Abstract
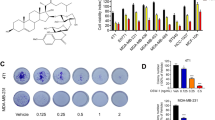
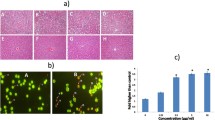
Triple-negative breast cancer is a subtype of breast cancer with poor clinical outcome, and currently, no effective targeted therapies are available. Since cancer develops owing to deregulation of apoptosis, employing therapeutic strategies with the ability to target the molecules involved in apoptosis induction would provide a valid approach to hinder tumor progression. Hydrazide-hydrazones and oxamide molecules are the subject of intense studies due to their anticancer effects via apoptosis induction. In the present study, we attempted to elucidate the mechanism of action of a synthesized compound (compound A) in inducing cell death. Annexin/PI and Western blotting analyses, DAPI staining, mitochondrial membrane potential probe, and flow cytometry were applied for the in vitro evaluations. 4T1 syngeneic mouse model and immunohistochemistry were used for the in vivo assessments. Compound A caused cell death by inducing apoptosis in MDA-MB-231 cells in a mitochondrial-dependent manner at high concentrations after 72 h of incubation. Compound A also impeded tumor growth in a 4T1 syngeneic mouse model as evidenced by hematoxylin and eosin staining of the tumors. Furthermore, it significantly diminished the expression of pro-caspase-3, Ki67, and CD31 markers in the tumor sections. Conclusively, this study for the first time reports the anti-cancer efficacy of compound A in both in vitro and in vivo models and its potential in the treatment of triple-negative breast cancer.







Similar content being viewed by others
Data availability
All relevant data can be found within the manuscript.
Abbreviations
- ANOVA:
-
Analysis of variance
- CCCP:
-
Carbonyl cyanide 3-chlorophenylhydrazone
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- DMEM:
-
Dulbecco’s Modified Eagle’s Medium
- DMSO:
-
Dimethyl sulfoxide
- EDTA:
-
Ethylenediaminetetraacetic acid
- FBS:
-
Fetal bovine serum
- H&E:
-
Hematoxylin and eosin
- HER2:
-
Human epidermal growth factor receptor 2
- IC50 :
-
Inhibitory concentration
- IHC:
-
Immunohistochemistry
- MMP:
-
Mitochondrial membrane potential
- NBF:
-
Neutral buffered formalin
- NCBI:
-
National Cell Bank of Pasture Institute of Iran
- PBS:
-
Phosphate-buffered saline
- PI:
-
Propidium iodide
- PVDF:
-
Polyvinylidene fluoride
- SDS-PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- SEM:
-
Standard error of mean
- TNBC:
-
Triple-negative breast cancer
References
Attanzio A, Ippolito M, Girasolo MA, Saiano F, Rotondo A, Rubino S et al (2018) Anti-cancer activity of di-and tri-organotin (IV) compounds with D-(+)-Galacturonic acid on human tumor cells. J Inorg Biochem 188:102–112. https://doi.org/10.1016/j.jinorgbio.2018.04.006
Barreyro FJ, Holod S, Finocchietto PV, Camino AM, Aquino JB, Avagnina A et al (2015) The pan-caspase inhibitor Emricasan (IDN-6556) decreases liver injury and fibrosis in a murine model of non-alcoholic steatohepatitis. Liver Int 35(3):953–966. https://doi.org/10.1111/liv.12570
Bialvaei AZ, Kafil CHS (2015) Colistin, mechanisms and prevalence of resistance. Med Res Opin 31(4):707–721. https://doi.org/10.1185/03007995.2015.1018989
