Abstract
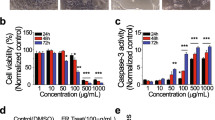
Bartogenic acid (BA), a natural pentacyclic triterpenoid, proved to have chemomodulatory, anticancer, antidiabetic, anti-arthritic, and anti-inflammatory activity. Based on structure–activity relationship (SAR) approaches, BA has close structural resemblance to oleanolic acid and ursolic acid. These two pentacyclic triterpenoids are well accepted with respect to their therapeutic value in various ailments including anti-cancer activity. The aim of this study is to evaluate the efficacy of BA as a possible antitumor agent, along with its safety in SKOV-3 ovarian cancer. In vitro cytotoxicity of BA and paclitaxel on human ovarian cancer cells (SKOV-3) was assessed using MTT assay. Antitumor potential of BA alone, standard anticancer drug (paclitaxel) alone, and BA in combination with paclitaxel were evaluated in SKOV-3 xenografted SCID mice. Immunohistochemical analysis of NF-κB was performed and analyzed in SKOV-3 tumors. BA alone and BA in combination with paclitaxel significantly inhibited the tumor growth. IC50 of BA was found to be 15.72 μM. Similarly, paclitaxel showed significant antitumor effect with IC50 of 3.234 μM. Treatments of paclitaxel, BA, and combination of BA with paclitaxel were well tolerated during treatment period. Immunohistochemical analysis of NF-κB in SKOV-3 tumors treated with BA in combination with paclitaxel revealed antitumor effect in terms of inhibition of NF-κB. Our results suggested that BA exhibits promising antitumor effect in the restriction of SKOV-3 cells and tumors with considerable safety.







Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
Change history
21 September 2021
A Correction to this paper has been published: https://doi.org/10.1007/s00210-021-02150-x
References
Akhtar MF, Saleem A, Rasul A, Faran Ashraf Baig MM, Bin-Jumah M, Abdel Daim MM (2020) Anticancer natural medicines: an overview of cell signaling and other targets of anticancer phytochemicals. Eur J Pharmacol. https://doi.org/10.1016/j.ejphar.2020.173488
An JH, Kim GJ, Jo HJ, Lee KJ, Choi JW (2018) Oleanolic acid induces p53-dependent apoptosis via the ERK/JNK/AKT pathway in cancer cell lines in prostatic cancer xenografts in mice. Oncotarget 9(41):26370–26386
Belotti D, Paganoni P, Manenti L, Garofalo A, Marchini S, Taraboletti G, Giavazzi R (2003) Matrix metalloproteinases (MMP9 and MMP2) induce the release of vascular endothelial growth factor (VEGF) by ovarian carcinoma cells: implications for ascites formation. Cancer Res 63(17):5224–5229
Bishayee A, Ahmed S, Brankov N, Perloff M (2011) Triterpenoids as potential agents for the chemoprevention and therapy of breast cancer. Front Biosci 16:980–996
Chiang YC, Chen CA, Chiang CJ, Hsu TH, Lin MC, You SL, Lai M, Cheng WF, Lai MS (2013) Trends in incidence and survival outcome of epithelial ovarian cancer: 30-year national population-based registry in Taiwan. J Gynecol Oncol 4:342–351. https://doi.org/10.3802/jgo.2013.24.4.342
Feng XM, Su XL (2019) Anticancer effect of ursolic acid via mitochondria-dependent pathways (Review). Oncol Lett 17:4761–4767. https://doi.org/10.3892/ol.2019.10171
Ge GB, Zou LW, Dou TY, Wang P, Lei W, Weng ZM, Hou J, Wang DD, Fan YM, Zhang WD, Yang L (2017) Structure activity relationship of pentacyclic triterpenoids as potent and selective inhibitors against human carboxylesterase 1. Front Pharmacol 8:435. https://doi.org/10.3389/fphar.2017.00435
