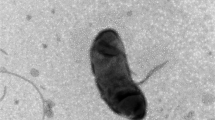
Abstract
A facultative anaerobic nitrogen-fixing bacterium, designated SG131T, was isolated from paddy soil. Strain SG131T showed high 16S rRNA gene sequence similarities with type strains Propionivibrio limicola DSM 6832T (96.9%), Propionivibrio pelophilus asp 66T (96.0%) and Propionivibrio dicarboxylicus DSM 5885T (95.7%). The phylogenetic trees (based on 16S rRNA gene sequences and 120 conserved genes from genomes, respectively) indicated that strain SG131T clustered with members of the genus Propionivibrio. Growth of strain SG131T was observed at 25–40 °C, pH 5.5–10.5 and 0–0.5% (w/v) NaCl. The quinone was Q-7, and the main fatty acids were C16:1 ω6c and/or C16:1 ω7c (25.9%), C16:0 (23.3%), C17:0-cyclo (11.7%), C12:0 (6.0%) and C17:0 (5.9%). The genomic DNA G + C content of strain SG131T was 60.3%. The average nucleotide identity (ANI) values between strain SG131T and its most closely related species P. limicola DSM 6832T, P. pelophilus DSM 12018T and P. dicarboxylicus DSM 5885T were 74.4%, 74.9% and 75.6%, respectively. The digital DNA-DNA hybridization (dDDH) values between strain SG131T and its most closely related species P. limicola DSM 6832T, P. pelophilus DSM 12018T and P. dicarboxylicus DSM 5885T were 19.9%, 20.6% and 20.5%, respectively. All these values were lower than the recommended species delineation thresholds of ANI (95–96%) and dDDH (70%). Strain SG131T possessed core genes (nifHDK) of nitrogen fixation and was confirmed its nitrogen-fixing ability by the ARA method. According to the above-described analysis, strain SG131T represents a novel species of the genus Propionivibrio, for which the name Propionivibrio soli sp. nov. is proposed. The type strain is SG131T (= GDMCC 1.3313T = JCM 35595T).


Similar content being viewed by others
Data availability
The GenBank accession numbers for 16S rRNA gene and genome sequence of strain SG131T are OP345262 and JANZKY000000000, respectively.
Abbreviations
- ANI:
-
Average nucleotide identity
- dDDH:
-
Digital DNA-DNA hybridization
- ARA:
-
Acetylene reduction activity
References
Besemer J, Lomsadze A, Borodovsky M (2001) GeneMarkS: a self-training method for prediction of gene starts in microbial genomes. Implications for finding sequence motifs in regulatory regions. Nucleic Acids Res 29:2607–2618. https://doi.org/10.1093/nar/29.12.2607
Blasco F, Iobbi C, Ratouchniak J, Bonnefoy V, Chippaux M (1990) Nitrate reductases of Escherichia coli: sequence of the second nitrate reductase and comparison with that encoded by the narGHJI operon. Mol Gen Genet 222:104–111. https://doi.org/10.1007/BF00283030
Brune A, Ludwig W, Schink B (2002) Propionivibrio limicola sp. nov., a fermentative bacterium specialized in the degradation of hydroaromatic compounds, reclassification of Propionibacter pelophilus as Propionivibrio pelophilus comb. nov. and amended description of the genus Propionivibrio. Int J Syst Evol Microbiol 52:441–444. https://doi.org/10.1099/00207713-52-2-441
Chor B, Hendy MD, Snir S (2006) Maximum likelihood Jukes-Cantor triplets: analytic solutions. Mol Biol Evol 23:626–632. https://doi.org/10.1093/molbev/msj069
Collins MD, Pirouz T, Goodfellow M, Minnikin DE (1977) Distribution of menaquinones in actinomycetes and corynebacteria. J Gen Microbiol 100:221–230. https://doi.org/10.1099/00221287-100-2-221
Dong ZY, Narsing Rao MP, Wang HF, Fang BZ, Liu YH et al (2019) Transcriptomic analysis of two endophytes involved in enhancing salt stress ability of Arabidopsis thaliana. Sci Total Environ 686:107–117. https://doi.org/10.1016/j.scitotenv.2019.05.483
Dos Santos PC, Fang Z, Mason SW, Setubal JC, Dixon R (2012) Distribution of nitrogen fixation and nitrogenase-like sequences amongst microbial genomes. BMC Genomics 13:162. https://doi.org/10.1186/1471-2164-13-162
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/BF01734359
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Fitch WM (1971) Toward defining the course of evolution: minimum change for a specific tree topology. Syst Zool 20:406–416. https://doi.org/10.1093/sysbio/20.4.406
Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P et al (2007) DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol 57:81–91. https://doi.org/10.1099/ijs.0.64483-0
Graninger M, Nidetzky B, Heinrichs DE, Whitfield C, Messner P (1999) Characterization of dTDP-4-dehydrorhamnose 3,5-epimerase and dTDP-4-dehydrorhamnose reductase, required for dTDP-L-rhamnose biosynthesis in Salmonella enterica serovar Typhimurium LT2. J Biol Chem 274:25069–25077. https://doi.org/10.1074/jbc.274.35.25069
Gregersen T (1978) Rapid method for distinction of Gram-negative from gram-positive bacteria. Eur J Appl Microbiol Biotechnol 5:123–127. https://doi.org/10.1007/BF00498806
Ju XT, Xing GX, Chen XP, Zhang SL, Zhang LJ et al (2009) Reducing environmental risk by improving N management in intensive Chinese agricultural systems. Proc Natl Acad Sci USA 106:3041–3046. https://doi.org/10.1073/pnas.0813417106
Kanehisa M, Goto S, Kawashima S, Okuno Y, Hattori M (2004) The KEGG resource for deciphering the genome. Nucleic Acids Res 32:D277–D280. https://doi.org/10.1093/nar/gkh063
Kanehisa M, Goto S, Hattori M, Aoki-Kinoshita KF, Itoh M et al (2006) From genomics to chemical genomics: new developments in KEGG. Nucleic Acids Res 34:D354–D357. https://doi.org/10.1093/nar/gkj102
Kanehisa M, Sato Y, Kawashima M, Furumichi M, Tanabe M (2016) KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res 44:D457–D462. https://doi.org/10.1093/nar/gkv1070
Kim M, Oh HS, Park SC, Chun J (2014) Towards a taxonomic coherence between average nucleotide identity and 16S rRNA gene sequence similarity for species demarcation of prokaryotes. Int J Syst Evol Microbiol 64:346–351. https://doi.org/10.1099/ijs.0.059774-0
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/BF01731581
Kovacs N (1956) Identification of Pseudomonas pyocyanea by the oxidase reaction. Nature 178:703–704. https://doi.org/10.1038/178703a0
Kroppenstedt RM (1982) Separation of bacterial menaquinones by HPLC using reverse phase (RP18) and a silver loaded ion exchanger as stationary phases. J Liq Chromatogr 5:2359–2367. https://doi.org/10.1080/01483918208067640
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Lagesen K, Hallin P, Rødland EA (2007) RNAmmer: consistent and rapid annotation of ribosomal RNA genes. Nucleic Acids Res 35:3100–3108. https://doi.org/10.1093/nar/gkm160
Li R, Li Y, Kristiansen K (2008) SOAP: short oligonucleotide alignment program. Bioinformatics 24:713–714. https://doi.org/10.1093/bioinformatics/btn025
Liu X, Wang C, Yan B, Lyu L, Takiff HE, Gao Q (2020) The potassium transporter KdpA affects persister formation by regulating ATP levels in Mycobacterium marinum. Emerg Microbes Infect 9:129–139. https://doi.org/10.1080/22221751.2019.1710090
Liu GH, Yang S, Tang R, Xie CJ, Zhou SG (2022) Genome analysis and description of three novel diazotrophs Geomonas species isolated from paddy soils. Front Microbiol 12:801462. https://doi.org/10.3389/fmicb.2021.801462
Lowe TM, Eddy SR (1997) tRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res 25:0955–0964. https://doi.org/10.1093/nar/25.5.955
Meier-Kolthoff JP, Carbasse JS, Peinado-Olarte RL, Göker M (2022) TYGS and LPSN: a database tandem for fast and reliable genome-based classification and nomenclature of prokaryotes. Nucleic Acids Res 50:D801–D807. https://doi.org/10.1093/nar/gkab902
Nakajima A, Aono T, Tsukada S, Siarot L, Ogawa T et al (2012) Lon protease of Azorhizobium caulinodans ORS571 is required for suppression of reb gene expression. Appl Environ Microbiol 78:6251–6261. https://doi.org/10.1128/AEM.01039-12
Narsing Rao MP, Dong ZY, Kan Y, Dong L, Li S et al (2020) Description of Paenibacillus tepidiphilus sp. nov., isolated from a tepid spring. Int J Syst Evol Microbiol 70:1977–1981. https://doi.org/10.1099/ijsem.0.004004
Ogawa K, Akagawa E, Yamane K, Sun ZW, LaCelle M et al (1995) The nasB operon and nasA gene are required for nitrate/nitrite assimilation in Bacillus subtilis. J Bacteriol 177:1409–1413. https://doi.org/10.1128/jb.177.5.1409-1413.1995
