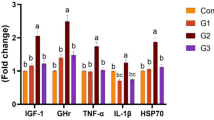
Abstract
Bacillus subtilis (BS) has been used as an excellent probiotic; however, some BS strains seem to be opportunist pathogens or do not present inhibitory effects in the pathogenic bacteria, so the characterization of BS strains for use in animals is mandatory. This study aimed to select nonpathogenic strains of BS, which can inhibit Salmonella spp., avian pathogenic Escherichia coli (APEC), and Campylobacter jejuni (CJ) using a chicken embryo as a model. We tested nine (9) strains of BS isolated from several sources (named A to I) in in vitro by tests of mucin degradation activity, haemolytic activity, apoptosis, and necrosis in fibroblasts from chickens. After the in vitro test, we tested the remaining seven (7) strains (strains A to G) in a chicken embryo (CE) as an in vivo model and target animal. We inoculated 3 log CFU/CE of each strain via allantoic fluid at the 10th day postincubation (DPI). Each treatment group consisted of eight CEs. At the 17th DPI we checked CE mortality, gross lesions, CE weight, and whether BS strains were still viable. To perform the cytokine, total protein, albumin, and reactive C protein analysis, we collected the CE blood from the allantoic vessel and intestine fragments in the duodenum portion for histomorphometric analysis. After the results in CEs, we tested the inhibition capacity of the selected BS strains for diverse strains of Salmonella Heidelberg (SH), S. Typhimurium (ST), S. Enteritidis (SE), S. Minnesota (SM), S. Infantis (SI), Salmonella var. monophasic (SVM), APEC and C. jejuni. After the in vitro trial (mucin degradation activity, haemolytic activity, apoptosis, and necrosis), we removed two (2) strains (H and I) that showed β-haemolysis, mucin degradation, and/or high apoptosis and necrosis effects. Although all strains of BS were viable in CEs at the 17th DPI, we removed four (4) strains (A, B, D, F) once they led to the highest mortality in CEs or a high albumin/protein ratio. C. jejuni inoculated with strain G had greater weight than the commercial strain, which could be further used for egg inoculation with benefits to the CE. From the tests in CEs, we selected the strains C, E, and G for their ability to inhibit pathogenic strains of relevant foodborne pathogens. We found that the inhibition effect was strain dependent. In general, strains E and/or G presented better or similar results than commercial control strains in the inhibition of SH, ST, SI, APEC, and two (2) strains of CJ. In this study, we selected BS strains C, E and G due to their in vitro and in vivo safety and beneficial effects. In addition, we emphasize the value of CE as an in vivo experimental model for assessing BS’s safety and possible benefits for poultry and other animals.




Similar content being viewed by others
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- BS:
-
Bacillus subtilis
- APEC:
-
Avian pathogenic Escherichia coli
- CJ:
-
Campylobacter jejuni
- DPI:
-
Day postincubation
- SH:
-
Salmonella Heidelberg
- ST:
-
Salmonella Typhimurium
- SE:
-
Salmonella Enteritidis
- SM:
-
Salmonella Minnesota
- SI:
-
Salmonella Infantis
- SVM:
-
Salmonella Var. monophasic
- DSM 17299:
-
Commercial probiotic of BS
- CLSI:
-
Clinical and Laboratory Standards Institute
- MIC:
-
Minimal inhibitory concentration
- EFSA:
-
European Food Safety Authority
- NC:
-
Negative control
- PC:
-
Positive control
- YP:
-
Yo Pro-01
- PI:
-
Propide iodate
- DAPI:
-
4′,6-Diamidino-2-phenylindole
References
Al-Sadi R, Ye D, Dokladny K, Ma TY (2008) Mechanism of IL-1β-induced increase in intestinal epithelial tight junction permeability. J Immunol 180:5653–5661
Anadón A, Rosa Martínez-Larrañaga M, Aranzazu Martínez M (2006) Probiotics for animal nutrition in the European Union. Regulation and safety assessment. Regulat Toxicol Pharmacol 45:91–95
Cheng YH, Zhang N, Han JC, Chang CW, Hsiao FSH, Yu YH (2018) Optimization of surfactin production from Bacillus subtilis in fermentation and its effects on Clostridium perfringens-induced necrotic enteritis and growth performance in broilers. J Anim Physiol Anim Nutr (berl) 102:1232–1244
