Abstract
Objective and design
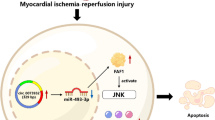
We aim to explore the molecular mechanism of myocardial ischemia–reperfusion injury (MIRI).
Methods
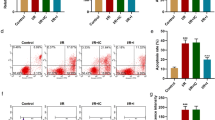
The H9C2 cells were cultured under hypoxia/reoxygenation (H/R) condition to induce myocardial injury in vitro. The expression of miR-451-3p and MAP1LC3B was detected by RT-qPCR. Dual-luciferase reporter assay and RNA pull-down assay were performed to examine the relationship between microRNA (miR)-451-3p and MAP1LC3B. CCK8 was used to test cell viability. The level of LDH and CK was evaluated via ELISA. Immunofluorescence assay and flow cytometry were applied to detect autophagy and apoptosis, respectively. Autophagy-related protein expressions were determined by western blotting. Furthermore, an in vivo rat model of MIRI was established by subjection to 30 min ischemia and subsequently 24 h reperfusion for validation of the role of miR-451-3p in regulating MIRI in vivo.
Results
miR-451-3p was down-regulated in MIRI, and miR-451-3p mimics transfection alleviated autophagy and apoptosis induced by MIRI. miR-451-3p could target MAP1LC3B directly. Co-transfection miR-451-3p mimics and pcDNA 3.1 MAP1LC3B curbed the protected effects of miR-451-3p mimics on MIRI.
Conclusions
miR-451-3p played a protective role in MIRI via inhibiting MAP1LC3B-mediated autophagy, which may provide new molecular targets for the treatment of MIRI and further improves the clinical outcomes of heart diseases.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- MIRI:
-
Myocardial ischemia–reperfusion injury
- H/R:
-
Hypoxia/reoxygenation
- AMI:
-
Acute myocardial infarction
- MAP1LC3/ LC3:
-
Microtubule-associated protein 1 light chain 3
- FITC:
-
Fluorescein isothiocyanate
- MAP1LC3B-WT:
-
MAP1LC3B wild-type
- 3′-UTR:
-
3′-Untranslated region
- MAP1LC3B –MUT:
-
MAP1LC3B mutated-type
- LAD:
-
The left anterior descending coronary artery
- AI:
-
Apoptosis index
- ANOVA:
-
Analysis of variance
References
Hu SS, Kong LZ, Gao RL, Zhu ML, Wang W, Wang YJ, et al. Outline of the report on cardiovascular disease in China, 2010. Biomed Environ Sci. 2012;25(3):251–6. https://doi.org/10.3967/0895-3988.2012.03.001.
Armstrong PW, Gershlick AH, Goldstein P, Wilcox R, Danays T, Lambert Y, et al. Fibrinolysis or primary PCI in ST-segment elevation myocardial infarction. N Engl J Med. 2013;368(15):1379–87. https://doi.org/10.1056/NEJMoa1301092.
Libby P. Mechanisms of acute coronary syndromes and their implications for therapy. N Engl J Med. 2013;368(21):2004–13. https://doi.org/10.1056/NEJMra1216063.
Keeley EC, Boura JA, Grines CL. Primary angioplasty versus intravenous thrombolytic therapy for acute myocardial infarction: a quantitative review of 23 randomised trials. Lancet. 2003;361(9351):13–20. https://doi.org/10.1016/S0140-6736(03)12113-7.
Cecconi F, Levine B. The role of autophagy in mammalian development: cell makeover rather than cell death. Dev Cell. 2008;15(3):344–57. https://doi.org/10.1016/j.devcel.2008.08.012.
Rabinovich-Nikitin I, Lieberman B, Martino TA, Kirshenbaum LA. Circadian-regulated cell death in cardiovascular diseases. Circulation. 2019;139(7):965–80. https://doi.org/10.1161/CIRCULATIONAHA.118.036550.
Li D, Wang J, Hou J, Fu J, Liu J, Lin R. Salvianolic acid B induced upregulation of miR-30a protects cardiac myocytes from ischemia/reperfusion injury. BMC Complement Altern Med. 2016;16:336. https://doi.org/10.1186/s12906-016-1275-x.
Zhou LY, Zhai M, Huang Y, Xu S, An T, Wang YH, et al. The circular RNA ACR attenuates myocardial ischemia/reperfusion injury by suppressing autophagy via modulation of the Pink1/ FAM65B pathway. Cell Death Differ. 2019;26(7):1299–315. https://doi.org/10.1038/s41418-018-0206-4.