Gowri PM, Tiwari AK, Ali AZ, Rao JM (2007) Inhibition of α-glucosidase and amylase by bartogenic acid isolated from Barringtonia racemosa Roxb. Seeds. Phytother Res 21:796–799. https://doi.org/10.1002/ptr.2176
Gowri PM, Radhakrishnan SV, Basha SJ, Sarma AV, Rao JM (2009) Oleanane-type isomeric triterpenoids from Barringtonia racemosa. J Nat Prod 72(4):791–795. https://doi.org/10.1021/np8007396
Guglielmo GMD, Soares IN, Viana R, Trelford CB, Chan E, Thai B, Cino EA (2020) The synthetic oleanane triterpenoid CDDO-Me binds and inhibits pyruvate kinase M2. Pharmacol Rep 72:631–640. https://doi.org/10.1007/s43440-019-00045-6
Guo W, Xu B, Wang X, Zheng B, Du J, Liu S (2020) The analysis of the anti-tumor mechanism of ursolic acid using connectively map approach in breast cancer cells line MCF-7. Cancer Manag Res 12:3469–3476. https://doi.org/10.2147/CMAR.S241957
Harrington BS, Annunziata CM (2019) NF-κB signaling in ovarian cancer. Cancers 11:1182. https://doi.org/10.3390/cancers11081182
Huang L, Li J, Ye H, Li C, Wang H, Liu B, Zhang Y (2012) Molecular characterization of the pentacyclic triterpenoid biosynthetic pathway in Catharanthus roseus. Planta 236:1571–1581. https://doi.org/10.1007/s00425-012-1712-0
Jäger S, Trojan H, Kopp T, Laszczyk MN, Scheffler A (2009) Pentacyclic triterpene distribution in various plants–rich sources for a new group of multi-potent plant extracts. Molecules 14:2016–2031. https://doi.org/10.3390/molecules14062016
Jo H, Oh JH, Park DW, Lee C, Min CK (2018) Oleanolic acid 3-acetate, a minor element of ginsenosides, induces apoptotic cell death in ovarian carcinoma and endometrial carcinoma cells via the involvement of a reactive oxygen specieseindependent mitochondrial pathway. J Ginseng Res 44:96–104. https://doi.org/10.1016/j.jgr.2018.09.003
Kriplani D, Patel MM (2013) Immunohistochemistry: a diagnostic aid in differentiating primary epithelial ovarian tumors and tumors metastatic to the ovary. South Asian J Cancer 2(4):254–258. https://doi.org/10.4103/2278-330X.119888
Kundu CN, Patil KR, Mohapatra P, Patel HM, Goyal SN, Ojha S, Patil CR (2015) Pentacyclic triterpenoids inhibit IKKβ mediated activation of NF-κB pathway: in silico and in vitro evidences. Plos One 10(5):e0125709. https://doi.org/10.1371/journal.pone.0125709
Leng S, Hao Y, Du D, Xie S, Hong L, Gu H, Zhu X, Zhang J, Fan D, Kung HF (2013) Ursolic acid promotes cancer cell death by inducing Atg5-dependent autophagy. Int J Cancer 133:2781–2790. https://doi.org/10.1002/ijc.28301
Li G, Wang J, Jin M, Jin C, Ye C, Zhou Y, Wang R, Cui H, Zhou W (2020) A new pentacyclic triterpenoid from the leaves of Rhododendron dauricum L. with inhibition of NO production in LPS-induced RAW 264.7 cells. Nat Prod Res 34(23):3313–3319. https://doi.org/10.1080/14786419.2019.1566822
Litaudon M, Jolly C, Callonec CL, Cuong DD, Retailleau P, Nosjean O, Nguyen VH, Pfeiffer B, Boutin JA, Guéritte F (2009) Cytotoxic pentacyclic triterpenoids from Combretum sundaicum and Lantana camara as inhibitors of Bcl-xL/BakBH3 domain peptide interaction. J Nat Prod 72:1314–1320. https://doi.org/10.1021/np900192r
Morsy RA, Khater D, Kasem RF (2017) Immunohistochemical analysis of Nf-κB expression and its relation to apoptosis and proliferation in different odontogenic tumors. Int J Cancer res 13(2):76–83. https://doi.org/10.3923/ijcr.2017.76.83
