Abstract
Background
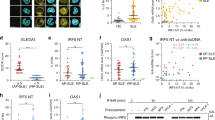
Previous studies reported that IL-38 was abnormally expressed in patients with systemic lupus erythematosus (SLE). However, the involvement of IL-38 in the pathophysiology of SLE remains unknown.
Methods
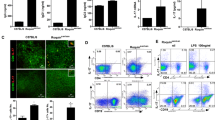
The therapeutic potential of IL-38 was tested in pristane-treated wild-type (WT) and IL-38−/− mice. Thus, SLE was induced via pristane in WT and IL-38−/− mice. Afterwards, the liver, spleen, and kidney of each mouse were obtained. The flow cytometric analysis of the immune cells, serologic expression of inflammatory cytokines and autoantibodies, renal histopathology, and inflammatory signaling were evaluated.
Results
WT mice with pristane-induced lupus exhibited hepatomegaly, splenomegaly, severe kidney damages, increased lymphoproliferation, enhanced lymphoproliferation, and upregulated inflammatory cytokines, such as IL-6, IL-13, IL-17A, MIP-3α, IL-12p70, and IFNγ, and elevated levels of autoantibodies, such as ANA IgG, anti-dsDNA IgG, and total IgG. IL-38−/− mice whose lupus progressed, had elevated cells of CD14+, CD19+, CD3+, and Th1, upregulated inflammatory cytokines and autoantibodies, and severe pathological changes in kidney. Administration of recombinant murine IL-38 to pristane-treated IL-38−/− mice improved their renal histopathology, which depended on ERK1/2, JNK1/2, p38, NF-κB p65, and STAT5 signaling pathways.
Conclusion
IL-38 regulates SLE pathogenesis. Furthermore, targeting IL-38 is critical in the treatment of SLE.






Similar content being viewed by others
Data availability
Datasets are available from the corresponding author on reasonable request.
Abbreviations
- SLE:
-
Systemic lupus erythematosus
- WT:
-
Wild-type
- IL-38:
-
Interleukin-38
- LN:
-
Lupus nephritis
- IL-1Ra:
-
IL-1 receptor antagonist
- IL-36Ra:
-
IL-36 receptor antagonist
- RA:
-
Rheumatoid arthritis
References
Xu WD, Huang AF. Role of interleukin-38 in chronic inflammatory diseases: a comprehensive review. Front Immunol. 2018;9:1462.
Esmaeilzadeh A, Bahmaie N, Nouri E, Hajkazemi MJ, Zareh RM. Immunobiological properties and clinical applications of interleukin-38 for immune-mediated disorders: a systematic review study. Int J Mol Sci. 2021;22(22):12552.
Han Y, Huard A, Mora J, da Silva P, Brüne B, Weigert A. IL-36 family cytokines in protective versus destructive inflammation. Cell Signal. 2020;75: 109773.
Hao Z, Liu Y. IL-38 and IL-36 target autophagy for regulating synoviocyte proliferation, migration, and invasion in rheumatoid arthritis. Dis Markers. 2021;2021:7933453.
Huang L, Zhang H, Zhao D, Hu H, Lu Z. Interleukin-38 suppresses cell migration and proliferation and promotes apoptosis of colorectal cancer cell through negatively regulating extracellular signal-regulated kinases signaling. J Interferon Cytokine Res. 2021;41(10):375–84.
Xu WD, Su LC, Liu XY, et al. IL-38: a novel cytokine in systemic lupus erythematosus pathogenesis. J Cell Mol Med. 2020;24(21):12379–89.
Xu WD, Su LC, He CS, Huang AF. Plasma interleukin-38 in patients with rheumatoid arthritis. Int Immunopharmacol. 2018;65:1–7.
Freitas EC, de Oliveira MS, Monticielo OA. Pristane-induced lupus: considerations on this experimental model. Clin Rheumatol. 2017;36(11):2403–14.
Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997;40(9):1725.
Markowitz GS, D’Agati VD. The ISN/RPS 2003 classification of lupus nephritis: an assessment at 3 years. Kidney Int. 2007;71(6):491–5.
Satoh M, Kumar A, Kanwar YS, Reeves WH. Anti-nuclear antibody production and immune-complex glomerulonephritis in BALB/c mice treated with pristane. Proc Natl Acad Sci USA. 1995;92(24):10934–8.
Reeves WH, Lee PY, Weinstein JS, Satoh M, Lu L. Induction of autoimmunity by pristane and other naturally occurring hydrocarbons. Trends Immunol. 2009;30(9):455–64.
