Abstract
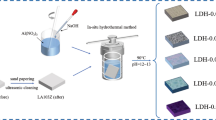
In this study, a series of ZnAl layered double hydroxide (ZnAl-LDH) thin films were synthesized on an AA6082 alloy by a single-step hydrothermal process at different synthesis parameters, including reaction temperature, reaction time, pH, and the relation between the LDH structural variations, and the corresponding corrosion resistance properties are briefly reported. The as-prepared synthetic coatings were characterized by scanning electron microscopy and X-ray diffraction. The corresponding corrosion properties were evaluated through potentiodynamic polarization curves and through electrochemical impedance spectra. The findings demonstrated that synthesis parameter variations impart an influential effect on the geometry of LDH, film thickness, and structural morphologies which have a significant impact on LDH corrosion resistance properties. The ZnAl-LDH corrosion resistance was found to increase with respect to extended high temperature and aging time, while the synthetic pH conditions also promoted LDH growth and film thickness, relative to lower-pH reaction conditions. The optimization results are attributed to high ZnAl-LDH corrosion resistance and act as a strong barrier film and ion-exchange surface area. The ZnAl-LDH films at 80°C-18r-6.5pH have shown much lower corrosion current density compared to bare AA6082, and a decrease of up to five orders of magnitude is observed.









Similar content being viewed by others
References
Davis, JR, (ed), Corrosion of Aluminum and Aluminum Alloys. ASM International, 1999
Winston Revie, R, Uhlig’s Corrosion Handbook, 3rd ed. John Willey & Sons Inc., New York, USA (2011)
Xhanari, K, Finšgar, M. “Organic Corrosion Inhibitors for Aluminum and Its Alloys in Chloride and Alkaline Solutions: A Review.” Arabian J. Chem. (2016)
Kumar, N, et al., “Effect of Cryorolling and Annealing on Recovery, Recrystallisation, Grain Growth and Their Influence on Mechanical and Corrosion Behaviour of 6082 Al Alloy.” Mater. Chem. Phys., 165 177–187 (2015)
Liang, WJ, et al., “General Aspects Related to the Corrosion of 6xxx Series Aluminium Alloys: Exploring the Influence of Mg/Si Ratio and Cu.” Corros. Sci., 76 119–128 (2013)
Larsen, MH, et al., “Intergranular Corrosion of Copper-Containing AA6x xx AlMgSi Aluminum Alloys.” J. Electrochem. Soc., 155 (11) C550–C556 (2008)
Schäfer, H, Stock, H-R, “Improving the Corrosion Protection of Aluminium Alloys Using Reactive Magnetron Sputtering.” Corros. Sci., 47 (4) 953–964 (2005)
Diesselberg, M, Stock, HR, Mayr, P, “Corrosion Protection of Magnetron Sputtered TiN Coatings Deposited on High Strength Aluminium Alloys.” Surf. Coat. Technol., 177 399–403 (2004)
Arenas, MA, Conde, A, De Damborenea, JJ, “Effect of Acid Traces on Hydrothermal Sealing of Anodising Layers on 2024 Aluminium Alloy.” Electrochim. Acta, 55 (28) 8704–8708 (2010)
Venugopal, A, Panda, R, Manwatkar, S, Sreekumar, K, Krishna, LR, Sundararajan, G, “Effect of Micro Arc Oxidation Treatment on Localized Corrosion Behaviour of AA7075 Aluminum Alloy in 3.5% NaCl Solution.” Trans. Nonferrous Met. Soc. China, 22 (3) 700–710 (2012)
Dalmoro, V, et al., “A Synergistic Combination of Tetraethylorthosilicate and Multiphosphonic Acid Offers Excellent Corrosion Protection to AA1100 Aluminum Alloy.” Appl. Surf. Sci., 273 758–768 (2013)
Voevodin, NN, Kurdziel, JW, Mantz, R, “Corrosion Protection for Aerospace Aluminum Alloys by Modified Self-Assembled Nanophase Particle (MSNAP) Sol–Gel.” Surf. Coat. Technol., 201 (3–4) 1080–1084 (2006)
