C, Myneni RB, Piao S, Thornton P (2013) Carbon and other biogeochemical cycles. In: Stocker TF, Quin TF, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate Change 2013: the Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge and New York, pp. 465–570
Clark TD, Raby GD, Roche DG, Binning SA, Speers-Roesch B, Jutfelt F, Sundin J (2020) Ocean acidification does not impair the behaviour of coral reef fishes. Nature 577:370–375. https://doi.org/10.1038/s41586-019-1903-y
Clements JC, Hunt HL (2015) Marine animal behaviour in a high CO2 ocean. Mar Ecol Prog Ser 536:259–279. https://doi.org/10.3354/meps11426
Colton AR, Hurst TP (2010) Behavioral responses to light gradients, olfactory cues, and prey in larvae of two North Pacific gadids (Gadus macrocephalus and Theragra chalcogramma). Environ Biol Fishes 88:39–49. https://doi.org/10.1007/s10641-010-9616-y
Cooley SR, Doney SC (2009) Anticipating ocean acidification’s economic consequences for commercial fisheries. Environ Res Lett. https://doi.org/10.1088/1748-9326/4/2/024007
Copeman LA, Parrish CC, Brown JA, Harel M (2002) Effects of docosahexaenoic, eicosapentaenoic, and arachidonic acids on the early growth, survival, lipid composition and pigmentation of yellowtail flounder (Limanda ferruginea): a live food enrichment experiment. Aquaculture 210:285–304. https://doi.org/10.1016/S0044-8486(01)00849-3
Copeman L, Ryer C, Spencer M, Ottmar M, Iseri P, Sremba A, Wells J, Parrish C (2018) Benthic enrichment by diatom-sourced lipid promotes growth and condition in juvenile Tanner crabs around Kodiak Island, Alaska. Mar Ecol Prog Ser 597:161–178. https://doi.org/10.3354/meps12621
Copeman L, Laurel B (2010) Experimental evidence of fatty acid limited growth and survival in Pacific cod larvae. Mar Ecol Prog Ser 412:259–272. https://doi.org/10.3354/meps08661
Copeman L, Laurel B, Spencer M, Sremba A (2017) Temperature impacts on lipid allocation among juvenile gadid species at the Pacific Arctic-Boreal interface: an experimental laboratory approach. Mar Ecol Prog Ser 566:183–198. https://doi.org/10.3354/meps12040
Crespel A, Zambonino-Infante J-L, Mazurais D, Koumoundouros G, Fragkoulis S, Quazuguel P, Huelvan C, Madec L, Servili A, Claireaux G (2017) The development of contemporary European sea bass larvae (Dicentrarchus labrax) is not affected by projected ocean acidification scenarios. Mar Biol 164:155. https://doi.org/10.1007/s00227-017-3178-x
Cripps IL, Munday PL, McCormick MI (2011) Ocean acidification affects prey detection by a predatory reef fish. PLoS ONE. https://doi.org/10.1371/journal.pone.0022736
Czesny SJ, Graeb BDS, Dettmers JM (2005) Ecological consequences of swim bladder noninflation for larval yellow perch. Trans Am Fish Soc 134:1011–1020. https://doi.org/10.1577/T04-016.1
Davis BE, Miller NA, Flynn EE, Todgham AE (2016) Juvenile Antarctic rockcod (Trematomus bernacchii) are physiologically robust to CO2-acidified seawater. J Exp Biol 219:1203–1213. https://doi.org/10.1242/jeb.133173
Denman K, Christian JR, Steiner N, Po H, Portner H-O, Nojiri Y (2011) Potential impacts of future ocean acidification on marine ecosystems and fisheries: current knowledge and recommendations for future research. ICES J Mar Sci 68:1019–1029. https://doi.org/10.1093/icesjms/fsr074
