Abstract
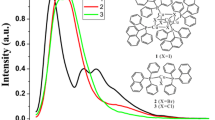
The homoleptic metal-TPPTS complex Cs8[Au(TPPTS)3].5.25 H2O has been characterized structurally. The x-ray data for the triclinic crystal, P-1, with cell constants a = 13.7003(4), b = 18.0001(6), c = 18.2817(2), α = 100.249(2), β = 99.593(2), ψ = 109.818(2) shows trigonal planar Au(I) centers coordinated by three TPPTS ligands having Au-P distances equal to 2.374(6), 2.394(5), and 2.417(5)A. The structure shows a complex network of bonding involving the Cs+ ions, the sulfate groups of the ligand, and H2O solvent molecules. The [Au(TPPTS)3]8− luminesces in the solid state and in aqueous solution with a broad emission atca 520 nm which is quenched by O2. and other small molecules. The forbidden singlet to triplet absorption atca 552 nm with ε≈ 0.8Lmol−1 cm−1 is also observed.
Similar content being viewed by others
References
‘Homogeneous Catalysis with Metal Phosphine Complexes’, ed. L.H. Pignolet, Plenum Press, 1983
‘Aqueous-Phase Organometallic Catalysis’, ed. B. Cornils and W.A. Hermann, Wiley-VCH, 1998
C. Washam, ‘Reaction Times’, American Chemical Society: Washington, D.C., April 1998
I.T. Horváth and F. Joó, ‘Aqueous Organometallic Chemistry and Catalysis’, Kluwer Acad., Dordrecht, 1995
C. Amatore, E. Blart, J.P. Genêt, A. Jutand, S. Lemaire-Audoire and M. Savignac,J. Org. Chem., 1995,60, 6829
F. Monteil, L. Miquel, R. Queau and P. Kalck, in reference 3, ‘ pp.131–147
J.C. Vickery, M.M. Olmstead, E.Y. Fung and A.L. Balch,Angew. Chem. Int. Ed. Engl., 1997,36, 1179
M.A. Mansour, W.B. Connick, R.J. Lachicotte, H.J. Gysling and R. Eisenberg,J. Am. Chem. Soc., 1998,120, 1329
M.N.I. Khan, R.J. Staples, C. King, J.P. Fackler; Jr and R.E.P. Winpenny,Inorg. Chem., 1993,32, 5800
C. King, M.N.I. Khan, R.J. Staples and J.P. Fackler, Jr,Inorg. Chem., 1992,31, 3236
T.M. McCleskey and H.B. Gray,Inorg. Chem., 1992,31, 1734
J.M. Forward, J.P., Fackler, Jr and Z. Assefa, in ‘Optoelectronic Properties of Inorganic Compounds’, ed. D.M. Roundhill and J.P. Fackler, Jr, Plenum Press, 1999, pp.195-229
J.M. Forward, Z. Assefa and J.P. Fackler, Jr,J. Am. Chem. Soc., 1995,117, 9103
D.J. Darensbourg, C.J. Bischoff and J.H. Reibenspies,Inorg. Chem., 1991,30, 1144
T. Bartik, B. Bartik, B.E. Hanson, K.H. Whitmire and I. Guo,Inorg. Chem., 1993,32, 5833
D.J. Darensbourg, T. J. Decuir and J.H. Reibenspies, ‘Aqueous Organometallic Chemistry and Catalysis’, ed. I.T. Horrath and F. Joo, NATO ASI Series, Kluwer Academic Publishers, 1995, p. 61
The crystal structure of 1 was solved and refined in the triclinic P-1 space group. Pale yellow crystals were obtained from a water solution. Crystallographic data are given as a,b,c (Å); α,β,ψ (deg); volume (Å3), Z, R1 [I>2sigma(I)], and GOF: a = 13.7003(4), 18.0001(6), 18.2817(2); 100.249(2), 99.593(2), 109.818(2); 4046.3(2), 2, 0.0659, 0.946
B. Cornils and E.G. Kuntz, in reference 2, p. 276
J. Kang, Masters Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, 1997
M.C. Gimeno and A. Laguna,Chem. Rev., 1997,97, 511
J.M. Forward, J.P. Fackler, Jr and R.J. Staples,Organometallics, 1995,14, 4194
J.M. Forward, R.J. Staples, C.W. Liu and J.P. Fackler, Jr,Acta Cryst. Sect. C, 1997, 195
D.E. Berning, K.V. Katti, C.L. Barnes and W.A. Volkert,Chem. Ber./Recueil, 1997,130, 907
A.L. Casalunuovo and J.C. Calabrese,J. Am. Chem. Soc., 1990,112, 4324
F. Monteil and P.J. Kalck,Organomet. Chem., 1994,482, 45–51
Photoluminesce data were obtained using a SLM AMINCO, Model 8100 spectrofluorometer with a xenon lamp. Radiation was filtered through a 0.10M KNO2 solution. Solution measurements, unless stated otherwise, were done in 0.50 M NaCl formed with doubly distilled water, deoxygenated by a 15 min N2 (g) purge.
T.M. Dunn, in ‘Modern Coordination Chemistry’, ed. J. Lewis and R.G. Wilkins, Interscience, 1969, p. 280
The shift observed suggests a strengthening of the Au-P bonds in the excited state by about 5 kcal.
C.-M. Che, H.-L. Kwong, C.-K. Poon and V. W.-W. Yam, J. Chem. Soc., Dalton Trans., 1990, 3215
D. Li, C.-M. Che, H.-L. Kwong and V.H.-H. Yam,. J. Chem. Soc., Dalton Trans., 1992, 3325
A. Mills, A. Lepre, B.R.C. Theobald, E. Slade and B. Murrer,Anal. Chem., 1997,69, 2842.
A. Mills, A. Lepre, B.R.C. Theobald, E. Slade and B.A. Murrer,Gold Bull., 1998,31, 68
Author information
Authors and Affiliations
Additional information
John P Fackler, Jr is Distinguished Professor of Chemistry at Texas A&M University and amongst his more than 280 publications there are over 100 papers relating to gold chemistry. Tiffany A Grant studied for her PhD with Fackler on the luminescent properties of gold(I) complexes and developed the crystallization of the water soluble phosphine complex reported here. Brian E Hanson is Professor of Chemistry at Virginia Polytechnic Institute and State University and is an expert in the use of biphasic systems for hydroformylation which involve sulfonated phosphine complexes of metal ions. Richard J Staples is staff crystallographer for the Chemistry Department at Harvard University and has structurally characterized over 50 gold compounds.
Rights and permissions
About this article
Cite this article
Fackler, J.P., Grant, T.A., Hanson, B.E. et al. Characterization of the Luminescent, Homoleptic, Three-Coordinate, Water Soluble Au(I) Complex of Trisulfonated Triphenylphosphine (TPPTS) as the Cesium Salt, Cs8[Au(TPPTS)3].5.25 H2O. Gold Bull 32, 20–23 (1999). https://doi.org/10.1007/BF03214785
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03214785