Johansen, HD, Brett, CMA, Motheo, CMA, “Corrosion Protection of Aluminium Alloy by Cerium Conversion and Conducting Polymer Duplex Coatings.” Corros. Sci., 63 342–350 (2012)
Shi, H, Han, EH, Liu, F, Kallip, S, “Protection of 2024-T3 Aluminium Alloy by Corrosion Resistant Phytic Acid Conversion Coating.” Appl. Surf. Sci., 280, 325–331 (2013)
Leroux, F, Taviot-Guého, C, “Fine Tuning Between Organic and Inorganic Host Structure: New Trends in Layered Double Hydroxide Hybrid Assemblies.” J. Mater. Chem., 15 (35–36) 3628–3642 (2005)
Evans, DG, Duan, X, “Preparation of Layered Double Hydroxides and Their Applications as Additives in Polymers, as Precursors to Magnetic Materials and in Biology and Medicine.” Chem. Commun., 5 485–496 (2006)
Cavani, F, Trifiro, F, Vaccari, A, “Hydrotalcite-Type Anionic Clays: Preparation, Properties and Applications.” Catalysis Today, 11 (2) 173–301 (1991)
Xu, ZP, et al., “The Effect of Zn, Al Layered Double Hydroxide on Thermal Decomposition of Poly (Vinyl Chloride).” Polym. Degrad. Stab., 91 (12) 3237–3244 (2006)
Rives, V, Layered Double Hydroxides: Present and Future. Nova Publishers, 2001
Williams, GR, O’Hare, D, “Towards Understanding, Control and Application of Layered Double Hydroxide Chemistry.” J. Mater. Chem., 16 (30) 3065–3074 (2006)
Lei, X, Y, L, Z, F, Evans, DG, Duan, X, “Synthesis of Oriented Layered Double Hydroxide Thin Films on Sulfonated Polystyrene Substrates.” Chem. Letters, 34 (12) 1610–1611 (2005)
Chen, H, Zhang, F, Fu, S, Duan, X, “In Situ Microstructure Control of Oriented Layered Double Hydroxide Monolayer Films with Curved Hexagonal Crystals as Superhydrophobic Materials.” Adv. Mater., 18 (23) 3089–3093 (2006)
Wang, L, Li, C, Liu, M, Evans, DG, Duan, X, “Large Continuous, Transparent and Oriented Self-Supporting Films of Layered Double Hydroxides with Tunable Chemical Composition.” Chem. Commun., 2 123–125 (2007)
Zheludkevich, ML, Poznyak, SK, Rodrigues, LM, Raps, D, Hack, T, Dick, LF, Ferreira, MGS, “Active Protection Coatings with Layered Double Hydroxide Nanocontainers of Corrosion Inhibitor.” Corros. Sci., 52 (2) 602–611 (2010)
Zhang, F, Zhao, L, Chen, H, Xu, S, Evans, DG, Duan, X, “Corrosion Resistance of Superhydrophobic Layered Double Hydroxide Films on Aluminum.” Angewandte Chemie, 120 (13) 2500–2503 (2008)
Zeng, RC, Li, XT, Liu, ZG, Zhang, F, Li, SQ, Cui, HZ, “Corrosion Resistance of Zn–Al Layered Double Hydroxide/Poly (Lactic Acid) Composite Coating on Magnesium Alloy AZ31.” Front. Mater. Sci., 9 (4) 355–365 (2015)
Cao, Y, Zheng, D, Li, X, Lin, J, Wang, C, Dong, S, Lin, C, “Enhanced Corrosion Resistance of Superhydrophobic Layered Double Hydroxide Films with Long-Term Stability on Al Substrate.” ACS Appl. Mater. Interfaces, 10 (17) 15150–15162 (2018)
Tedim, J, Zheludkevich, ML, Salak, AN, Lisenkov, A, Ferreira, MGS, “Nanostructured LDH-container layer with active protection functionality”. J. Mater. Chem., 21 (39), 15464–15470 (2011)
Tedim, J, Zheludkevich, ML, Bastos, AC, Salak, AN, Lisenkov, AD, Ferreira, MGS. “Influence of Preparation Conditions of Layered Double Hydroxide Conversion Films on Corrosion Protection.” Electrochim. Acta, 117, 164–171 (2014)
Tedim, J, Bastos, AC, Kallip, S, Zheludkevich, ML, Ferreira, MGS. “Corrosion Protection of AA2024-T3 by LDH Conversion Films. Analysis of SVET Results.” Electrochim. Acta, 210, 215–224 (2016)
Buchheit, RG, Guan, H, Mahajanam, S, Wong, F, “Active Corrosion Protection and Corrosion Sensing in Chromate-Free Organic Coatings.” Prog. Org. Coat., 47 (3–4) 174–182 (2003)
Hoshino, K, Furuya, S, Buchheit, RG, “Effect of Solution pH on Layered Double Hydroxide Formation on Electrogalvanized Steel Sheets.” J. Mater. Eng. Perform., 1–8.