Díaz-Gil C, Catalán IA, Palmer M, Faulk CK, Fuiman LA (2015) Ocean acidification increases fatty acids levels of larval fish. Biol Lett 11:20150331. https://doi.org/10.1098/rsbl.2015.0331
Dickson AG, Millero FJ (1987) A comparison of the equilibrium constants for the dissociation of carbonic acid in seawater media. Deep Sea Res Part A Oceanogr Res Pap 34:1733–1743. https://doi.org/10.1016/0198-0149(87)90021-5
Ekstrom JA, Suatoni L, Cooley SR, Pendleton LH, Waldbusser GG, Cinner JE, Ritter J, Langdon C, van Hooidonk R, Gledhill D, Wellman K, Beck MW, Brander LM, Rittschof D, Doherty C, Edwards PET, Portela R (2015) Vulnerability and adaptation of US shellfisheries to ocean acidification. Nat Clim Chang 5:207–214. https://doi.org/10.1038/nclimate2508
Fabry VJ, Seibel BA, Feely RA, Orr JC (2008) Impacts of ocean acidification on marine fauna and ecosystem processes. ICES J Mar Sci 65:414–432. https://doi.org/10.1093/icesjms/fsn048
Fabry V, McClintock J, Mathis J, Grebmeier J (2009) Ocean acidification at high latitudes: the bellwether. Oceanography 22:160–171. https://doi.org/10.5670/oceanog.2009.105
Fissel B, Dalton M, Garber-Yonts B, Haynie A, Kasperski S, Lee J, Lew D, Seung C, Sparks K, Szymkowiak M, Wise S (2019) Stock assessment and fishery evaluation report for the groundfish fisheries for the Gulf of Alaska and Bering Sea/Aleutian Islands Area: Economic status of the groundfish fisheries off Alaska p 297
Folch J, Lees M, Sloane Stanley GH (1956) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509. https://doi.org/10.1016/j.athoracsur.2011.06.016
Forsgren E, Dupont S, Jutfelt F, Amundsen T (2013) Elevated CO2 affects embryonic development and larval phototaxis in a temperate marine fish. Ecol Evol 3:3637–3646. https://doi.org/10.1002/ece3.709
Franke A, Clemmesen C (2011) Effect of ocean acidification on early life stages of Atlantic herring (Clupea harengus L.). Biogeosciences Discuss 8:7097–7126. https://doi.org/10.5194/bgd-8-7097-2011
Fraser AJ (1989) Triacylglycerol content as a condition index for fish, bivalve, and crustacean larvae. Can J Fish Aquat Sci 46:1868–1873. https://doi.org/10.1139/f89-235
Frommel AY, Brauner CJ, Allan BJM, Nicol S, Parsons DM, Pether SMJ, Setiawan AN, Smith N, Munday PL (2019) Organ health and development in larval kingfish are unaffected by ocean acidification and warming. PeerJ 2019:e8266. https://doi.org/10.7717/peerj.8266
Frommel AY, Maneja R, Lowe D, Malzahn AM, Geffen AJ, Folkvord A, Piatkowski U, Reusch TBH, Clemmesen C (2012) Severe tissue damage in Atlantic cod larvae under increasing ocean acidification. Nat Clim Chang 2:42–46. https://doi.org/10.1038/nclimate1324
Frommel AY, Maneja R, Lowe D, Pascoe CK, Geffen AJ, Folkvord A, Piatkowski U, Clemmesen C (2014) Organ damage in Atlantic herring larvae as a result of ocean acidification. Ecol Appl 24:1131–1143. https://doi.org/10.1890/13-0297.1
Frommel AY, Margulies D, Wexler JB, Stein MS, Scholey VP, Williamson JE, Bromhead D, Nicol S, Havenhand J (2016) Ocean acidification has lethal and sub-lethal effects on larval development of yellowfin tuna, Thunnus albacares. J Exp Mar Bio Ecol 482:18–24. https://doi.org/10.1016/j.jembe.2016.04.008
Frommel AY, Hermann BT, MIchael, K, Clemmesen C, Hanel R, Reusch TBH (2020) Differential gene expression patterns related to lipid metabolism in larvae and juveniles of Atlantic cod. Comp Biochem Physiol A 247:110740. https://doi.org/10.1016/j.cbpa.2020.110740
