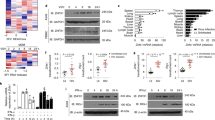
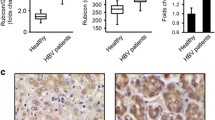
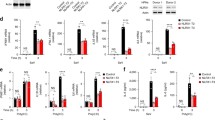
Abstract
DExD/H-box helicase (DDX) 5 belongs to the DExD/H-box helicase family. DDX family members play differential roles in the regulation of innate antiviral immune response. However, whether DDX5 is involved in antiviral immunity remains unclear. In this study, we found that DDX5 serves as a negative regulator of type I interferon (IFN) response. Overexpression of DDX5 inhibited IFN production induced by Spring viremia of carp virus (SVCV) and poly(I:C) and enhanced virus replication by targeting key elements of the RLR signaling pathway (MAVS, MITA, TBK1, IRF3 and IRF7). Mechanistically, DDX5 directly interacted with TBK1 to promote its autophagy-mediated degradation. Moreover, DDX5 was shown to block the interaction between TRAF3 and TBK1, hence preventing nuclear translocation of IRF3. Together, these data shed light on the roles of DDX5 in regulating IFN response.









Similar content being viewed by others
Availability of data and materials
All data generated in this study are included in the article and the Supplementary Material files, and will be made available on request.
References
Takeuchi O, Akira S (2010) Pattern recognition receptors and inflammation. Cell 140(6):805–820. https://doi.org/10.1016/j.cell.2010.01.022
Chen IY, Ichinohe T (2015) Response of host inflammasomes to viral infection. Trends Microbiol 23(1):55–63. https://doi.org/10.1016/j.tim.2014.09.007
Tan XJ, Sun LJ, Chen JQ, Chen ZJJ (2018) Detection of microbial infections through innate immune sensing of nucleic acids. Annu Rev Microbiol 72:447–478. https://doi.org/10.1146/annurev-micro-102215-095605
Schlee M, Hartmann G (2016) Discriminating self from non-self in nucleic acid sensing. Nat Rev Immunol 16(9):566–580. https://doi.org/10.1038/nri.2016.78
Yoneyama M, Kikuchi M, Natsukawa T, Shinobu N, Imaizumi T, Miyagishi M et al (2004) The RNA helicase RIG-I has an essential function in double-stranded RNA-induced innate antiviral responses. Nat Immunol 5(7):730–737. https://doi.org/10.1038/ni1087
Yoneyama M, Kikuchi M, Matsumoto K, Imaizumi T, Miyagishi M, Taira K et al (2005) Shared and unique functions of the DExD/H-box helicases RIG-I, MDA5, and LGP2 in antiviral innate immunity. J Immunol 175(5):2851–2858. https://doi.org/10.4049/jimmunol.175.5.2851
Saito T, Hirai R, Loo YM, Owen D, Johnson CL, Sinha SC et al (2007) Regulation of innate antiviral defenses through a shared repressor domain in RIG-I and LGP2. Proc Natl Acad Sci USA 104(2):582–587. https://doi.org/10.1073/pnas.0606699104
Xu LG, Wang YY, Han KJ, Li LY, Zhai ZH, Shu HB (2005) VISA is an adapter protein required for virus-triggered IFN-β signaling. Mol Cell 19(6):727–740. https://doi.org/10.1016/j.molcel.2005.08.014
Kawai T, Takahashi K, Sato S, Coban C, Kumar H, Kato H et al (2005) IPS-1, an adaptor triggering RIG-I- and Mda5-mediated type I interferon induction. Nat Immunol 6(10):981–988. https://doi.org/10.1038/ni1243
Meylan E, Curran J, Hofmann K, Moradpour D, Binder M, Bartenschlager R et al (2005) Cardif is an adaptor protein in the RIG-I antiviral pathway and is targeted by hepatitis C virus. Nature 437(7062):1167–1172. https://doi.org/10.1038/nature04193