Fu R, Xia Y, Li M, et al. Pim-1 as a therapeutic target in lupus nephritis. Arthritis Rheumatol. 2019;71(8):1308–18.
Zarrabi M, Nazarinia M, Rahimi Jaberi A, Gholijani N, Amirghofran Z. Elevated IL-38 serum levels in newly diagnosed multiple sclerosis and systemic sclerosis patients. Med Princ Pract. 2021;30(2):146–53.
Xie C, Yan W, Quan R, et al. Interleukin-38 is elevated in inflammatory bowel diseases and suppresses intestinal inflammation. Cytokine. 2020;127: 154963.
Luo D, Chen Y, Zhou N, Li T, Wang H. Blockade of Th17 response by IL-38 in primary Sjogren’s syndrome. Mol Immunol. 2020;127:107–11.
Rudloff I, Godsell J, Nold-Petry CA, et al. Brief report: interleukin-38 exerts anti-inflammatory functions and is associated with disease activity in systemic lupus erythematosus. Arthritis Rheumatol. 2015;67(12):3219–25.
Chu M, Tam LS, Zhu J, et al. In vivo anti-inflammatory activities of novel cytokine IL-38 in Murphy Roths Large (MRL)/lpr mice. Immunobiology. 2017;222(3):483–93.
Chu M, Chu IM, Yung EC, et al. Aberrant expression of novel cytokine IL-38 and regulatory T lymphocytes in childhood asthma. Molecules. 2016;21(7):933.
Ge Y, Huang M, Wu Y, Dong N, Yao YM. Interleukin-38 protects against sepsis by augmenting immunosuppressive activity of CD4(+) CD25(+) regulatory T cells. J Cell Mol Med. 2020;24(2):2027–39.
Sigdel KR, Duan L, Wang Y, et al. Serum cytokines Th1, Th2, and Th17 expression profiling in active lupus nephritis-IV: from a Southern Chinese Han population. Mediators Inflamm. 2016;2016:4927530.
Mao YM, Zhao CN, Leng J, et al. Interleukin-13: a promising therapeutic target for autoimmune disease. Cytokine Growth Factor Rev. 2019;45:9–23.
Xiao ZX, Hu X, Zhang X, et al. High salt diet accelerates the progression of murine lupus through dendritic cells via the p38 MAPK and STAT1 signaling pathways. Signal Transduct Target Ther. 2020;5(1):34.
Goropevšek A, Holcar M, Pahor A, Avčin T. STAT signaling as a marker of SLE disease severity and implications for clinical therapy. Autoimmun Rev. 2019;18(2):144–54.
Sun X, Hou T, Cheung E, et al. Anti-inflammatory mechanisms of the novel cytokine interleukin-38 in allergic asthma. Cell Mol Immunol. 2020;17(6):631–46.
Gao X, Chan PKS, Lui GCY, et al. Interleukin-38 ameliorates poly(I:C) induced lung inflammation: therapeutic implications in respiratory viral infections. Cell Death Dis. 2021;12(1):53.
Yu H, Liu Y, Xie W, Xie Q, Liu Q, Cheng L. IL-38 alleviates the inflammatory response and the degeneration of nucleus pulposus cells via inhibition of the NF-κB signaling pathway in vitro. Int Immunopharmacol. 2020;85: 106592.
Ma K, Du W, Xiao F, et al. IL-17 sustains the plasma cell response via p38-mediated Bcl-xL RNA stability in lupus pathogenesis. Cell Mol Immunol. 2021;18(7):1739–50.
Hou LF, He SJ, Li X, et al. Oral administration of artemisinin analog SM934 ameliorates lupus syndromes in MRL/lpr mice by inhibiting Th1 and Th17 cell responses. Arthritis Rheum. 2011;63(8):2445–55.
Chang A, Clark MR, Ko K. Cellular aspects of the pathogenesis of lupus nephritis. Curr Opin Rheumatol. 2021;33(2):197–204.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81701606), and Sichuan Provincial Natural Science Foundation (2022NSFSC0697, 2022NSFSC0694).
Author information
Authors and Affiliations
Contributions
Study conception and design: WX, LS, and AH. Acquisition of data: LF, YL, and XL. Analysis and interpretation of data: QH, QW, and JZ. Drafting the article: WX, LS, and AH. Final approval of the version of the article to be published: all the authors, and all the authors agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
This study was approved Animal Ethics Committee of Southwest Medical University.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, WD., Su, LC., Fu, L. et al. IL-38, a potential therapeutic agent for lupus, inhibits lupus progression. Inflamm. Res. 71, 963–975 (2022). https://doi.org/10.1007/s00011-022-01581-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-022-01581-3