Gaichas S, Aydin K, Francis RC (2015) Wasp waist or beer belly? Modeling food web structure and energetic control in Alaskan marine ecosystems, with implications for fishing and environmental forcing. Prog Oceanogr 138:1–17. https://doi.org/10.1016/j.pocean.2015.09.010
Hamilton TJ, Holcombe A, Tresguerres M (2014) CO2-induced ocean acidification increases anxiety in rockfish via alteration of GABA a receptor functioning. Proc R Soc B Biol Sci 281:20132509. https://doi.org/10.1098/rspb.2013.2509
Holmberg RJ, Wilcox-Freeburg E, Rhyne AL, Tlusty MF, Stebbins A, Nye SW, Honig A, Johnston AE, San Antonio CM, Bourque B, Hannigan RE (2019) Ocean acidification alters morphology of all otolith types in Clark’s anemonefish (Amphiprion clarkii). PeerJ. https://doi.org/10.7717/peerj.6152
Holsman KK, Hazen EL, Haynie A, Gourguet S, Hollowed A, Bograd SJ, Samhouri JF, Aydin K, Anderson E (2019) Towards climate resiliency in fisheries management. ICES J Mar Sci 76:1368–1378. https://doi.org/10.1093/icesjms/fsz031
Hurst TP, Copeman LA, Haines SA, Meredith SD, Daniels K, Hubbard KM (2019) Elevated CO2 alters behavior, growth, and lipid composition of Pacific cod larvae. Mar Environ Res 145:52–65. https://doi.org/10.1016/j.marenvres.2019.02.004
Hurst TP, Fernandez ER, Mathis JT (2013) Effects of ocean acidification on hatch size and larval growth of walleye pollock (Theragra chalcogramma). ICES J Mar Sci 70:812–822. https://doi.org/10.1093/icesjms/fst053
Hurst TP, Laurel BJ, Mathis JT, Tobosa LR (2016) Effects of elevated CO2 levels on eggs and larvae of a North Pacific flatfish. ICES J Mar Sci 73:981–990. https://doi.org/10.1093/icesjms/fsv050
Hurst T, Fernandez E, Mathis J, Miller J, Stinson C, Ahgeak E (2012) Resiliency of juvenile walleye pollock to projected levels of ocean acidification. Aquat Biol 17:247–259. https://doi.org/10.3354/ab00483
Jones ER, Simning D, Serafin J, Sepúlveda MS, Griffitt RJ (2020) Acute exposure to oil induces age and species-specific transcriptional responses in embryo-larval estuarine fish. Environ Pollut 263:114325. https://doi.org/10.1016/j.envpol.2020.114325
Kroeker KJ, Kordas RL, Crim RN, Singh GG (2010) Meta-analysis reveals negative yet variable effects of ocean acidification on marine organisms. Ecol Lett 13:1419–1434. https://doi.org/10.1111/j.1461-0248.2010.01518.x
Laurel BJ, Copeman LA, Hurst TP, Parrish CC (2010) The ecological significance of lipid/fatty acid synthesis in developing eggs and newly hatched larvae of Pacific cod (Gadus macrocephalus). Mar Biol 157:1713–1724. https://doi.org/10.1007/s00227-010-1445-1
Laurel BJ, Copeman LA, Iseri P, Spencer ML, Hutchinson G, Nordtug T, Donald CE, Meier S, Allan SE, Boyd DT, Ylitalo GM, Cameron JR, French BL, Linbo TL, Scholz NL, Incardona JP (2019) Embryonic crude oil exposure impairs growth and lipid allocation in a keystone Arctic forage fish. Science 19:1101–1113. https://doi.org/10.1016/j.isci.2019.08.051
Lu Y, Ludsin SA, Fanslow DL, Pothoven SA (2008) Comparison of three microquantity techniques for measuring total lipids in fish. Can J Fish Aquat Sci 65:2233–2241. https://doi.org/10.1139/F08-135
MacPherson JC, Pavlovich JG, Jacobs RS (1998) Phospholipid composition of the granular amebocyte from the horseshoe crab, Limulus polyphemus. Lipids 33:931–940. https://doi.org/10.1007/s11745-998-0290-y