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424. https://doi.org/10.3322/caac.21492
De Figueiredo LP, Ibiapino AL, Do Amaral DN, Ferraz LS, Rodrigues T, Barreiro EJ et al (2017) Structural characterization and cytotoxicity studies of different forms of a combretastatin A4 analogue. J Mol Struct 1147:226–234. https://doi.org/10.1016/j.molstruc.2017.06.093
Dehbid M, Tahmasvand R, Tasharofi M, Shojaie F, Aghamaali MR, Almasirad A et al (2023) Synthesis of oxamide-hydrazone hybrid derivatives as potential anticancer agents. RPS 18(1):24–38. https://doi.org/10.4103/1735-5362.363593
Dehghani S, Kooshafar Z, Almasirad A, Tahmasvand R, Moayer F, Muhammadnejad A et al (2019) A novel hydrazide compound exerts anti-metastatic effect against breast cancer. Biol Res 52:1–8. https://doi.org/10.1186/s40659-019-0247-2
Do Amaral DN, Cavalcanti BC, Bezerra DP, Ferreira PMP, de Paula CR, Sabino JR et al (2014) Docking, synthesis and antiproliferative activity of N-acylhydrazone derivatives designed as combretastatin A4 analogues. PLoS ONE 9(3):e85380. https://doi.org/10.1371/journal.pone.0085380
Fan C, Su H, Zhao J, Zhao B, Zhang S, Miao J (2010) A novel copper complex of salicylaldehyde pyrazole hydrazone induces apoptosis through up-regulating integrin β4 in H322 lung carcinoma cells. Eur J Med Chem 45(4):1438–1446. https://doi.org/10.1016/j.ejmech.2009.12.048
Fulda S (2015) Targeting apoptosis for anticancer therapy. Sem Cancer Biol 31:84–88. https://doi.org/10.1016/j.semcancer.2014.05.002
He F (2011) BCA (bicinchoninic acid) protein assay. Bio-Protoc 101(5):e44. https://doi.org/10.21769/BioProtoc.44
Iranmanesh M, Mohebbati R, Forouzanfar F, Roshan MK, GhorbaniA Nik MJ et al (2018) In vivo and in vitro effects of ethanolic extract of Trigonella foenum-graecum L. seeds on proliferation, angiogenesis and tube formation of endothelial cells. Res Pharm Sci 13(4):343. https://doi.org/10.4103/1735-5362.235161
Jan R, Chaudhry GES (2019) Understanding apoptosis and apoptotic pathways targeted cancer therapeutics. Adv Pharm Bull 9(2):205–218. https://doi.org/10.15171/apb.2019.024.
Kim H, Cho HJ, Kim SW, Liu B, Choi YJ, Lee J, Sohn YD, Lee MY, Houge MA, Yoon YS (2010) CD31+ cells represent highly angiogenic and vasculogenic cells in bone marrow: novel role of non-endothelial CD31+ cells in neovascularization and their therapeutic effects on ischemic vascular disease. Circ Res 107(5):602–614. https://doi.org/10.1161/CIRCRESAHA.110.218396
Kristiansen A, Grgic M, Altermark B, Leiros I (2014) Properties and distribution of a metallo-β-lactamase (ALI-1) from the fish pathogen Aliivibrio salmonicida LFI1238. J Antimicrob Chemother 70(3):766–772. https://doi.org/10.1093/jac/dku433
Küçükgüzel ŞG, Koç D, Çıkla-Süzgün P, Özsavcı D, Bingöl-Özakpınar Ö, Mega-Tiber P et al (2015) Synthesis of tolmetin hydrazide–hydrazones and discovery of a potent apoptosis inducer in colon cancer cells. Arch Pharm 348(10):730–742. https://doi.org/10.1002/ardp.201500178
Lim B, Greer Y, Lipkowitz S, Takebe N (2019) Novel apoptosis-inducing agents for the treatment of cancer, a new arsenal in the toolbox. Cancers (basel) 11(8):1087. https://doi.org/10.3390/cancers11081087