Sato M, Suemori H, Hata N, Asagiri M, Ogasawara K, Nakao K et al (2000) Distinct and essential roles of transcription factors IRF-3 and IRF-7 in response to viruses for IFN-α/β gene induction. Immunity 13(4):539–548. https://doi.org/10.1016/s1074-7613(00)00053-4
Honda K, Takaoka A, Taniguchi T (2006) Type I inteferon gene induction by the interferon regulatory factor family of transcription factors. Immunity 25(3):349–360. https://doi.org/10.1016/j.immuni.2006.08.009
Munz C, Lunemann JD, Getts MT, Miller SD (2009) Antiviral immune responses: triggers of or triggered by autoimmunity? Nat Rev Immunol 9(4):246–258. https://doi.org/10.1038/nri2527
Li S, Lu LF, Li ZC, Zhang C, Zhou XY, Zhou Y et al (2019) Zebrafish MVP recruits and degrades TBK1 to suppress IFN production. J Immunol 202(2):559–566. https://doi.org/10.4049/jimmunol.1801325
Chen LT, Hu MM, Xu ZS, Liu Y, Shu HB (2016) MSX1 modulates RLR-mediated innate antiviral signaling by facilitating assembly of TBK1-associated complexes. J Immunol 197(1):199–207. https://doi.org/10.4049/jimmunol.1600039
Xia Z, Xu G, Nie L, Liu L, Peng N, He Q et al (2019) NAC1 potentiates cellular antiviral signaling by bridging MAVS and TBK1. J Immunol 203(4):1001–1011. https://doi.org/10.4049/jimmunol.1801110
Iggo R, Picksley S, Southgate J, McPheat J, Lane DP (1990) Identification of a putative RNA helicase in E. coli. Nucleic acids Res 18(18):5413–5417. https://doi.org/10.1093/nar/18.18.5413
Iggo RD, Jamieson DJ, MacNeill SA, Southgate J, McPheat J, Lane DP (1991) p68 RNA helicase: identification of a nucleolar form and cloning of related genes containing a conserved intron in yeasts. Mol Cell Biol 11(3):1326–1333. https://doi.org/10.1128/mcb.11.3.1326
Miyashita M, Oshiumi H, Matsumoto M, Seya T (2011) DDX60, a DEXD/H box helicase, is a novel antiviral factor promoting RIG-I-like receptor-mediated signaling. Mol Cell Biol 31(18):3802–3819. https://doi.org/10.1128/mcb.01368-10
Pattabhi S, Knoll ML, Gale M, Loo YM (2019) DHX15 is a coreceptor for RLR signaling that promotes antiviral defense against RNA virus infection. J Interferon Cytokine Res 39(6):331–346. https://doi.org/10.1089/jir.2018.0163
Goncalves A, Bauch A, Stefanovic A, Hantschel O, Bennett KL, Decker T, Superti-Furga G (2008) The DEAD-box helicase DDX3X is a critical component of the TANK-binding kinase 1-dependent innate immune response. EMBO J 27(15):2135–2146. https://doi.org/10.1038/emboj.2008.126
Schroder M, Baran M, Bowie AG (2008) Viral targeting of DEAD box protein 3 reveals its role in TBK1/IKK epsilon-mediated IRF activation. EMBO J 27(15):2147–2157. https://doi.org/10.1038/emboj.2008.143
Gu LL, Fullam A, Brennan R, Schroder M (2013) Human DEAD box helicase 3 couples IκB kinase epsilon to interferon regulatory factor 3 activation. Mol Cell Biol 33(10):2004–2015. https://doi.org/10.1128/mcb.01603-12
Fullam A, Gu LL, Hohn Y, Schroder M (2018) DDX3 directly facilitates IKK α activation and regulates downstream signalling pathways. Biochem J 475:3595–3607. https://doi.org/10.1042/bcj20180163