Maneja RH, Frommel AY, Browman HI, Clemmesen C, Geffen AJ, Folkvord A, Piatkowski U, Durif CMF, Bjelland R, Skiftesvik AB (2013) The swimming kinematics of larval Atlantic cod, Gadus morhua L., are resilient to elevated seawater pCO2. Mar Biol 160:1963–1972. https://doi.org/10.1007/s00227-012-2054-y
Mathis JT, Cooley SR, Lucey N, Colt S, Ekstrom J, Hurst T, Hauri C, Evans W, Cross JN, Feely RA (2015) Ocean acidification risk assessment for Alaska’s fishery sector. Prog Oceanogr 136:71–91. https://doi.org/10.1016/j.pocean.2014.07.001
Mathis JT, Cross JN, Bates NR (2011) The role of ocean acidification in systemic carbonate mineral suppression in the Bering Sea. Geophys Res Lett. https://doi.org/10.1029/2011GL048884
Melzner F, Gutowska MA, Langenbuch M, Dupont S, Lucassen M, Thorndyke MC, Bleich M, Pörtner H-O (2009) Physiological basis for high CO2 tolerance in marine ectothermic animals: Pre-adaptation through lifestyle and ontogeny? Biogeosciences 6:2313–2331. https://doi.org/10.5194/bg-6-2313-2009
Miller CB, Morgan CA, Prahl FG, Sparrow MA (1998) Storage lipids of the copepod Calanus Jinmarchicus from Georges Bank and the Gulf of Maine. Limnol Oceanogr 43:488–497. https://doi.org/10.4319/lo.1998.43.3.0488
Munday PL, Dixson DL, McCormick MI, Meekan M, Ferrari MCO, Chivers DP (2010) Replenishment of fish populations is threatened by ocean acidification. Proc Natl Acad Sci U S A 107:12930–12934. https://doi.org/10.1073/pnas.1004519107
Munday P, Gagliano M, Donelson J, Dixson D, Thorrold S (2011) Ocean acidification does not affect the early life history development of a tropical marine fish. Mar Ecol Prog Ser 423:211–221. https://doi.org/10.3354/meps08990
Murray CS, Baumann H (2020) Are long-term growth responses to elevated pCO2 sex-specific in fish? PLoS ONE 15:e0235817. https://doi.org/10.1371/journal.pone.0235817
Murray CS, Fuiman LA, Baumann H (2017) Consequences of elevated CO2 exposure across multiple life stages in a coastal forage fish. ICES J Mar Sci 74:1051–1061. https://doi.org/10.1093/icesjms/fsw179
Murray CS, Wiley D, Baumann H (2019) High sensitivity of a keystone forage fish to elevated CO2 and temperature. Conserv Physiol. https://doi.org/10.1093/conphys/coz084
Nagelkerken I, Munday PL (2016) Animal behaviour shapes the ecological effects of ocean acidification and warming: moving from individual to community-level responses. Glob Chang Biol 22:974–989. https://doi.org/10.1111/gcb.13167
Nilsson GE, Dixson DL, Domenici P, McCormick MI, Sørensen C, Watson S-A, Munday PL (2012) Near-future carbon dioxide levels alter fish behaviour by interfering with neurotransmitter function. Nat Clim Chang 2:201–204. https://doi.org/10.1038/nclimate1352
Parrish CC (1987) Separation of aquatic lipid classes by chromarod thin-layer chromatography with measurement by iatroscan flame ionization detection. Can J Fish Aquat Sci 44:722–731. https://doi.org/10.1139/f87-087
Parrish CC (1988) Dissolved and particulate marine lipid classes: a review. Mar Chem 23:17–40. https://doi.org/10.1016/0304-4203(88)90020-5
Parrish CC (2013) Lipids in Marine Ecosystems. ISRN Oceanogr. https://doi.org/10.5402/2013/604045
Perry AL, Low PJ, Ellis JR, Reynolds JD (2005) Climate change and distribution shifts in marine fishes. Science 308:1912–1915. https://doi.org/10.1126/science.1111322