Rogov V, Dotsch V, Johansen T, Kirkin V. Interactions between autophagy receptors and ubiquitin-like proteins form the molecular basis for selective autophagy. Mol Cell. 2014;53(2):167–78. https://doi.org/10.1016/j.molcel.2013.12.014.
Othman EQ, Kaur G, Mutee AF, Muhammad TS, Tan ML. Immunohistochemical expression of MAP1LC3A and MAP1LC3B protein in breast carcinoma tissues. J Clin Lab Anal. 2009;23(4):249–58. https://doi.org/10.1002/jcla.20309.
Singh R, Cuervo AM. Autophagy in the cellular energetic balance. Cell Metab. 2011;13(5):495–504. https://doi.org/10.1016/j.cmet.2011.04.004.
Shi J, Bei Y, Kong X, Liu X, Lei Z, Xu T, et al. miR-17-3p contributes to exercise-induced cardiac growth and protects against myocardial ischemia-reperfusion injury. Theranostics. 2017;7(3):664–76. https://doi.org/10.7150/thno.15162.
Hinkel R, Penzkofer D, Zuhlke S, Fischer A, Husada W, Xu QF, et al. Inhibition of microRNA-92a protects against ischemia/reperfusion injury in a large-animal model. Circulation. 2013;128(10):1066–75. https://doi.org/10.1161/CIRCULATIONAHA.113.001904.
Joladarashi D, Garikipati VNS, Thandavarayan RA, Verma SK, Mackie AR, Khan M, et al. Enhanced cardiac regenerative ability of stem cells after ischemia-reperfusion injury: role of human CD34+ Cells Deficient in MicroRNA-377. J Am Coll Cardiol. 2015;66(20):2214–26. https://doi.org/10.1016/j.jacc.2015.09.009.
Przyklenk K. microRNA-144: the “what” and “how” of remote ischemic conditioning? Basic Res Cardiol. 2014;109(5):429. https://doi.org/10.1007/s00395-014-0429-6.
Lee TL, Lee MH, Chen YC, Lee YC, Lai TC, Lin HY, et al. Vitamin D attenuates ischemia/reperfusion-induced cardiac injury by reducing mitochondrial fission and mitophagy. Front Pharmacol. 2020;11: 604700. https://doi.org/10.3389/fphar.2020.604700.
Lv X, Xu T, Wu Q, Zhou Y, Huang G, Xu Y, et al. 6-gingerol activates PI3K/Akt and inhibits apoptosis to attenuate myocardial ischemia/reperfusion injury. Evid Based Complement Altern Med. 2018;2018:9024034. https://doi.org/10.1155/2018/9024034.
Yang CF. Clinical manifestations and basic mechanisms of myocardial ischemia/reperfusion injury. Ci ji yi xue za zhi = Tzu-Chi Med J. 2018;30(4):209–15. https://doi.org/10.4103/tcmj.tcmj_33_18.
Sun T, Li MY, Li PF, Cao JM. MicroRNAs in cardiac autophagy: small molecules and big role. Cells. 2018. https://doi.org/10.3390/cells7080104.
Ma S, Wang Y, Chen Y, Cao F. The role of the autophagy in myocardial ischemia/reperfusion injury. Biochem Biophys Acta. 2015;1852(2):271–6. https://doi.org/10.1016/j.bbadis.2014.05.010.
Yang J, He J, Ismail M, Tweeten S, Zeng F, Gao L, et al. HDAC inhibition induces autophagy and mitochondrial biogenesis to maintain mitochondrial homeostasis during cardiac ischemia/reperfusion injury. J Mol Cell Cardiol. 2019;130:36–48. https://doi.org/10.1016/j.yjmcc.2019.03.008.
Condello M, Pellegrini E, Caraglia M, Meschini S. Targeting autophagy to overcome human diseases. Int J Mol Sci. 2019. https://doi.org/10.3390/ijms20030725.
Hang P, Zhao J, Su Z, Sun H, Chen T, Zhao L, et al. Choline inhibits ischemia-reperfusion-induced cardiomyocyte autophagy in rat myocardium by activating Akt/mTOR signaling. Cell Physiol Biochem. 2018;45(5):2136–44. https://doi.org/10.1159/000488049.
