Abstract
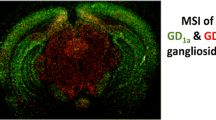
Matrix-assisted laser desorption/ionization imaging mass spectrometry (MALDI-IMS) is a histological method used for various molecules including gangliosides. The method is based on mass spectrometry, and thus target molecules with small structural differences are directly and independently detected. Here we describe a general procedure and related key notes for analyzing major brain gangliosides by MALDI-IMS.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Norris J, Caprioli R (2013) Analysis of tissue specimens by matrix-assisted laser desorption/ionization imaging mass spectrometry in biological and clinical research. Chem Rev 113:2309–2342
Kitakaze K, Mizutani Y, Sugiyama E et al (2016) Protease-resistant modified human β-hexosaminidase B ameliorates symptoms in GM2 gangliosidosis model. J Clin Invest 126:1691–1703
Sugiura Y, Shimma S, Konishi Y et al (2008) Imaging mass spectrometry technology and application on ganglioside study; visualization of age-dependent accumulation of C20-ganglioside molecular species in the mouse hippocampus. PLoS One 3:e3232
Colsch B, Jackson SN, Dutta S, Woods AS (2011) Molecular microscopy of brain gangliosides: Illustrating their distribution in hippocampal cell layers. ACS Chem Neurosci 2:213–222
Zhang Y, Wang J, Liu J et al (2016) Combination of ESI and MALDI mass spectrometry for qualitative, semi-quantitative and in situ analysis of gangliosides in brain. Sci Rep 6:25289
Hirano-Sakamaki W, Sugiyama E, Hayasaka T et al (2015) Alzheimer’s disease is associated with disordered localization of ganglioside GM1 molecular species in the human dentate gyrus. FEBS Lett 589:3611–3616
Schwartz SA, Reyzer ML, Caprioli RM (2003) Direct tissue analysis using matrix-assisted laser desorption/ionization mass spectrometry: practical aspects of sample preparation. J Mass Spectrom 38:699–708
Yang J, Caprioli RM (2011) Matrix sublimation/recrystallization for imaging proteins by mass spectrometry at high spatial resolution. Anal Chem 83:5728–5734
Angel PM, Spraggins JM, Baldwin HS, Caprioli RM (2012) Enhanced sensitivity for high spatial resolution lipid analysis by negative ion mode matrix assisted laser desorption ionization imaging mass spectrometry. Anal Chem 84:1557–1564
Shimma S, Takashima Y, Hashimoto J et al (2013) Alternative two-step matrix application method for imaging mass spectrometry to avoid tissue shrinkage and improve ionization efficiency. J Mass Spectrom 48:1285–1290
Sugiura Y, Konishi Y, Zaima N et al (2009) Visualization of the cell-selective distribution of PUFA-containing phosphatidylcholines in mouse brain by imaging mass spectrometry. J Lipid Res 50:1776–1788
Pirman DA, Kiss A, Heeren RMA, Yost RA (2013) Identifying tissue-specific signal variation in MALDI mass spectrometric imaging by use of an internal standard. Anal Chem 85:1090–1096
Takai N, Tanaka Y, Saji H (2014) Quantification of small molecule drugs in biological tissue sections by imaging mass spectrometry using surrogate tissue-based calibration standards. Mass Spectrom (Tokyo, Japan) 3:A0025
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Sugiyama, E., Setou, M. (2018). Visualization of Brain Gangliosides Using MALDI Imaging Mass Spectrometry. In: Sonnino, S., Prinetti, A. (eds) Gangliosides. Methods in Molecular Biology, vol 1804. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8552-4_10
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8552-4_10
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8551-7
Online ISBN: 978-1-4939-8552-4
eBook Packages: Springer Protocols