Zhang KL, Zhang YF, Xue J, Meng QW, Liu HY, Bi CH et al (2019) DDX19 inhibits type I interferon production by disrupting TBK1-IKK epsilon-IRF3 interactions and promoting TBK1 and IKK epsilon degradation. Cell Rep 26(5):1258–1272. https://doi.org/10.1016/j.celrep.2019.01.029
Li D, Fu SZ, Wu ZQ, Yang WP, Ru Y, Shu HB et al (2020) DDX56 inhibits type I interferon by disrupting assembly of IRF3-IPO5 to inhibit IRF3 nucleus import. J Cell Sci 133(4):1. https://doi.org/10.1242/jcs.244681
Ma Z, Moore R, Xu XX, Barber GN (2013) DDX24 negatively regulates cytosolic RNA-mediated innate immune signaling. PLoS Pathog 9(10):13. https://doi.org/10.1371/journal.ppat.1003721
Chen JY, Chen WN, Poon KMV, Zheng BJ, Lin X, Wang YX et al (2009) Interaction between SARS-CoV helicase and a multifunctional cellular protein (Ddx5) revealed by yeast and mammalian cell two-hybrid systems. Arch Virol 154(3):507–512. https://doi.org/10.1007/s00705-009-0323-y
Zhou XX, Luo J, Mills L, Wu SX, Pan T, Geng GN et al (2013) DDX5 facilitates HIV-1 replication as a cellular co-factor of Rev. PLoS One 8(5):12. https://doi.org/10.1371/journal.pone.0065040
Li C, Ge LL, Li PP, Wang Y, Sun MX, Huang L et al (2013) The DEAD-box RNA helicase DDX5 acts as a positive regulator of Japanese encephalitis virus replication by binding to viral 3′ UTR. Antiviral Res 100(2):487–499. https://doi.org/10.1016/j.antiviral.2013.09.002
Goh PY, Tan YJ, Lim SP, Tan YH, Lim SG, Fuller-Pace F et al (2004) Cellular RNA helicase p68 relocalization and interaction with the hepatitis C virus (HCV) NS5B protein and the potential role of p68 in HCV RNA replication. J Virol 78(10):5288–5298. https://doi.org/10.1128/jvi.78.10.5288-5298.2004
Zhang H, Xing Z, Mani SKK, Bancel B, Durantel D, Zoulim F et al (2016) RNA helicase DEAD box protein 5 regulates polycomb repressive complex 2/Hox transcript antisense intergenic RNA function in hepatitis B virus infection and hepatocarcinogenesis. Hepatology 64(4):1033–1048. https://doi.org/10.1002/hep.28698
Zhao S, Ge X, Wang X, Liu A, Guo X, Zhou L et al (2015) The DEAD-box RNA helicase 5 positively regulates the replication of porcine reproductive and respiratory syndrome virus by interacting with viral Nsp9 in vitro. Virus Res 195:217–224. https://doi.org/10.1016/j.virusres.2014.10.021
Kolluru V, Pal D, John A, Ankem MK, Freedman JH, Damodaran C (2017) Induction of Plac8 promotes pro-survival function of autophagy in cadmium-induced prostate carcinogenesis. Cancer Lett 408:121–129. https://doi.org/10.1016/j.canlet.2017.08.023
Ugalde AP, Bretones G, Rodriguez D, Quesada V, Llorente F, Fernandez-Delgado R et al (2022) Autophagy-linked plasma and lysosomal membrane protein PLAC8 is a key host factor for SARS-CoV-2 entry into human cells. Embo J 41(21):22. https://doi.org/10.15252/embj.2022110727
Adamek M, Rakus KŁ, Chyb J, Brogden G, Huebner A, Irnazarow I, Steinhagen D (2012) Interferon type I responses to virus infections in carp cells: in vitro studies on Cyprinid herpesvirus 3 and Rhabdovirus carpio infections. Fish Shellfish Immunol 33(3):482–493. https://doi.org/10.1016/j.fsi.2012.05.031
Winton J, Batts W, deKinkelin P, LeBerre M, Bremont M, Fijan N (2010) Current lineages of the epithelioma papulosum cyprini (EPC) cell line are contaminated with fathead minnow, Pimephales promelas, cells. J Fish Dis 33(8):701–704. https://doi.org/10.1111/j.1365-2761.2010.01165.x