Meyerholz DK, Gibson-Corley KN, Olivier AK (2013) Principles for valid histopathologic scoring in research. Vet Pathol 50(6):1007–1015. https://doi.org/10.1177/0300985813485099
Ojha S, Patil CR, Sonara BM, Mahajan UB, Patil KR, Patil DD, Jadhav RB, Goyal SN (2016) Chemomodulatory potential of bartogenic acid against DMBA/croton oil induced two-step skin carcinogenesis in mice. J Cancer 7(14):2139–2147. https://doi.org/10.7150/jca.16446
Patil KR, Patil CR (2017) Anti-inflammatory activity of bartogenic acid containing fraction of fruits of Barringtonia racemosa Roxb. in acute and chronic animal models of inflammation. J Tradit Complement Med 7:86–93. https://doi.org/10.1016/j.jtcme.2016.02.001
Patil KR, Patil CR, Jadhav RB, Mahajan VK, Patil PR, Gaikwad PS (2011) Anti-arthritic activity of bartogenic acid isolated from fruits of Barringtonia racemosa Roxb. (Lecythidaceae). Evid Based Complement Alternat Med. https://doi.org/10.1093/ecam/nep148
Patil CR, Kamble SM, Goyal SN (2014) Multifunctional pentacyclic triterpenoids as adjuvants in cancer chemotherapy: a review. RSC Adv 4:33370. https://doi.org/10.1039/C4RA02784A
Paduch R, Kandefer-Szerszeń M (2014) Antitumor and antiviral activity of pentacyclic triterpenes. Mini-Rev Org Chem 11:262–268. https://doi.org/10.2174/1570193X1103140915105240
Rajkumar T, Natarajan A, Ramachandran B, Gopisetty G, Jayavelu S, Sundersingh S (2020) Pioglitazone modulates doxorubicin resistance in a in vivo model of drug resistant osteosarcoma xenograft. Naunyn-Schmiedeberg’s Arch Pharmacol. https://doi.org/10.1007/s00210-020-01982-3
Reid BM, Permuth JB, Sellers TA (2017) Epidemiology of ovarian cancer: a review. Cancer Biol Med 14(1):9–32. https://doi.org/10.20892/j.issn.2095-3941.2016.0084
Patlolla JMR, Rao CV (2012) Triterpenoids for cancer prevention and treatment: current status and future prospects. Curr Pharm Biotechnol 13:147–155. https://doi.org/10.2174/138920112798868719
Shanmugam MK, Nguyen AH, Kumar AP, Tan BKH, Sethi G (2012) Targeted Inhibition of tumor proliferation, survival, and metastasis by pentacyclic triterpenoids: potential role in prevention and therapy of cancer. Cancer Lett 320(2):158–170. https://doi.org/10.1016/j.canlet.2012.02.037
Shi Z, Chen ZS, Yan XJ, Gong LH, Zheng FY, Cheng KJ (2014) Triterpenoids as reversal agents for anticancer drug resistance treatment. Drug Discovery Today 19(4):482–488. https://doi.org/10.1016/j.drudis.2013.07.018
Shishodia S, Majumdar S, Banerjee S, Aggarwal BB (2003) Ursolic acid inhibits nuclear factor-kappaB activation induced by carcinogenic agents through suppression of IkappaBalpha kinase and p65 phosphorylation: correlation with down-regulation of cyclooxygenase 2, matrix metalloproteinase 9, and cyclin D1. Cancer Res 63(15):4375–4383
Shukla S, Shankar E, Fu P, MacLennan GT, Gupta S (2015) Suppression of NF-κB and NF-κB-regulated gene expression by apigenin through IκBα and IKK pathway in TRAMP mice. Plos One 10(9):e0138710. https://doi.org/10.1371/journal.pone.0138710
Thomas TJ, Panikkar B, Subramoniam A, Nair MK, Panikkar KR (2002) Antitumour property and toxicity of Barringtonia racemosa Roxb seed extract in mice. J Ethnopharmacol 82(2–3):223–227. https://doi.org/10.1016/S0378-8741(02)00074-0
Wang Y, Xiang J, Wang J, Ji Y (2018) Downregulation of TGF-β1 suppressed proliferation and increased chemosensitivity of ovarian cancer cells by promoting BRCA1/Smad3 signaling. Biol Res 51:58. https://doi.org/10.1186/s40659-018-0205-4
