Abstract
Background
Recent evidence suggests that the rabbit subscapularis tendon may be anatomically, biomechanically, and histologically suitable to study rotator cuff pathology and repair. However, biomechanical comparisons of rotator cuff repairs in this model have not been evaluated and compared to those in human cadaveric specimens.
Questions/purposes
We quantified the biomechanical properties of the repaired rabbit subscapularis tendon after (1) single-row, (2) double-row, and (3) transosseous-equivalent rotator cuff repair techniques and compared the ratios of repairs to previously published data for human repairs.
Methods
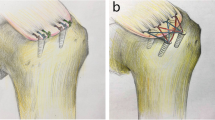
Tensile testing was performed on 21 New Zealand White rabbit subscapularis tendon-humerus complexes for single-row repair, double-row repair, and transosseous-equivalent repair (n = 7 for each group). Video digitizing software was used to quantify deformation. Load elongation data were then used to quantify structural properties. We compared the ratios of rotator cuff repairs for the rabbit data to data from human supraspinatus repair studies previously performed in our laboratory. For our primary end points (linear stiffness, yield load, ultimate load, and energy absorbed to failure), with the numbers available, our statistical power to detect a clinically important difference (defined as 15%) was 85%.
Results
The ratios of single-row/double-row repair were 0.72, 0.73, 0.71, and 0.66 for human supraspinatus and 0.77, 0.74, 0.79, and 0.89 for rabbit subscapularis repair for linear stiffness, yield load, ultimate load, and energy absorbed to failure, respectively. The ratios of double-row/transosseous-equivalent repair were 1.0, 0.86, 0.70, and 0.41 for human supraspinatus and 1.22, 0.85, 0.76, and 0.60 for rabbit subscapularis for linear stiffness, yield load, ultimate load, and energy absorbed to failure, respectively. There were no differences comparing rabbit to human repair ratios for any parameter (p > 0.09 for all comparisons).
Conclusions
Subscapularis repairs in the rabbit at Time 0 result in comparable ratios to human supraspinatus repairs.
Clinical Relevance
The biomechanical similarities between the different types of rotator cuff repair in the rabbit subscapularis and human supraspinatus at Time 0 provide more evidence that the rabbit subscapularis may be an appropriate model to study rotator cuff repairs.




Similar content being viewed by others
References
Ahmad CS, Stewart AM, Izquierdo R, Bigliani LU. Tendon-bone interface motion in transosseous suture and suture anchor rotator cuff repair techniques. Am J Sports Med. 2005;33:1667–1671.
Ahmad CS, Vorys GC, Covey A, Levine WN, Gardner TR, Bigliani LU. Rotator cuff repair fluid extravasation characteristics are influenced by repair technique. J Shoulder Elbow Surg. 2009;18:976–981.
Aoki M, Oguma H, Fukushima S, Ishii S, Ohtani S, Murakami G. Fibrous connection to bone after immediate repair of the canine infraspinatus: the most effective bony surface for tendon attachment. J Shoulder Elbow Surg. 2001;10:123–128.
Apreleva M, Ozbaydar M, Fitzgibbons PG, Warner JJ. Rotator cuff tears: the effect of the reconstruction method on three-dimensional repair site area. Arthroscopy. 2002;18:519–526.
Banas MP, Miller RJ, Totterman S. Relationship between the lateral acromion angle and rotator cuff disease. J Shoulder Elbow Surg. 1995;4:454–461.
Barton ER, Gimbel JA, Williams GR, Soslowsky LJ. Rat supraspinatus muscle atrophy after tendon detachment. J Orthop Res. 2005;23:259–265.
Bigliani LU, Ticker JB, Flatow EL, Soslowsky LJ, Mow VC. The relationship of acromial architecture to rotator cuff disease. Clin Sports Med. 1991;10:823–838.
Bjorkenheim JM. Structure and function of the rabbit’s supraspinatus muscle after resection of its tendon. Acta Orthop Scand. 1989;60:461–463.
Bowser JE, Elder SH, Rashmir-Raven AM, Swiderski CE. A cryogenic clamping technique that facilitates ultimate tensile strength determinations in tendons and ligaments. Vet Comp Orthop Traumatol. 2011;24:370–373.
Burkhart SS, Lo IK. Arthroscopic rotator cuff repair. J Am Acad Orthop Surg. 2006;14:333–346.
Coleman SH, Fealy S, Ehteshami JR, MacGillivray JD, Altchek DW, Warren RF, Turner AS. Chronic rotator cuff injury and repair model in sheep. J Bone Joint Surg Am. 2003;85:2391–2402.