Luo XL, Lin L, Hu H, Hu FL, Lin Y, Luo ML, Wang L, He YQ (2020) Development and characterization of mammary intraductal (MIND) spontaneous metastasis models for triple-negative breast cancer in syngeneic mice. Sci Rep 10(1):4681. https://doi.org/10.1038/s41598-020-61679-8
Majumder M, Debnath S, Gajbhiye RL, Saikia R, Gogoi B, Samanta SK et al (2019) Fruit extract inhibits migration/invasion, induces apoptosis in breast cancer cells and arrests tumor progression in vivo. Sci Rep 9:1–14. https://doi.org/10.1038/s41598-019-50769-x
Marotti JD, Abreu FB, Wells WA, Tsongalis GJ (2017) Triple-negative breast cancer: next-generation sequencing for target identification. Am J Pathol 187(10):2133–2138. https://doi.org/10.1016/j.ajpath.2017.05.018
McIlwain DR, Berger T, Mak TW (2015) Caspase functions in cell death and disease. Cold Spring Harb Perspect Biol 7(4):a026716. https://doi.org/10.1101/cshperspect.a026716
Michel A, Downey P, Nicolas JM, Scheller D (2014) Unprecedented therapeutic potential with a combination of A2A/NR2B receptor antagonists as observed in the 6-OHDA lesioned rat model of Parkinson’s disease. PLoS ONE 9:e114086. https://doi.org/10.1371/journal.pone.0114086
Mrouj K, Andrés-Sánchez N, Dubra G, Singh P, Sobecki M, Chahar D et al (2021) Ki-67 regulates global gene expression and promotes sequential stages of carcinogenesis. Proc Natl Acad Sci USA 118(10):e2026507118. https://doi.org/10.1073/pnas.2026507118
Pfeffer CM, Singh ATK (2018) Apoptosis: a target for anticancer therapy. Int J Mol Sci 19(2):448. https://doi.org/10.3390/ijms19020448
Pistritto G, Trisciuoglio D , Ceci C , Garufi A, D’Orazi G (2016) Apoptosis as anticancer mechanism: function and dysfunction of its modulators and targeted therapeutic strategies. Aging (Albany NY) 8(4):603–619. https://doi.org/10.18632/aging.100934.
Ramya PS, Guntuku L, Angapelly S, Digwal CS, Lakshmi UJ, Sigalapalli DK et al (2018) Synthesis and biological evaluation of curcumin inspired imidazo [1, 2-a] pyridine analogues as tubulin polymerization inhibitors. Bioorg Chem 143:216–231. https://doi.org/10.1016/j.ejmech.2017.11.010
Rashidi M, Khalilnezhad A, Amani D, Jamshidi H, Muhammadnejad A, Bazi A et al (2018) Umbelliprenin shows antitumor, antiangiogenesis, antimetastatic, anti-inflammatory, and immunostimulatory activities in 4T1 tumor-bearing Balb/c mice. J Cell Physiol 233(11):8908–8918. https://doi.org/10.1002/jcp.26814
Sakamuru S, Attene-Ramos MS, Xia M (2016) Mitochondrial membrane potential assay. Methods Mol Biol 1473:17–22. https://doi.org/10.1007/978-1-4939-6346-1_2
Salucci S, Burattini S, Buontempo F, Orsini E, Furiassi L, Mari M et al (2018) Marine bisindole alkaloid: a potential apoptotic inducer in human cancer cells. Eur J Histochem. 62:2881. https://doi.org/10.4081/ejh.2018.2881
Shilpi S, Shivvedi R, Khatri K (2018) Triple Negative Breast Cancer (TNBC): A challenge for current cancer therapy. J Hum Virol Retrovirol. 6(1): 00189. https://doi.org/10.15406/jhvrv.2018.06.00189.
Singhal J, Nagaprashantha L, Chikara S, Awasthi S, Horne D, Singhal SS (2017) 2'-Hydroxyflavanone: a novel strategy for targeting breast cancer. Oncotarget 8(43): 75025–75037. https://doi.org/10.18632/oncotarget.20499.