Pimentel MS, Faleiro F, Dionisio G, Repolho T, Pousao-Ferreira P, Machado J, Rosa R (2014) Defective skeletogenesis and oversized otoliths in fish early stages in a changing ocean. J Exp Biol 217:2062–2070. https://doi.org/10.1242/jeb.092635
Pörtner HO, Langenbuch M, Reipschläger A (2004) Biological impact of elevated ocean CO2 concentrations: lessons from animal physiology and Earth history. J Oceanogr 60:705–718. https://doi.org/10.1007/s10872-004-5763-0
Sanders JL, Peterson TS, Kent ML (2014) Early development and tissue distribution of Pseudoloma neurophilia in the zebrafish, Danio rerio. J Eukaryot Microbiol 61:238–246. https://doi.org/10.1111/jeu.12101
Sargent J, McEvoy L, Estevez A, Bell G, Bell M, Henderson J, Tocher D (1999) Lipid nutrition of marine fish during early development: current status and future directions. Aquaculture 179:217–229. https://doi.org/10.1016/S0044-8486(99)00191-X
Schmidt M, Gerlach G, Leo E, Kunz K, Swoboda S, Pörtner H, Bock C, Storch D (2017) Impact of ocean warming and acidification on the behaviour of two co-occurring gadid species, Boreogadus saida and Gadus morhua, from Svalbard. Mar Ecol Prog Ser 571:183–191. https://doi.org/10.3354/meps12130
Schwebel LN, Stuart K, Lowery MS, Wegner NC (2018) Swim bladder inflation failure affects energy allocation, growth, and feed conversion of California yellowtail (Seriola dorsalis) in aquaculture. Aquaculture 497:117–124. https://doi.org/10.1016/j.aquaculture.2018.07.050
Sørhus E, Incardona JP, Furmanek T, Goetz GW, Scholz NL, Meier S, Edvardsen RB, Jentoft S (2017) Novel adverse outcome pathways revealed by chemical genetics in a developing marine fish. Elife. https://doi.org/10.7554/eLife.20707
Sørhus E, Donald CE, da Silva D, Thorsen A, Karlsen Ø, Meier (2021) Untangling mechanisms of crude oil toxicity: linking gene expression, morphology and PAHs at two developmental stages in a cold water fish. Science of the Total Environ 757:143896. https://doi.org/10.1016/j.scitotenv.2020.143896
Stiasny MH, Sswat M, Mittermayer FH, Falk-Petersen I, Schnell NK, Puvanendran V, Mortensen A, Reusch TBH, Clemmesen C (2019) Divergent responses of Atlantic cod to ocean acidification and food limitation. Glob Chang Biol 25:839–849. https://doi.org/10.1111/gcb.14554
Suthers IM (1998) Bigger? fatter? or is faster growth better? considerations on condition in larval and juvenile coral-reef fish. Austral Ecol 23:265–273. https://doi.org/10.1111/j.1442-9993.1998.tb00730.x
Tresguerres M, Hamilton TJ (2017) Acid–base physiology, neurobiology and behaviour in relation to CO2-induced ocean acidification. J Exp Biol 220:2136–2148. https://doi.org/10.1242/jeb.144113
Williams CR, Dittman AH, McElhany P, Busch DS, Maher MT, Bammler TK, MacDonald JW, Gallagher EP (2019) Elevated CO2 impairs olfactory-mediated neural and behavioral responses and gene expression in ocean-phase coho salmon (Oncorhynchus kisutch). Glob Chang Biol 25:963–977. https://doi.org/10.1111/gcb.14532
Wolf JC, Baumgartner WA, Blazer VS, Camus AC, Engelhardt JA, Fournie JW, Frasca S, Groman DB, Kent ML, Khoo LH, Law JM, Lombardini ED, Ruehl-Fehlert C, Segner HE, Smith SA, Spitsbergen JM, Weber K, Wolfe MJ (2015) Nonlesions, misdiagnoses, missed diagnoses, and other interpretive challenges in fish histopathology studies. Toxicol Pathol 43:297–325. https://doi.org/10.1177/0192623314540229