Wu YJ, Muldoon LL, Dickey DT, Lewin SJ, Varallyay CG, Neuwelt EA (2009) Cyclophosphamide enhances human tumor growth in nude rat xenografted tumor models. Neoplasia 11(2):187–195. https://doi.org/10.1593/neo.81352
Xia Y, Verma IM, Shen S (2014) NF-kB, an active player in human cancers. Cancer Immunol Res 2(9):823–830. https://doi.org/10.1158/2326-6066.CIR-14-0112
Yadav VR, Aggarwal BB, Prasad S, Sung B, Kannappan R (2010) Targeting inflammatory pathways by triterpenoids for prevention and treatment of cancer. Toxins 2:2428–2466. https://doi.org/10.3390/toxins2102428
Yilmaz E, Gul M, Melekoglu R, Koleli I (2018) Immunhistochemical analysis of nuclear factor kappa beta expression in etiopathogenesis of ovarian tumors. Acta Cir Bras 33(7):641–650. https://doi.org/10.1590/s0102-865020180070000009
Yu M, Si L, Wang Y, Wu Y, Yu F, Jiao P, Shi Y, Wang H, Xiao S, Fu G, Tian K, Wang YT, Guo Z, Ye XS, Zhang LH, Zhou D (2014) Discovery of pentacyclic triterpenoids as potential entry inhibitors of influenza viruses. J Med Chem. https://doi.org/10.1021/jm5014067
Zhang Y, Luo G, Li M, Guo P, Xiao Y, Ji H, Hao Y (2019) Global patterns and trends in ovarian cancer incidence: age, period and birth cohort analysis. BMC Cancer 19:984–998. https://doi.org/10.1186/s12885-019-6139-6
Zhao YC, Zhang L, Feng SS, Hong L, Zheng HL, Chen LL, Zheng XL, Ye YQ, Zhao MD, Wang WX, Zheng CH (2016) Efficient delivery of Notch1 siRNA to SKOV3 cells by cationic cholesterol derivative-based liposome. Int J Nanomedicine 20(11):5485–5496. https://doi.org/10.2147/IJN.S115367
Zheng J (1998) Immunohistochemistry and immunocytochemistry. Methods Mol Biol 105:307–314. https://doi.org/10.1385/0-89603-491-7:307
Zheng L, Liu J, Zhong J, Wu N, Liu G, Lin X (2014) Oleanolic acid induces protective autophagy in cancer cells through the JNK and mTOR pathways. Oncol Rep 32(2):567–572. https://doi.org/10.3892/or.2014.3239
Acknowledgements
We thank Dr P. Mangala Gowri, IICT, Hyderabad, India, for technical assistance and guidance for executing phyto work.
Author information
Authors and Affiliations
Contributions
The authors declare that all the data were generated in-house and that no paper mill was used. VKD and SKR conceptualized and designed the experiments. VKD and AB performed extraction, isolation, and characterization of bartogenic acid at Dabur Research Foundation. VKD and AB performed in vitro and in vivo experiment at Dabur Research Foundation. VKD and SKR analyzed the data. VKD drafted the manuscript. MJ, ATS, and SKR critically reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants. All animal studies were carried out with prior approval from the Institutional Animal Ethics Committee (IAEC) of Dabur Research Foundation, India. Care of animals complied according to CPCSEA (Committee for the purpose of Control and Supervision of Experiments on Animals) guidelines, Government of India.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised. Figure 7 is now corrected.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dubey, V.K., Budhauliya, A., Jaggi, M. et al. Tumor-suppressing effect of bartogenic acid in ovarian (SKOV-3) xenograft mouse model. Naunyn-Schmiedeberg's Arch Pharmacol 394, 1815–1826 (2021). https://doi.org/10.1007/s00210-021-02088-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-021-02088-0