Hoshino, K, Furuya, S, Buchheit, RG, “Effect of NO3 – Intercalation on Corrosion Resistance of Conversion Coated Zn–Al–CO3 LDHs on Electrogalvanized Steel.” J. Electrochem. Soc., 165 (9) C461–C468 (2018)
Guo, X, Xu, S, Zhao, L, Lu, W, Zhang, F, Evans, DG, Duan, X, “One-Step Hydrothermal Crystallization of a Layered Double Hydroxide/Alumina Bilayer Film on Aluminum and Its Corrosion Resistance Properties.” Langmuir, 25 (17) 9894–9897 (2009)
Iqbal, MA, Fedel, M, “The Effect of the Surface Morphologies on the Corrosion Resistance of In Situ Growth MgAl-LDH Based Conversion Film on AA6082,” Surf. Coat. Technol. (2018). https://doi.org/10.1016/j.surfcoat.2018.08.006
Iqbal, M, Fedel, M, “Effect of Synthesis Conditions on the Controlled Growth of MgAl–LDH Corrosion Resistance Film: Structure and Corrosion Resistance Properties.” Coatings, 9 (1) 30 (2019)
Abderrazek, K, Srasra, NF, Srasra, E, “Synthesis and Characterization of [Zn–Al] Layered Double Hydroxides: Effect of the Operating Parameters.” J. Chinese Chem. Soc., 64 (3) 346–353 (2017)
Seftel, EM, Popovici, E, Mertens, M, De Witte, K, Van Tendeloo, G, Cool, P, Vansant, EF, “Zn–Al Layered Double Hydroxides: Synthesis, Characterization and Photocatalytic Application.” Microporous Mesoporous Mater., 113 (1–3) 296–304 (2008)
Ke, X, Bernal, SA, Provis, JL, “Uptake of Chloride and Carbonate by Mg–Al and Ca–Al Layered Double Hydroxides in Simulated Pore Solutions of Alkali-Activated Slag Cement.” Cement Concrete Res., 100 1–13 (2017)
Zhang, C, Luo, X, Pan, X, Liao, L, Wu, X, Liu, Y, “Self-Healing Li-Al Layered Double Hydroxide Conversion Coating Modified with Aspartic Acid for 6N01 Al Alloy.” Appl. Surf. Sci., 394 275–281 (2017)
Wang, F, Guo, Z, “Insitu Growth of du Rable Superhydrophobic Mg–Al Layered Double Hydroxides Nanoplatelets on Aluminum Alloys for Corrosion Resistance.” J. Alloys Compd., 767 382–391 (2018)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iqbal, M.A., Fedel, M. Effect of operating parameters on the structural growth of ZnAl layered double hydroxide on AA6082 and corresponding corrosion resistance properties. J Coat Technol Res 16, 1423–1433 (2019). https://doi.org/10.1007/s11998-019-00227-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11998-019-00227-0