Huang WJ, Ji N, Zhao X, Guo JH, Feng JH, Chen KY, Wu YX, Wang JY, Zou J (2022) RNA-Seq analysis of a zebrafish caudal fin cell line in response to infection with spring viraemia of carp virus. Aquac Fish. https://doi.org/10.1016/j.aaf.2022.11.004
Huang WJ, Zhao X, Ji N, Guo JH, Feng JH, Chen KY et al (2022) IRF2 cooperates with phosphoprotein of spring viremia of carp virus to suppress antiviral response in zebrafish. J Virol 96(22):17. https://doi.org/10.1128/jvi.01314-22
Costes B, Fournier G, Michel B, Delforge C, Raj VS, Dewals B et al (2008) Cloning of the koi herpesvirus genome as an infectious bacterial artificial chromosome demonstrates that disruption of the thymidine kinase locus induces partial attenuation in Cyprinus carpio koi. J Virol 82(10):4955–4964. https://doi.org/10.1128/jvi.00211-08
Sun F, Zhang YB, Liu TK, Shi J, Wang B, Gui JF (2011) Fish MITA serves as a mediator for distinct fish IFN gene activation dependent on IRF3 or IRF7. J Immunol 187(5):2531–2539. https://doi.org/10.4049/jimmunol.1100642
Michel B, Fournier G, Lieffrig F, Costes B, Vanderplasschen A (2010) Cyprinid herpesvirus 3. Emerg Infect Dis 16(12):1835–1843. https://doi.org/10.3201/eid1612.100593
Cheng WY, Chen GH, Jia HJ, He XB, Jing ZZ (2018) DDX5 RNA helicases: emerging roles in viral infection. Int J Mol Sci 19(4):15. https://doi.org/10.3390/ijms19041122
Zhang H, Zhang YQ, Zhu XY, Chen C, Zhang C, Xia YZ et al (2019) DEAD box protein 5 inhibits liver tumorigenesis by stimulating autophagy via interaction with p62/SQSTM1. Hepatology 69(3):1046–1063. https://doi.org/10.1002/hep.30300
Cai B, Wu J, Yu X, Su X-z, Wang R-F (2017) FOSL1 inhibits type I interferon responses to malaria and viral infections by blocking TBK1 and TRAF3/TRIF interactions. MBio. https://doi.org/10.1128/mBio.02161-16
Seth RB, Sun LJ, Ea CK, Chen ZJJ (2005) Identification and characterization of MAVS, a mitochondrial antiviral signaling protein that activates NF-κ B and IRF3. Cell 122(5):669–682. https://doi.org/10.1016/j.cell.2005.08.012
Zhang K, Zhang Y, Xue J, Meng Q, Liu H, Bi C et al (2019) DDX19 inhibits type I interferon production by disrupting TBK1-IKKepsilon-IRF3 interactions and promoting TBK1 and IKKepsilon degradation. Cell Rep 26(5):1258–1272. https://doi.org/10.1016/j.celrep.2019.01.029. (e4)
Zhang ZQ, Kim T, Bao MS, Facchinetti V, Jung SY, Ghaffari AA et al (2011) DDX1, DDX21, and DHX36 helicases form a complex with the adaptor molecule TRIF to sense dsRNA in dendritic cells. Immunity 34(6):866–878. https://doi.org/10.1016/j.immuni.2011.03.027
Zhang ZQ, Yuan B, Bao MS, Lu N, Kim T, Liu YJ (2011) The helicase DDX41 senses intracellular DNA mediated by the adaptor STING in dendritic cells. Nat Immunol 13(2):196. https://doi.org/10.1038/ni0212-196b
Sadler AJ, Latchoumanin O, Hawkes D, Mak J, Williams BRG (2009) An antiviral response directed by PKR phosphorylation of the RNA helicase A. PLoS Pathog 5(2):11. https://doi.org/10.1371/journal.ppat.1000311
Zhao LC, Zhao YZ, Liu QZ, Huang JJ, Lu YL, Ping JH (2022) DDX5/METTL3-METTL14/YTHDF2 axis regulates replication of influenza A virus. Microbiol Spectr 10(3):17. https://doi.org/10.1128/spectrum.01098-22