Clinical and Laboratory Standards Institute (2015) Antimicrobial dilution and disk susceptibility testing of infrequently isolated or fastidious bacteria. 3rd ed. CLSI, editor
CLSI (2012) Performance standards for antimicrobial disk susceptibility testing; twenty-second information supplement [Internet]. Clinical and Laboratory Institute;. Available from: www.clsi.org
dela Cruz PJD, Dagaas CT, Mangubat KMM, Angeles AA, Abanto OD (2019) Dietary effects of commercial probiotics on growth performance, digestibility, and intestinal morphometry of broiler chickens. Trop Anim Health Prod 51:1105–15
Coelho-Rocha ND, de Jesus LCL, Barroso FAL et al (2022) Evaluation of Probiotic Properties of Novel Brazilian Lactiplantibacillus plantarum Strains. Probiotics & Antimicro. Prot. https://doi.org/10.1007/s12602-022-09978-6
Danilova I, Sharipova M (2020) The practical potential of bacilli and their enzymes for industrial production. Front Microbiol 4(11):1782. https://doi.org/10.3389/fmicb.2020.01782
de Oliveira JE, van der Hoeven-Hangoor E, van de Linde IB, Montijn RC, van der Vossen JMBM (2014) In ovo inoculation of chicken embryos with probiotic bacteria and its effect on posthatch Salmonella susceptibility. Poult Sci 93:818–829
Earl A, Losick R, Kolter R. Ecology and genomics of Bacillus subtilis. Trends Microbiol [Internet]. 2008;16. Available from: https://doi.org/10.1016/j.tim.2008.03.004
EFSA (2012) Guidance on the assessment of bacterial susceptibility to antimicrobials of human and veterinary importance. EFSA J. https://doi.org/10.2903/j.efsa.2012.2740
EFSA (2018) The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2017. EFSA Journal. Wiley-Blackwell Publishing Ltd, 16
Elisashvili V, Kachlishvili E, Chikindas ML (2019) Recent advances in the physiology of spore formation for bacillus probiotic production. Probiot Antimicrob Proteins 11:731–747
Elshaghabee FMF, Rokana N, Gulhane RD, Sharma C, Panwar H (2017) Bacillus as potential probiotics: status, concerns, and future perspectives. Front Microbiol 8:1–15
FAO/WHO. Guideline for the Evaluation of Probiotics in Food. [Internet]. London,Ontario, Canada.: Food and Agriculture Organization of the United Nations/World Health Organization; 2002 [cited 2022 Aug 26]. Available from: www.who.int/foodsafety/fs_management/en/probiotic_guidelines.pdf
Fonseca BB, Rossi DA, Maia CA, Nalevaiko PC, Melo RT, Cuccato LP, et al (2014) Characterization of the virulence, growth temperature and antibiotic resistance of the Campylobacter jejuni IAL 2383 strain isolated from humans [Internet]. Available from: https://doi.org/10.1590/S1517-83822014000100039
Foster N, Tang Y, Berchieri A, Geng S, Jiao X, Barrow P (2021) Revisiting persistent salmonella infection and the carrier state: what do we know? Pathogens 10(10):1299. https://doi.org/10.3390/pathogens10101299
Fujisawa S, Romin Y, Barlas A, Petrovic LM, Turkekul M, Fan N et al (2014) Evaluation of YO-PRO-1 as an early marker of apoptosis following radiofrequency ablation of colon cancer liver metastases. Cytotechnology 66:259–273
Gray KM, Banada PP, O’Neal E, Bhunia AK (2005) Rapid Ped-2E9 cell-based cytotoxicity analysis and genotyping of Bacillus species. J Clin Microbiol 43:5865–5872
Gueimonde M, Sánchez BG, de Los Reyes-Gavilán C, Margolles A (2013) Antibiotic resistance in probiotic bacteria. Front Microbiol 4:202. https://doi.org/10.3389/fmicb.2013.00202
Hayashi RM, Lourenço MC, Kraieski AL, Araujo RB, Gonzalez-Esquerra R, Leonardecz E et al (2018) Effect of feeding bacillus subtilis spores to broilers challenged with Salmonella enterica serovar Heidelberg Brazilian strain UFPR1 on performance, immune response, and gut health. Front Vet Sci 5:1–12
Heak C, Sukon P, Sornplang P (2018) Effect of direct-fed microbials on culturable gut microbiotas in broiler chickens: a meta-analysis of controlled trials. Asian-Australas J Anim Sci 31:1781–1794