Pandey A, Suter H, He J, Hu H, Chen D (2019) Dissimilatory nitrate reduction to ammonium dominates nitrate reduction in long-term low nitrogen fertilized rice paddies. Soil Biol Biochem 131:149–156. https://doi.org/10.1016/j.soilbio.2019.01.007
Parks DH, Chuvochina M, Waite DW, Rinke C, Skarshewski A et al (2018) A standardized bacterial taxonomy based on genome phylogeny substantially revises the tree of life. Nat Biotechnol 36:996–1004. https://doi.org/10.1038/nbt.4229
Parte AC, Sardà Carbasse J, Meier-Kolthoff JP, Reimer LC, Göker M (2020) List of Prokaryotic names with Standing in Nomenclature (LPSN) moves to the DSMZ. Int J Syst Evol Microbiol 70:5607–5612. https://doi.org/10.1099/ijsem.0.004332
Postgate JR (1972) Chapter XIII The acetylene reduction test for nitrogen fixation. Methods Microbiol. Elsevier, Amsterdam, pp 343–356
Priyadarshini P, Choudhury S, Tilgam J, Bharati A, Sreeshma N (2021) Nitrogen fixing cereal: a rising hero towards meeting food security. Plant Physiol Biochem 167:912–920. https://doi.org/10.1016/j.plaphy.2021.09.012
Richter M, Rosselló-Móra R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci USA 106:19126–19131. https://doi.org/10.1073/pnas.0906412106
Rohdich F, Hecht S, Gärtner K, Adam P, Krieger C et al (2002) Studies on the nonmevalonate terpene biosynthetic pathway: metabolic role of IspH (LytB) protein. Proc Natl Acad Sci U S A 99:1158–1163. https://doi.org/10.1073/pnas.032658999
Rzhetsky A, Nei M (1993) Theoretical foundation of the minimum-evolution method of phylogenetic inference. Mol Biol Evol 10:1073–1095. https://doi.org/10.1093/oxfordjournals.molbev.a040056
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Sasser M (1990) Identification of bacteria by gas chromatography of cellular fatty acids. USFCC News 20:16
Tamura K, Nei M (1993) Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol Biol Evol 10:512–526. https://doi.org/10.1093/oxfordjournals.molbev.a040023
Tanaka K, Nakamura K, Mikami E (1990) Fermentation of maleate by a gram-negative strictly anaerobic non-spore former, Propionivibrio dicarboxylicus gen. nov., sp. nov. Arch Microbiol 154:323–328. https://doi.org/10.1099/00207713-41-2-331
Yang Z, Sadakane T, Hosokawa H, Kuroda M, Inoue D et al (2021) Factors affecting antimonate bioreduction by Dechloromonas sp AR-2 and Propionivibrio sp AR-3. 3 Biotech 11:163. https://doi.org/10.1007/s13205-021-02703-0
Yang S, Liu GH, Tang R, Han S, Xie CJ et al (2022) Description of two nitrogen-fixing bacteria, Geomonas fuzhouensis sp. nov and Geomonas agri sp. nov, isolated from paddy soils. Antonie Van Leeuwenhoek 115(3):435–444. https://doi.org/10.1007/s10482-021-01704-6
Yoon SH, Ha SM, Kwon S, Lim J, Kim Y et al (2017a) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67:1613–1617. https://doi.org/10.1099/ijsem.0.001755
Yoon SH, Ha SM, Lim JM, Kwon SJ, Chun J (2017b) A large-scale evaluation of algorithms to calculate average nucleotide identity. Antonie Van Leeuwenhoek 110:1281–1286. https://doi.org/10.1007/s10482-017-0844-4
Acknowledgements
We thank Prof. A. Oren for his kindly help and advice on nomenclature.
Funding
This work was supported by the National Natural Science Foundation of China (grant no: 41925028 and 42277257) and the Natural Science Foundation of Fujian Province (grant no: 2022G02014).
Author information
Authors and Affiliations
Contributions
SGZ and GHL designed the research and project outline. CJX and SY performed deposition and polyphasic taxonomy. RT performed isolation. SH performed genome analysis. CJX drafted the manuscript. SGZ, CR and GHL revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declared that they had no conflict of interest.
Ethical approval
Not applicable.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xie, CJ., Tang, R., Yang, S. et al. A novel nitrogen-fixing bacterium, Propionivibrio soli sp. nov. isolated from paddy soil. Arch Microbiol 205, 68 (2023). https://doi.org/10.1007/s00203-023-03413-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03413-2