Xu J, Cai YH, Ma ZB, Jiang B, Liu WX, Cheng J et al (2021) The RNA helicase DDX5 promotes viral infection via regulating N-6-methyladenosine levels on the DHX58 and NFκB transcripts to dampen antiviral innate immunity. PLoS Pathog 17(4):30. https://doi.org/10.1371/journal.ppat.1009530
Rios-Marco P, Romero-Lopez C, Berzal-Herranz A (2016) The cis-acting replication element of the Hepatitis C virus genome recruits host factors that influence viral replication and translation. Sci Rep 6:15. https://doi.org/10.1038/srep25729
Sparrer KMJ, Gableske S, Zurenski MA, Parker ZM, Full F, Baumgart GJ et al (2017) TRIM23 mediates virus-induced autophagy via activation of TBK1. Nat Microbiol 2(11):1543–1557. https://doi.org/10.1038/s41564-017-0017-2
Sun MW, Yu SX, Ge HL, Wang T, Li YF, Zhou PP et al (2022) The A137R protein of African swine fever virus inhibits type I interferon production via the autophagy-mediated lysosomal degradation of TBK1. J Virol 96(9):16. https://doi.org/10.1128/jvi.01957-21
Deng TJ, Hu BL, Wang XB, Lin LL, Zhou JW, Xu YT et al (2021) Inhibition of antiviral innate immunity by avibirnavirus VP3 via blocking TBK1-TRAF3 complex formation and IRF3 activation. MSystems 6(3):17. https://doi.org/10.1128/mSystems.00016-21
Zhou ZH, Sun Y, Xu JY, Tang XY, Zhou L, Li QN et al (2021) Swine acute diarrhea syndrome coronavirus nucleocapsid protein antagonizes interferon-beta production via blocking the interaction between TRAF3 and TBK1. Front Immunol 12:13. https://doi.org/10.3389/fimmu.2021.573078
Zhang WW, Weng JH, Yao L, Jia P, Yi MS, Jia KT (2022) Nectin4 antagonises type I interferon production by targeting TRAF3 for autophagic degradation and disrupting TRAF3-TBK1 complex formation. Int J Biol Macromol 218:654–664. https://doi.org/10.1016/j.ijbiomac.2022.07.151
Cai BW, Wu J, Yu X, Su XZ, Wang RF (2017) FOSL1inhibits type I interferon responses to malaria and viral infections by blocking TBK1 and TRAF3/TRIF interactions. MBio 8(1):14. https://doi.org/10.1128/mBio.02161-16
Sui C, Xiao TY, Zhang SY, Zeng HX, Zheng Y, Liu BY et al (2022) SARS-CoV-2 NSP13 inhibits type I IFN production by degradation of TBK1 via p62-dependent selective autophagy. J Immunol 208(3):753–761. https://doi.org/10.4049/jimmunol.2100684
Acknowledgements
The authors thank Dr Mingxian Chang for providing SVCV.
Funding
This work is funded by the National Natural Science Foundation of China (32030112 and U21A20268).
Author information
Authors and Affiliations
Contributions
YZ: investigation, methodology, data curation, writing original draft. Jing Cen, GY, ZJ, KC, WG, Jing Chen: investigation, methodology. MA, ZJ: methodology, materials, manuscript editing. JZ: conceptualization, funding acquisition, project administration, supervision, review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval and consent to participate
Not applicable.
Consent for publication
All authors have approved this article for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Cen, J., Yuan, G. et al. DDX5 inhibits type I IFN production by promoting degradation of TBK1 and disrupting formation of TBK1 − TRAF3 complex. Cell. Mol. Life Sci. 80, 212 (2023). https://doi.org/10.1007/s00018-023-04860-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-023-04860-2