Fabis J, Kordek P, Bogucki A, Mazanowska-Gajdowicz J. Function of the rabbit supraspinatus muscle after large detachment of its tendon: 6-week, 3-month, and 6-month observation. J Shoulder Elbow Surg. 2000;9:211–216.
Flatow EL, Soslowsky LJ, Ticker JB, Pawluk RJ, Hepler M, Ark J, Mow VC, Bigliani LU. Excursion of the rotator cuff under the acromion: patterns of subacromial contact. Am J Sports Med. 1994;22:779–788.
Galatz LM, Ball CM, Teefey SA, Middleton WD, Yamaguchi K. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86:219–224.
Gamradt SC, Gallo RA, Adler RS, Maderazo A, Altchek DW, Warren RF, Fealy S. Vascularity of the supraspinatus tendon three months after repair: characterization using contrast-enhanced ultrasound. J Shoulder Elbow Surg. 2010;19:73–80.
Gazielly DF, Gleyze P, Montagnon C. Functional and anatomical results after rotator cuff repair. Clin Orthop Relat Res. 1994;304:43–53.
Gerber C, Fuchs B, Hodler J. The results of repair of massive tears of the rotator cuff. J Bone Joint Surg Am. 2000;82:505–515.
Gerber C, Meyer DC, Frey E, von Rechenberg B, Hoppeler H, Frigg R, Jost B, Zumstein MA. Neer Award 2007. Reversion of structural muscle changes caused by chronic rotator cuff tears using continuous musculotendinous traction: an experimental study in sheep. J Shoulder Elbow Surg. 2009;18:163–171.
Goutallier D, Postel JM, Gleyze P, Leguilloux P, Van Driessche S. Influence of cuff muscle fatty degeneration on anatomic and functional outcomes after simple suture of full-thickness tears. J Shoulder Elbow Surg. 2003;12:550–554.
Grumet RC, Hadley S, Diltz MV, Lee TQ, Gupta R. Development of a new model for rotator cuff pathology: the rabbit subscapularis muscle. Acta Orthop. 2009;80:97–103.
Gupta R, Lee TQ. Contributions of the different rabbit models to our understanding of rotator cuff pathology. J Shoulder Elbow Surg. 2007;16(5 suppl):S149–S157.
Harryman DT 2nd, Mack LA, Wang KY, Jackins SE, Richardson ML, Matsen FA 3rd. Repairs of the rotator cuff: correlation of functional results with integrity of the cuff. J Bone Joint Surg Am. 1991;73:982–989.
Hashimoto T, Nobuhara K, Hamada T. Pathologic evidence of degeneration as a primary cause of rotator cuff tear. Clin Orthop Relat Res. 2003;415:111–120.
Irlenbusch U, Gansen HK. Muscle biopsy investigations on neuromuscular insufficiency of the rotator cuff: a contribution to the functional impingement of the shoulder joint. J Shoulder Elbow Surg. 2003;12:422–426.
Isaac C, Gharaibeh B, Witt M, Wright VJ, Huard J. Biologic approaches to enhance rotator cuff healing after injury. J Shoulder Elbow Surg. 2012;21:181–190.
Isaksson H, Harjula T, Koistinen A, Iivarinen J, Seppanen K, Arokoski JP, Brama PA, Jurvelin JS, Helminen HJ. Collagen and mineral deposition in rabbit cortical bone during maturation and growth: effects on tissue properties. J Orthop Res. 2010;28:1626–1633.
Kim DH, Elattrache NS, Tibone JE, Jun BJ, DeLaMora SN, Kvitne RS, Lee TQ. Biomechanical comparison of a single-row versus double-row suture anchor technique for rotator cuff repair. Am J Sports Med. 2006;34:407–414.
Milgrom C, Schaffler M, Gilbert S, van Holsbeeck M. Rotator-cuff changes in asymptomatic adults: the effect of age, hand dominance and gender. J Bone Joint Surg Br. 1995;77:296–298.
Neer CS 2nd. Anterior acromioplasty for the chronic impingement syndrome in the shoulder: a preliminary report. J Bone Joint Surg Am. 1972;54:41–50.
Nixon AJ, Watts AE, Schnabel LV. Cell- and gene-based approaches to tendon regeneration. J Shoulder Elbow Surg. 2012;21:278–294.
Ozbaydar M, Elhassan B, Esenyel C, Atalar A, Bozdag E, Sunbuloglu E, Kopuz N, Demirhan M. A comparison of single-versus double-row suture anchor techniques in a simulated repair of the rotator cuff: an experimental study in rabbits. J Bone Joint Surg Br. 2008;90:1386–1391.