Su Q, Liu Y, Lv XW, Ye ZL, Sun YH, Kong BH, et al. Inhibition of lncRNA TUG1 upregulates miR-142-3p to ameliorate myocardial injury during ischemia and reperfusion via targeting HMGB1- and Rac1-induced autophagy. J Mol Cell Cardiol. 2019;133:12–25. https://doi.org/10.1016/j.yjmcc.2019.05.021.
Dong H, Lei J, Ding L, Wen Y, Ju H, Zhang X. MicroRNA: function, detection, and bioanalysis. Chem Rev. 2013;113(8):6207–33. https://doi.org/10.1021/cr300362f.
Gozuacik D, Akkoc Y, Ozturk DG, Kocak M. Autophagy-regulating microRNAs and cancer. Front Oncol. 2017;7:65. https://doi.org/10.3389/fonc.2017.00065.
Kouhkan F, Hafizi M, Mobarra N, Mossahebi-Mohammadi M, Mohammadi S, Behmanesh M, et al. miRNAs: a new method for erythroid differentiation of hematopoietic stem cells without the presence of growth factors. Appl Biochem Biotechnol. 2014;172(4):2055–69. https://doi.org/10.1007/s12010-013-0633-0.
Tsuchiya S, Oku M, Imanaka Y, Kunimoto R, Okuno Y, Terasawa K, et al. MicroRNA-338-3p and microRNA-451 contribute to the formation of basolateral polarity in epithelial cells. Nucleic Acids Res. 2009;37(11):3821–7. https://doi.org/10.1093/nar/gkp255.
Alural B, Duran GA, Tufekci KU, Allmer J, Onkal Z, Tunali D, et al. EPO mediates neurotrophic, neuroprotective, anti-oxidant, and anti-apoptotic effects via downregulation of miR-451 and miR-885-5p in SH-SY5Y neuron-like cells. Front Immunol. 2014;5:475. https://doi.org/10.3389/fimmu.2014.00475.
Bai H, Wu S. miR-451: a novel biomarker and potential therapeutic target for cancer. Onco Targets Ther. 2019;12:11069–82. https://doi.org/10.2147/ott.s230963.
Song L, Su M, Wang S, Zou Y, Wang X, Wang Y, et al. MiR-451 is decreased in hypertrophic cardiomyopathy and regulates autophagy by targeting TSC1. J Cell Mol Med. 2014;18(11):2266–74. https://doi.org/10.1111/jcmm.12380.
Bortnik S, Gorski SM. Clinical applications of autophagy proteins in cancer: from potential targets to biomarkers. Int J Mol Sci. 2017. https://doi.org/10.3390/ijms18071496.
Swaminathan B, Goikuria H, Vega R, Rodríguez-Antigüedad A, López Medina A, Freijo Mdel M, et al. Autophagic marker MAP1LC3B expression levels are associated with carotid atherosclerosis symptomatology. PLoS ONE. 2014;9(12): e115176. https://doi.org/10.1371/journal.pone.0115176.
Tang C, Han H, Liu Z, Liu Y, Yin L, Cai J, et al. Activation of BNIP3-mediated mitophagy protects against renal ischemia-reperfusion injury. Cell Death Dis. 2019;10(9):677. https://doi.org/10.1038/s41419-019-1899-0.
Acknowledgements
We would like to give our sincere gratitude to the reviewers for their constructive comments. This work was supported by Natural Science Foundation of Guangxi (No.2018GXNSFDA281039, No.2018GXNSFAA050110, No. 2020GXNSFAA297009).
Funding
This work was supported by Natural Science Foundation of Guangxi (No.2018GXNSFDA281039, No.2018GXNSFAA050110, No. 2020GXNSFAA297009).
Author information
Authors and Affiliations
Contributions
Conception and study design: XWL. Data acquisition: ZFH, PPZ. Data analysis: QYQ. Manuscript drafting: YXH. Manuscript revising: TTX. All authors have read and approved the final version of this manuscript to be published.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
Not applicable.
Consent for publication
Not applicable.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lv, XW., He, ZF., Zhu, PP. et al. miR-451-3p alleviates myocardial ischemia/reperfusion injury by inhibiting MAP1LC3B-mediated autophagy. Inflamm. Res. 70, 1089–1100 (2021). https://doi.org/10.1007/s00011-021-01508-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-021-01508-4