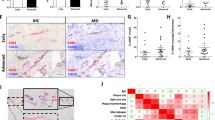
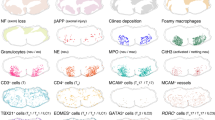
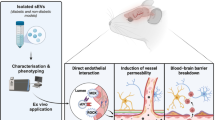
Abstract
Protein Spinster homolog 2 (Spns2) is a sphingosine-1-phosphate (S1P) transporter that releases S1P to regulate lymphocyte egress and trafficking. Global deletion of Spns2 (Spns2−/−) has been shown to reduce disease severity in several autoimmune disease models. To examine whether Spns2 could be exploited as a drug target, we generated and characterized the mice with postnatal knockout of Spns2 (Spns2-Mx1Cre). Our results showed that Spns2-Mx1Cre mice had significantly low number of lymphocytes in blood and lymphoid organs similar to Spns2−/− mice. Lymph but not plasma S1P levels were significantly reduced in both groups of knockout mice. Our lipidomic results also showed that Spns2 releases different S1P species into lymph. Interestingly, lymphatic vessels in the lymph nodes (LNs) of Spns2−/− and Spns2-Mx1Cre mice exhibited morphological defects. The structures of high endothelial venules (HEV) in the LNs of Spns2-Mx1Cre mice were disorganized. These results indicate that lack of Spns2 affects both S1P secretion and LN vasculatures. Nevertheless, blood vasculature of these Spns2 deficient mice was not different to controls under homeostasis and vascular insults. Importantly, Spns2-Mx1Cre mice were resistant to multiple sclerosis in experimental autoimmune encephalomyelitis (EAE) models with significant reduction of pathogenic Th17 cells in the central nervous system (CNS). This study suggests that pharmacological inhibition of Spns2 may be exploited for therapeutic applications in treatment of neuroinflammation.






Similar content being viewed by others
References
Yanagida K, Hla T (2017) Vascular and immunobiology of the circulatory sphingosine 1-phosphate gradient. Annu Rev Physiol 79:67–91. https://doi.org/10.1146/annurev-physiol-021014-071635
Tan ST, Ramesh T, Toh XR, Nguyen LN (2020) Emerging roles of lysophospholipids in health and disease. Prog Lipid Res 80:101068. https://doi.org/10.1016/j.plipres.2020.101068
Chun J, Rosen H (2006) Lysophospholipid receptors as potential drug targets in tissue transplantation and autoimmune diseases. Curr Pharm Des 12(2):161–171
Cyster JG, Schwab SR (2012) Sphingosine-1-phosphate and lymphocyte egress from lymphoid organs. Annu Rev Immunol 30:69–94. https://doi.org/10.1146/annurev-immunol-020711-075011
Maceyka M, Sankala H, Hait NC, Le Stunff H, Liu H, Toman R, Collier C, Zhang M, Satin LS, Merrill AH Jr, Milstien S, Spiegel S (2005) SphK1 and SphK2, sphingosine kinase isoenzymes with opposing functions in sphingolipid metabolism. J Biol Chem 280(44):37118–37129. https://doi.org/10.1074/jbc.M502207200
Fukuhara S, Simmons S, Kawamura S, Inoue A, Orba Y, Tokudome T, Sunden Y, Arai Y, Moriwaki K, Ishida J, Uemura A, Kiyonari H, Abe T, Fukamizu A, Hirashima M, Sawa H, Aoki J, Ishii M, Mochizuki N (2012) The sphingosine-1-phosphate transporter Spns2 expressed on endothelial cells regulates lymphocyte trafficking in mice. J Clin Invest 122(4):1416–1426. https://doi.org/10.1172/JCI60746
Vu TM, Ishizu AN, Foo JC, Toh XR, Zhang F, Whee DM, Torta F, Cazenave-Gassiot A, Matsumura T, Kim S, Toh SES, Suda T, Silver DL, Wenk MR, Nguyen LN (2017) Mfsd2b is essential for the sphingosine-1-phosphate export in erythrocytes and platelets. Nature 550(7677):524–528. https://doi.org/10.1038/nature24053
Tukijan F, Chandrakanthan M, Nguyen LN (2018) The signalling roles of sphingosine-1-phosphate derived from red blood cells and platelets. Br J Pharmacol 175(19):3741–3746. https://doi.org/10.1111/bph.14451
Pappu R, Schwab SR, Cornelissen I, Pereira JP, Regard JB, Xu Y, Camerer E, Zheng YW, Huang Y, Cyster JG, Coughlin SR (2007) Promotion of lymphocyte egress into blood and lymph by distinct sources of sphingosine-1-phosphate. Science 316(5822):295–298. https://doi.org/10.1126/science.1139221