Hwang JY, Park JH (2015) Characteristics of enterotoxin distribution, hemolysis, lecithinase, and starch hydrolysis of Bacillus cereus isolated from infant formulas and ready-to-eat foods. J Dairy Sci 98:1652–1660. https://doi.org/10.3168/jds.2014-9042
Jiang S, Yan FF, Hu JY, Mohammed A, Cheng HW (2021) Bacillus subtilis-based probiotic improves skeletal health and immunity in broiler chickens exposed to heat stress. Animals 11:1–21
Joerger RD, Ganguly A (2017) Current status of the preharvest application of pro- and prebiotics to farm animals to enhance the microbial safety of animal products. Microbiol Spectr 5:1–10
Kathayat D, Lokesh D, Ranjit S, Rajashekara G (2021) Avian pathogenic escherichia coli (Apec): an overview of virulence and pathogenesis factors, zoonotic potential, and control strategies. Pathogens 10(4):467. https://doi.org/10.3390/pathogens10040467
Khochamit N, Siripornadulsil S, Sukon P, Siripornadulsil W (2020) Bacillus subtilis and lactic acid bacteria improve the growth performance and blood parameters and reduce Salmonella infection in broilers. Vet World Veterinary World 13:2663–2672
Knap I, Kehlet AB, Bennedsen M, Mathis GF, Hofacre CL, Lumpkins BS et al (2011) Bacillus subtilis (DSM17299) significantly reduces Salmonella in broilers. Poult Sci 90:1690–1694
Lee NK, Kim WS, Paik HD (2019) Bacillus strains as human probiotics: characterization, safety, microbiome, and probiotic carrier. Food Sci Biotechnol 28:1297–1305
Lumeij JT (1997) Avian clinical biochemistry. In: Kaneko JJ, Harvey JW, Bruss ML (eds) Biochemistry of domestic animals, 5th edn. Academic Press, San Diego, pp 932–932
Mingmongkolchai S, Panbangred W (2018) Bacillus probiotics: an alternative to antibiotics for livestock production. J Appl Microbiol 124:1334–1346
Nishiyama T, Ashida N, Nakagawa K, Iwatani S, Yamamoto N (2021) Dietary bacillus subtilis c-3102 supplementation enhances the exclusion of salmonella enterica from chickens. J Poultry Sci. 58:138–145
O’Reilly EL, Eckersall PD (2014) Acute phase proteins: a review of their function, behaviour and measurement in chickens. Worlds Poult Sci J 27–43
Oakley BB, Kogut MH (2016) Spatial and temporal changes in the broiler chicken cecal and fecal microbiomes and correlations of bacterial taxa with cytokine gene expression. Front Vet Sci 3:11. https://doi.org/10.3389/fvets.2016.00011
Oh JK, Pajarillo EAB, Chae JP, Kim IH, Kang DK (2017) Protective effects of Bacillus subtilis against Salmonella infection in the microbiome of Hy-Line Brown layers. Asian-Australas J Anim Sci 30:1332–1339
Patterson L, Mora E (1964) Occurrence of a substance analogous to C-reactive protein in the blood of the domestic fowl. Rep Biol Med 22:716–721
Patterson LT, Mora E (1965) The C-reactive protein response and disease resistance in the domestic fowl. Tex Rep Biol Med 23(3):600–666
Pender CM, Kim S, Potter TD, Ritzi MM, Young M, Dalloul RA (2017) In ovo supplementation of probiotics and its effects on performance and immune-related gene expression in broiler chicks. Poult Sci 96:1052–1062
Pepys MB, Hirschfield GM (2003) C-reactive protein: a critical update. J Clin Investig 112(2):1805–1812
Ruas-Madiedo P, Gueimonde M, Fernández-García M, de Los Reyes-Gavilán CG, Margolles A (2008) Mucin degradation by Bifidobacterium strains isolated from the human intestinal microbiota. Appl Environ Microbiol 74:1936–1940
Saint-Cyr MJ, Guyard-Nicodème M, Messaoudi S, Chemaly M, Cappelier JM, Dousset X et al (2016) Recent advances in screening of anti-Campylobacter activity in probiotics for use in poultry. Front Microbiol 7:553. https://doi.org/10.3389/fmicb.2016.00553
Smyth D, McKay CM, Gulbransen BD, Phan VC, Wang A, McKay DM (2012) Interferon-gamma signals via an ERK1/2-ARF6 pathway to promote bacterial internalization by gut epithelia. Cell Microbiol 14:1257–1270
Sommerfeld S, Mundim AV, Silva RR, Queiroz JS, Rios MP, Notário FO et al (2022) Physiological changes in chicken embryos inoculated with drugs and viruses highlight the need for more standardization of this animal model. Animals 12(9):1156