Park MC, Cadet ER, Levine WN, Bigliani LU, Ahmad CS. Tendon-to-bone pressure distributions at a repaired rotator cuff footprint using transosseous suture and suture anchor fixation techniques. Am J Sports Med. 2005;33:1154–1159.
Park MC, Elattrache NS, Ahmad CS, Tibone JE. “Transosseous-equivalent” rotator cuff repair technique. Arthroscopy. 2006;22:1360.e1–5.
Park MC, ElAttrache NS, Tibone JE, Ahmad CS, Jun BJ, Lee TQ. Part I. Footprint contact characteristics for a transosseous-equivalent rotator cuff repair technique compared with a double-row repair technique. J Shoulder Elbow Surg. 2007;16:461–468.
Park MC, Tibone JE, ElAttrache NS, Ahmad CS, Jun BJ, Lee TQ. Part II. Biomechanical assessment for a footprint-restoring transosseous-equivalent rotator cuff repair technique compared with a double-row repair technique. J Shoulder Elbow Surg. 2007;16:469–476.
Reilly P, Amis AA, Wallace AL, Emery RJ. Supraspinatus tears: propagation and strain alteration. J Shoulder Elbow Surg. 2003;12:134–138.
Ricchetti ET, Aurora A, Iannotti JP, Derwin KA. Scaffold devices for rotator cuff repair. J Shoulder Elbow Surg. 2012;21:251–265.
Rowshan K, Hadley S, Pham K, Caiozzo V, Lee TQ, Gupta R. Development of fatty atrophy after neurologic and rotator cuff injuries in an animal model of rotator cuff pathology. J Bone Joint Surg Am. 2010;92:2270–2278.
Rudzki JR, Adler RS, Warren RF, Kadrmas WR, Verma N, Pearle AD, Lyman S, Fealy S. Contrast-enhanced ultrasound characterization of the vascularity of the rotator cuff tendon: age- and activity-related changes in the intact asymptomatic rotator cuff. J Shoulder Elbow Surg. 2008;17:96S–100S.
Ruotolo C, Fow JE, Nottage WM. The supraspinatus footprint: an anatomic study of the supraspinatus insertion. Arthroscopy. 2004;20:246–249.
Sher JS, Uribe JW, Posada A, Murphy BJ, Zlatkin MB. Abnormal findings on magnetic resonance images of asymptomatic shoulders. J Bone Joint Surg Am. 1995;77:10–15.
Soslowsky LJ, Carpenter JE, DeBano CM, Banerji I, Moalli MR. Development and use of an animal model for investigations on rotator cuff disease. J Shoulder Elbow Surg. 1996;5:383–392.
Thomazeau H, Boukobza E, Morcet N, Chaperon J, Langlais F. Prediction of rotator cuff repair results by magnetic resonance imaging. Clin Orthop Relat Res. 1997;344:275–283.
Tuoheti Y, Itoi E, Yamamoto N, Seki N, Abe H, Minagawa H, Okada K, Shimada Y. Contact area, contact pressure, and pressure patterns of the tendon-bone interface after rotator cuff repair. Am J Sports Med. 2005;33:1869–1874.
Yamaguchi K, Ditsios K, Middleton WD, Hildebolt CF, Galatz LM, Teefey SA. The demographic and morphological features of rotator cuff disease: a comparison of asymptomatic and symptomatic shoulders. J Bone Joint Surg Am. 2006;88:1699–1704.
Acknowledgments
The authors acknowledge Ranjan Gupta MD and Maxwell Park MD for their expertise and involvement in the development of this animal model for rotator cuff pathology.
Author information
Authors and Affiliations
Corresponding author
Additional information
The institution of one or more authors (KO, JW, TQL) has received funding, during the study period, from a grant from the VA Rehabilitation Research and Development Merit Review.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Each author certifies that his or her institution approved the animal protocol for this investigation and that all investigations were conducted in conformity with ethical principles of research.
This work was performed at the Orthopaedic Biomechanics Laboratory, Long Beach VA Healthcare System, Long Beach, CA, USA.
About this article
Cite this article
Otarodifard, K., Wong, J., Preston, C.F. et al. Relative Fixation Strength of Rabbit Subscapularis Repair Is Comparable to Human Supraspinatus Repair at Time 0. Clin Orthop Relat Res 472, 2440–2447 (2014). https://doi.org/10.1007/s11999-013-3439-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-013-3439-z