Camerer E, Regard JB, Cornelissen I, Srinivasan Y, Duong DN, Palmer D, Pham TH, Wong JS, Pappu R, Coughlin SR (2009) Sphingosine-1-phosphate in the plasma compartment regulates basal and inflammation-induced vascular leak in mice. J Clin Invest 119(7):1871–1879
Le TNU, Nguyen TQ, Kalailingam P, Nguyen YTK, Sukumar VK, Tan CKH, Tukijan F, Couty L, Hasan Z, Del Gaudio I, Wenk MR, Cazenave-Gassiot A, Camerer E, Nguyen LN (2022) Mfsd2b and Spns2 are essential for maintenance of blood vessels during development and in anaphylactic shock. Cell Rep 40(7):111208. https://doi.org/10.1016/j.celrep.2022.111208
Matloubian M, Lo CG, Cinamon G, Lesneski MJ, Xu Y, Brinkmann V, Allende ML, Proia RL, Cyster JG (2004) Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature 427(6972):355–360. https://doi.org/10.1038/nature02284
Liu Y, Wada R, Yamashita T, Mi Y, Deng CX, Hobson JP, Rosenfeldt HM, Nava VE, Chae SS, Lee MJ, Liu CH, Hla T, Spiegel S, Proia RL (2000) Edg-1, the G protein-coupled receptor for sphingosine-1-phosphate, is essential for vascular maturation. J Clin Invest 106(8):951–961. https://doi.org/10.1172/JCI10905
Kono M, Mi Y, Liu Y, Sasaki T, Allende ML, Wu YP, Yamashita T, Proia RL (2004) The sphingosine-1-phosphate receptors S1P1, S1P2, and S1P3 function coordinately during embryonic angiogenesis. J Biol Chem 279(28):29367–29373. https://doi.org/10.1074/jbc.M403937200
Mendoza A, Breart B, Ramos-Perez WD, Pitt LA, Gobert M, Sunkara M, Lafaille JJ, Morris AJ, Schwab SR (2012) The transporter Spns2 is required for secretion of lymph but not plasma sphingosine-1-phosphate. Cell Rep 2(5):1104–1110. https://doi.org/10.1016/j.celrep.2012.09.021
Nagahashi M, Kim EY, Yamada A, Ramachandran S, Allegood JC, Hait NC, Maceyka M, Milstien S, Takabe K, Spiegel S (2013) Spns2, a transporter of phosphorylated sphingoid bases, regulates their blood and lymph levels, and the lymphatic network. FASEB J 27(3):1001–1011. https://doi.org/10.1096/fj.12-219618
Chandrakanthan M, Nguyen TQ, Hasan Z, Muralidharan S, Vu TM, Li AWL, Le UTN, Thi Thuy Ha H, Baik SH, Tan SH, Foo JC, Wenk MR, Cazenave-Gassiot A, Torta F, Ong WY, Chan MYY, Nguyen LN (2021) Deletion of Mfsd2b impairs thrombotic functions of platelets. Nat Commun 12(1):2286. https://doi.org/10.1038/s41467-021-22642-x
Donoviel MS, Hait NC, Ramachandran S, Maceyka M, Takabe K, Milstien S, Oravecz T, Spiegel S (2015) Spinster 2, a sphingosine-1-phosphate transporter, plays a critical role in inflammatory and autoimmune diseases. FASEB J 29(12):5018–5028. https://doi.org/10.1096/fj.15-274936
Bian G, Yu C, Liu L, Fang C, Chen K, Ren P, Zhang Q, Liu F, Zhang K, Xue Q, Xiang J, Guo H, Song J, Zhao Y, Wu W, Chung SK, Sun R, Ju G, Wang J (2018) Sphingosine 1-phosphate stimulates eyelid closure in the developing rat by stimulating EGFR signaling. Sci Signal. https://doi.org/10.1126/scisignal.aat1470
Fang C, Bian G, Ren P, Xiang J, Song J, Yu C, Zhang Q, Liu L, Chen K, Liu F, Zhang K, Wu C, Sun R, Hu D, Ju G, Wang J (2018) S1P transporter SPNS2 regulates proper postnatal retinal morphogenesis. FASEB J 32(7):3597–3613. https://doi.org/10.1096/fj.201701116R
Chen J, Ingham N, Kelly J, Jadeja S, Goulding D, Pass J, Mahajan VB, Tsang SH, Nijnik A, Jackson IJ, White JK, Forge A, Jagger D, Steel KP (2014) Spinster homolog 2 (spns2) deficiency causes early onset progressive hearing loss. PLoS Genet 10(10):e1004688. https://doi.org/10.1371/journal.pgen.1004688
Lu E, Cyster JG (2019) G-protein coupled receptors and ligands that organize humoral immune responses. Immunol Rev 289(1):158–172. https://doi.org/10.1111/imr.12743