Wolf JC, Wolfe MJ (2005) A brief overview of nonneoplastic hepatic toxicity in fish. Toxicol Pathol 33:75–85. https://doi.org/10.1080/01926230590890187
Woolley LD, Qin JG (2010) Swimbladder inflation and its implication to the culture of marine finfish larvae. Rev Aquac 2:181–190. https://doi.org/10.1111/j.1753-5131.2010.01035.x
Xu C, Li E, Xu Z, Su Y, Lu M, Qin JG, Chen L, Wang X (2018) Growth and stress axis responses to dietary cholesterol in Nile tilapia (Oreochromis niloticus) in brackish water. Front Physiol 9:254. https://doi.org/10.3389/fphys.2018.00254
Yasumiishi EM, Cieciel K, Andrews AG, Murphy J, Dimond JA (2020) Climate-related changes in the biomass and distribution of small pelagic fishes in the eastern Bering Sea during late summer, 2002–2018. Deep Sea Res Part II Top Stud Oceanogr. https://doi.org/10.1016/j.dsr2.2020.104907
Acknowledgments
The authors thank S. Haines, P. Iseri, and M. Ottmar for assistance with laboratory culture. C. Magel and N. Monacci assisted with the CO2 dosing system and seawater chemical analyses. M. Spencer assisted with larval behavior observations. C. Salant assisted with lipid sample processing and extractions. C. Murray and C. Norrie provided valuable comments on earlier versions of this manuscript. Reference to trade names does not imply endorsement by the National Marine Fisheries Service. The findings and conclusions in this paper are those of the authors and do not necessarily represent the views of the National Marine Fisheries Service.
Funding
This work was supported by a grant to TPH from NOAA’s Ocean Acidification Program.
Author information
Authors and Affiliations
Contributions
TPH, LAC, and MLK designed the experiment. All authors collected the data. TPH, LAC, JFA and MS analyzed the data. TPH led manuscript preparation with contributions from LAC, JFA, and MLK. All authors reviewed, edited, and approved the manuscript for submission.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no conflicts of interest.
Ethical approval
This research was carried out in accordance with all applicable institutional and national guidelines at the time that the study was conducted. Ethical review and approval was not required for the animal study because this experiment was conducted in NOAA’s Alaska Fisheries Science Center Laboratory in Newport, Oregon. NOAA National Marine Fisheries Service does not have an Institutional Animal Care and Use Committee (IACUC) approval processes for research on fishes. All work followed American Fisheries Society policies on the Guidelines for Use of Fishes in Research (https://fisheries.org/docs/policy_useoffishes.pdf) and AVMA (American Veterinary Medical Association) Guidelines on Euthanasia (https://olaw.nih.gov/sites/default/ files/Euthanasia2007.pdf).
Additional information
Responsible Editor: W. Figueira.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewers: H. Baumann and an undisclosed expert
Rights and permissions
About this article
Cite this article
Hurst, T.P., Copeman, L.A., Andrade, J.F. et al. Expanding evaluation of ocean acidification responses in a marine gadid: elevated CO2 impacts development, but not size of larval walleye pollock. Mar Biol 168, 119 (2021). https://doi.org/10.1007/s00227-021-03924-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-021-03924-w