Sivandzade F, Bhalerao A, Cucullo L (2019) Analysis of the mitochondrial membrane potential using the cationic JC-1 dye as a sensitive fluorescent probe. Bio Protoc 9(1): e3128. https://doi.org/10.21769/BioProtoc.3128.
Song N, Tang Y, Wang Y, Guan X, Yu W, Jiang T, Lu L, Gu Y (2023) A SIRT6 inhibitor, marine-derived pyrrole-pyridinimidazole derivative 8a, suppresses angiogenesis. Mar Drugs 21(10):517. https://doi.org/10.3390/md21100517
Sreenivasulu R, Reddy KT, Sujitha P, Kumar CG, Raju RR (2019) Synthesis, antiproliferative and apoptosis induction potential activities of novel bis (indolyl) hydrazide-hydrazone derivatives. Bioorg Med Chem 27(6):1043–1055. https://doi.org/10.1016/j.bmc.2019.02.002
Tahmasvand R, Bayat P, Vahdaniparast SM, Dehghani S, Kooshafar Z, Khaleghi S et al (2020) Design and synthesis of novel 4-thiazolidinone derivatives with promising anti-breast cancer activity: synthesis, characterization, in vitro and in vivo results. Bioorg Chem 104:104276. https://doi.org/10.1016/j.bioorg.2020.104276
Tomao F, Papa A, Zaccarelli E, Rossi L, Caruso D, Minozzi M et al (2015) Triple-negative breast cancer: new perspectives for targeted therapies. Onco Targets Ther 8:177–193. https://doi.org/10.2147/OTT.S67673
Wang Y, Huang M, Zhou Z, Li H, Ma Z, Sun Ch (2023) Potential of natural flavonoids to target breast cancer angiogenesis (Review). Br J Pharmacol (online ahead of print). https://doi.org/10.1111/bph.16275.
Wolf P (2019) Tumor-specific induction of the intrinsic apoptotic pathway—a new therapeutic option for advanced prostate cancer? Front Oncol 9:590. https://doi.org/10.3389/fonc.2019.00590
Wong RS (2011) Apoptosis in cancer: from pathogenesis to treatment. J Exp Clin Cancer Res 30(1):87. https://doi.org/10.1186/1756-9966-30-87
Yamamoto A, Huang Y, Krajina BA, McBirney M, Doak AE, Qu S, Wang CL, Haffner MC, Cheung KJ (2023) Metastasis from the tumor interior and necrotic core formation are regulated by breast cancer-derived angiopoietin-like 7. Proc Natl Acad Sci U S A 120(10):e2214888120
Zhang J, Dai J, Zheng Q, Guo S, Yu Y, Hu W et al (2020) The fluoro-thiazolylhydrazone compound TSC-3C inhibits triple negative breast cancer (TNBC) cell line activity by promoting apoptosis, regulating the MAPK pathway and inducing mitochondrial dysfunction. Int J Mol Sci 21(3):1038. https://doi.org/10.3390/ijms21031038
Acknowledgements
We would like to thank the Physiology and Pharmacology Department of Pasteur Institute of Iran for kindly supplying the experimental instruments.
Funding
This work was financially supported by Pasteur Institute of Iran.
Author information
Authors and Affiliations
Contributions
RT made contributions to the biological experiments and collective data. SD and ZK contributed to the in vivo experiments. SAEN synthesized the compound. AA designed the structures. MS wrote the manuscript, provided the technical support, and supervised the whole project. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Ethics approval
This study was conducted in accordance with the animal ethics committee of Tehran Medical Sciences, Islamic Azad University (IR.IAU.PS.REC.1398.104).
Consent to participate
NA.
Consent for publication
NA.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tahmasvand, R., Dehghani, S., Kooshafar, Z. et al. In vitro and in vivo activity of a novel oxamide-hydrazone hybrid derivative against triple-negative breast cancer. Naunyn-Schmiedeberg's Arch Pharmacol 397, 5119–5129 (2024). https://doi.org/10.1007/s00210-023-02931-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02931-6