Suzuki T, Yoshinaga N, Tanabe S (2011) Interleukin-6 (IL-6) regulates claudin-2 expression and tight junction permeability in intestinal epithelium. J Biol Chem 286:31263–31271
Tang Y, Foster N, Jones MA, Barrow PA (2018) Model of persistent Salmonella infection: Salmonella enterica serovar Pullorum modulates the immune response of the chicken from a Th17-type response towards a Th2-type response Response. Infect Immun 86(8):e00307–18. https://doi.org/10.1128/IAI.00307-18
Tolosa EMC, Rodrigues CJ, Behmer OA, Freitas-Neto AG (2003) Manual de Técnicas Para Histologia Normal e Patológica. 1. edn
Ugwuodo CJ, Nwagu TN (2020) Stabilizing enzymes by immobilization on bacterial spores: a review of literature. Int J Biol Macromol 166:238
Xing JH, Zhao W, Li QY, Yang GL, Zhang RR, Chen HL et al (2021) Bacillus subtilis BSH has a protective effect on Salmonella infection by regulating the intestinal flora structure in chickens. Microb Pathog 155: 104898. https://doi.org/10.1016/j.micpath.2021.104898
Xu L, Yuan J, Chen X, Zhang S, Xie M, Chen C et al (2021) Screening of intestinal probiotics and the effects of feeding probiotics on the digestive enzyme activity, immune, intestinal flora and WSSV resistance of Procambarus clarkii. Aquaculture 540:736748
Yahav S, Berkovich Z, Ostrov I, Reifen R, Shemesh M. Encapsulation of beneficial probiotic bacteria in extracellular matrix from biofilm-forming Bacillus subtilis. Artif Cells Nanomed Biotechnol. Informa UK Limited, trading as Taylor & Francis Group; 2018;46:974–82.
Zeissig S, Burgel N, Gunzel D, Richter J, Mankertz J, Wahnschaffe U et al (2007) Changes in expression and distribution of claudin 2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active Crohn’s disease. Gut 56:61–72
Zhang Y, Chen M, Yu P, Yu S, Wang J, Guo H et al (2020) Prevalence, virulence feature, antibiotic resistance and MLST typing of Bacillus cereus isolated from retail aquatic products in China. Front Microbiol 3(11):1513
Zhou JS, Gopal PK, Gill HS (2001a) Potential probiotic lactic acid bacteria Lactobacillus rhamnosus (HN001), Lactobacillus acidophilus (HN017) and Bifidobacterium lactis (HN019) do not degrade gastric mucin in vitro. Int J Food Microbiol 63:81–90
Zhou JS, Gopal PK, Gill HS (2001b) Potential probiotic lactic acid bacteria Lactobacillus rhamnosus (HN001), Lactobacillus acidophilus (HN017) and Bifidobacterium lactis (HN019) do not degrade gastric mucin in vitro [Internet]. Int J Food Microbiol. Available from: https://doi.org/10.1016/S0168-1605(00)00398-6
Acknowledgements
The authors thank Luiz Ricardo Goulart Filho for all support, research partnership, and teachings. Your departure left us with a vast sadness, but your brilliance and generosity reached all who had the honour to learn from you. You live within us.
Funding
This research was funded by the National Institute of Science and Technology in Theranostics and Nanobiotechnology-INCT-Teranano (CNPq/CAPES/FAPEMIG, Grant # CNPq-403193/2022-2, FAPEMIG Processo nº C.B.B.-APQ-03613-17)).
Author information
Authors and Affiliations
Contributions
BBF, NDC, SS, TFMR, GRS, IMC, DAR, and PGH conducted the in vitro experiment. TFMR, GRS, MCS, AES, SS, and IMC made the collections and analyses in embryos. FON, AAMR, and IPC prepared the histopathological slides and performed the analysis. MSC performed the ELISA. PGH wrote the manuscript. BBF and VAC idealized, designed, analysed the data, and edited the article.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
The Ethics Committee on Animal Use of the U.F.U. certified under protocol number 11/2022/CEUA/PROPP/REITO, PROCESSO N°º23117.023808/2022-77.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
dos Reis, T.F.M., Hoepers, P.G., Azevedo, V.A. et al. Chicken embryos are a valuable model for the selection of Bacillus subtilis for probiotic purposes. Arch Microbiol 204, 715 (2022). https://doi.org/10.1007/s00203-022-03307-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03307-9