Dixit D, Okuniewska M, Schwab SR (2019) Secrets and lyase: Control of sphingosine 1-phosphate distribution. Immunol Rev 289(1):173–185. https://doi.org/10.1111/imr.12760
Mendoza A, Fang V, Chen C, Serasinghe M, Verma A, Muller J, Chaluvadi VS, Dustin ML, Hla T, Elemento O, Chipuk JE, Schwab SR (2017) Lymphatic endothelial S1P promotes mitochondrial function and survival in naive T cells. Nature 546(7656):158–161. https://doi.org/10.1038/nature22352
van der Weyden L, Arends MJ, Campbell AD, Bald T, Wardle-Jones H, Griggs N, Velasco-Herrera MD, Tuting T, Sansom OJ, Karp NA, Clare S, Gleeson D, Ryder E, Galli A, Tuck E, Cambridge EL, Voet T, Macaulay IC, Wong K, Sanger Mouse Genetics P, Spiegel S, Speak AO, Adams DJ (2017) Genome-wide in vivo screen identifies novel host regulators of metastatic colonization. Nature 541(7636):233–236. https://doi.org/10.1038/nature20792
Pham TH, Baluk P, Xu Y, Grigorova I, Bankovich AJ, Pappu R, Coughlin SR, McDonald DM, Schwab SR, Cyster JG (2010) Lymphatic endothelial cell sphingosine kinase activity is required for lymphocyte egress and lymphatic patterning. J Exp Med 207(1):17–27. https://doi.org/10.1084/jem.20091619
Simmons S, Sasaki N, Umemoto E, Uchida Y, Fukuhara S, Kitazawa Y, Okudaira M, Inoue A, Tohya K, Aoi K, Aoki J, Mochizuki N, Matsuno K, Takeda K, Miyasaka M, Ishii M (2019) High-endothelial cell-derived S1P regulates dendritic cell localization and vascular integrity in the lymph node. Elife. https://doi.org/10.7554/eLife.41239
Hisano Y, Kobayashi N, Yamaguchi A, Nishi T (2012) Mouse SPNS2 functions as a sphingosine-1-phosphate transporter in vascular endothelial cells. PLoS ONE 7(6):e38941. https://doi.org/10.1371/journal.pone.0038941
Nguyen TQ, Vu TM, Tukijan F, Muralidharan S, Foo JC, Li Chin JF, Hasan Z, Torta F, Nguyen LN (2021) Erythrocytes efficiently utilize exogenous sphingosines for S1P synthesis and export via Mfsd2b. J Biol Chem 296:100201. https://doi.org/10.1074/jbc.RA120.012941
Pereira JP, Kelly LM, Xu Y, Cyster JG (2009) EBI2 mediates B cell segregation between the outer and centre follicle. Nature 460(7259):1122–1126. https://doi.org/10.1038/nature08226
Lu E, Wolfreys FD, Muppidi JR, Xu Y, Cyster JG (2019) S-Geranylgeranyl-L-glutathione is a ligand for human B cell-confinement receptor P2RY8. Nature 567(7747):244–248. https://doi.org/10.1038/s41586-019-1003-z
Green JA, Suzuki K, Cho B, Willison LD, Palmer D, Allen CD, Schmidt TH, Xu Y, Proia RL, Coughlin SR, Cyster JG (2011) The sphingosine 1-phosphate receptor S1P(2) maintains the homeostasis of germinal center B cells and promotes niche confinement. Nat Immunol 12(7):672–680. https://doi.org/10.1038/ni.2047
Brinkmann V, Billich A, Baumruker T, Heining P, Schmouder R, Francis G, Aradhye S, Burtin P (2010) Fingolimod (FTY720): discovery and development of an oral drug to treat multiple sclerosis. Nat Rev Drug Discov 9(11):883–897. https://doi.org/10.1038/nrd3248
Chun J, Hartung HP (2010) Mechanism of action of oral fingolimod (FTY720) in multiple sclerosis. Clin Neuropharmacol 33(2):91–101. https://doi.org/10.1097/WNF.0b013e3181cbf825
Pham TH, Okada T, Matloubian M, Lo CG, Cyster JG (2008) S1P1 receptor signaling overrides retention mediated by G alpha i-coupled receptors to promote T cell egress. Immunity 28(1):122–133. https://doi.org/10.1016/j.immuni.2007.11.017
Chun J, Giovannoni G, Hunter SF (2021) Sphingosine 1-phosphate receptor modulator therapy for multiple sclerosis: differential downstream receptor signalling and clinical profile effects. Drugs 81(2):207–231. https://doi.org/10.1007/s40265-020-01431-8
Chun J, Kihara Y, Jonnalagadda D, Blaho VA (2019) Fingolimod: lessons learned and new opportunities for treating multiple sclerosis and other disorders. Annu Rev Pharmacol Toxicol 59:149–170. https://doi.org/10.1146/annurev-pharmtox-010818-021358
Saba JD (2019) Fifty years of lyase and a moment of truth: sphingosine phosphate lyase from discovery to disease. J Lipid Res 60(3):456–463. https://doi.org/10.1194/jlr.S091181
Osborne N, Brand-Arzamendi K, Ober EA, Jin SW, Verkade H, Holtzman NG, Yelon D, Stainier DY (2008) The spinster homolog, two of hearts, is required for sphingosine 1-phosphate signaling in zebrafish. Curr Biol 18(23):1882–1888. https://doi.org/10.1016/j.cub.2008.10.061
Kawahara A, Nishi T, Hisano Y, Fukui H, Yamaguchi A, Mochizuki N (2009) The sphingolipid transporter spns2 functions in migration of zebrafish myocardial precursors. Science 323(5913):524–527. https://doi.org/10.1126/science.1167449
Nguyen TQ, Vu TM, Tukijan F, Muralidharan S, Foo JC, Chin JFL, Hasan Z, Torta F, Nguyen LN (2020) Erythrocytes efficiently utilize exogenous sphingosines for S1P synthesis and export via Mfsd2b. J Biol Chem. https://doi.org/10.1074/jbc.RA120.012941
Han MH, Hwang SI, Roy DB, Lundgren DH, Price JV, Ousman SS, Fernald GH, Gerlitz B, Robinson WH, Baranzini SE, Grinnell BW, Raine CS, Sobel RA, Han DK, Steinman L (2008) Proteomic analysis of active multiple sclerosis lesions reveals therapeutic targets. Nature 451(7182):1076–1081. https://doi.org/10.1038/nature06559
Hasan Z, Koizumi SI, Sasaki D, Yamada H, Arakaki N, Fujihara Y, Okitsu S, Shirahata H, Ishikawa H (2017) JunB is essential for IL-23-dependent pathogenicity of Th17 cells. Nat Commun 8:15628. https://doi.org/10.1038/ncomms15628
Seifert HA, Gerstner G, Kent G, Vandenbark AA, Offner H (2019) Estrogen-induced compensatory mechanisms protect IL-10-deficient mice from developing EAE. J Neuroinflammation 16(1):195. https://doi.org/10.1186/s12974-019-1588-z
Narayanaswamy P, Shinde S, Sulc R, Kraut R, Staples G, Thiam CH, Grimm R, Sellergren B, Torta F, Wenk MR (2014) Lipidomic “deep profiling”: an enhanced workflow to reveal new molecular species of signaling lipids. Anal Chem 86(6):3043–3047. https://doi.org/10.1021/ac4039652
Gao L, Ji S, Burla B, Wenk MR, Torta F, Cazenave-Gassiot A (2021) LICAR: an application for isotopic correction of targeted lipidomic data acquired with class-based chromatographic separations using multiple reaction monitoring. Anal Chem 93(6):3163–3171. https://doi.org/10.1021/acs.analchem.0c04565
Funding
This study was supported in part by Singapore Ministry of Health’s National Research Council NMRC/OFIRG/0066/20, Ministry of Education MOE2018-T2-1-126, NUHSRO/2022/067/T1, T2EP30221-0012, NUSMED-FOS Joint Research Programme on Healthy Brain Aging grants (to L.N.N.).
Author information
Authors and Affiliations
Contributions
ZH designed and performed in vivo experiments and ex vivo experiments. TQN performed lipidomic analysis and in vivo experiments. BWSL and JW performed IF staining. CKHT assisted with Evans blue leakage. CCYW assisted with H&E staining. CHT collected lymph. JY helped with EAE experiments. VA supervised CHT. YZ supervised JY. LNN conceived and designed the study and performed confocal imaging. LNN and ZH prepared the figures and wrote the paper. We thank other NNL lab members for kind assistance with experiments in this project.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
All animal experiments were performed under R19-0567 protocol which was approved by the IACUC committee of the National University of Singapore.
Consent to publish
The authors approved for publication of this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hasan, Z., Nguyen, T.Q., Lam, B.W.S. et al. Postnatal deletion of Spns2 prevents neuroinflammation without compromising blood vascular functions. Cell. Mol. Life Sci. 79, 541 (2022). https://doi.org/10.1007/s00018-022-04573-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04573-y