Abstract
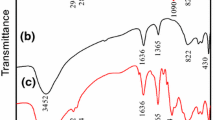
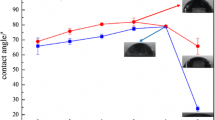
In this study, zinc aluminum hydrotalcite intercalated with 2-benzothiazolylthio-succinic acid (BTSA) as a container of corrosion inhibitor was prepared and incorporated in an epoxy coating. The HT-BTSA obtained was characterized using infrared spectroscopy and X-ray diffraction. BTSA release from HT-BTSA in 0.5 M NaCl solution was investigated using UV–Vis spectroscopy. The corrosion protection performance of the epoxy coating containing HT-BTSA was evaluated and compared to the pure epoxy coating and the epoxy coating containing HT by cathodic disbonding test, electrochemical impedance spectroscopy, salt spray test, and adhesion measurement. It was shown that the BTSA was intercalated in hydrotalcite and its loading was about 33.5%. The BTSA release from HT-BTSA increased with the pH of the NaCl solution. It was also shown that the presence of HT or HT-BTSA in epoxy coating improved the resistance to cathodic disbonding and the adhesion of the epoxy coating. Corrosion protection performance of epoxy coating containing HT-BTSA was higher than that of epoxy coating containing HT.










Similar content being viewed by others
References
Leggat, RB, Zhang, W, Buchheit, RG, Taylor, SR, “Performance of Hydrotalcite Conversion Treatments on AA2024-T3 When Used Within a Coating System.” Corrosion, 58 322–328 (2002)
Wang, H, Presuel, F, Kelly, RG, “Computational Modeling of Inhibitor Release and Transport from Multifunctional Organic Coatings.” Electrochim. Acta, 49 239–255 (2004)
Buchheit, RG, Guan, H, Mahajanam, S, Wong, F, “Active Corrosion Protection and Corrosion Sensing in Chromate-Free Organic Coatings.” Prog. Org. Coat., 47 174–182 (2003)
Mahajanam, PV, Buchheit, RG, “Characterization of Inhibitor Release from Zn-Al-[V10O28]6− Hydrotalcite Pigments and Corrosion Protection from Hydrotalcite-Pigmented Epoxy Coatings.” Corrosion, 64 230–240 (2008)
Zheludkevich, ML, Poznyak, SK, Rodrigues, LM, Raps, D, Hack, T, Dick, LF, Nunes, T, Ferreira, MGS, “Active Protection Coatings with Layered Double Hydroxide Nanocontainers of Corrosion Inhibitor.” Corros. Sci., 52 602–611 (2010)
Williams, G, McMurray, HN, “Inhibition of Filiform Corrosion on Polymer Coated AA2024-T3 by Hydrotalcite-Like Pigments Incorporating Organic Anions.” Electrochem. Solid-State Lett., 7 B13–B15 (2004)
Braig, A, “Advances in corrosion protection by organic coatings.” In: Sekine, I. (ed.) Electrochemical Society. Corrosion Division, pp. 18–30. (1998)
Hang, TTX, Truc, TA, Duong, NT, Pébère, N, Olivier, MG, “Layered Double Hydroxides as Containers of Inhibitors in Organic Coatings for Corrosion Protection of Carbon Steel.” Prog. Org. Coat., 74 343–348 (2012)
Hang, TTX, Truc, TA, Duong, NT, Vu, PG, Hoang, T, “Preparation and Characterization of Nanocontainers of Corrosion Inhibitor Based on Layered Double Hydroxides.” Appl. Clay Sci., 67 18–25 (2012)
Nyambo, C, Chen, D, Su, S, Wilkie, CA, “Variation of Benzyl Anions in MgAl-Layered Double Hydroxides: Fire and Thermal Properties in PMMA.” Polym. Degrad. Stab., 94 496–505 (2009)
ASTM B 117-09, Standard Practice for Operating Salt Spray (Fog) Apparatus. ASTM, Philadelphia, 2009
ASTM D1654, Standard Test Method for Evaluation of Painted or Coated Specimens Subjected to Corrosive Environments. ASTM, Philadelphia, 2008
ASTM D714, Evaluating Degree of Blistering of Paints. ASTM, Philadelphia, 1987
ASTM D4541-02, Standard Test Method for Pull-Off Strength of Coatings Using Portable Adhesion Testers. ASTM, Philadelphia, 2002
Xu, ZP, Braterman, PS, “Synthesis, Structure and Morphology of Organic Layered Double Hydroxide (LDH) Hybrids: Comparison Between Aliphatic Anions and Their Oxygenated Analogs.” Appl. Clay Sci., 48 235–242 (2010)
Simons, WW, The Sadtler Handbook of Infrared Spectra. Sadtler Research Laboratories Inc, Pennsylvania, 1978
Buchheit, RG, Guan, H, Mahajanam, S, Wong, F, “Active Corrosion Protection and Corrosion Sensing in Chromate-Free Organic Coatings.” Prog. Org. Coat., 47 174–182 (2003)
Qiu, DP, Hou, WG, “Synthesis and Characterization of Indole-3-Butyric Acid/Hydrotalcite-Like Compound Nanohybrids.” Colloids Surf. A: Physicochem. Eng. Aspects, 336 12–17 (2009)
Hussein, MZB, Zainal, Z, Yahaya, AH, Foo, DWV, “Controlled Release of a Plant Growth Regulator, a-Naphthaleneacetate from the Lamella of Zn–Al-Layered Double Hydroxide Nanocomposite.” J. Controll. Rel., 82 417–427 (2002)
Radha, AV, Kamath, PV, Shivakumara, C, “Mechanism of the Anion Exchange Reactions of the Layered Double Hydroxides (LDHs) of Ca and Mg with Al.” Solid State Sci., 7 1180–1187 (2005)
Beaunier, L, Epelboin, I, Lestrade, JC, Takenouti, H, “Electrochemical and Scanning Microscope Study of Pained Fe.” Surf. Technol., 4 237–254 (1976)
Pébère, N, Picaud, T, Duprat, M, Dabosi, F, “Evaluation of Corrosion Performance of Coated Steel by the Impedance Technique.” Corros. Sci., 29 1073–1086 (1989)
Williams, G, McMurray, HN, “Anion-Exchange Inhibition of Filiform Corrosion on Organic Coated AA2024-T3 Aluminum Alloy by Hydrotalcite-Like Pigments.” Electrochem. Solid-State Lett., 6 B9–B11 (2003)
Tedim, J, Kuznetsova, A, Salak, AN, Montemor, F, Snihirova, D, Pilz, M, Zheludkevich, ML, Ferreira, MGS, “Zn–Al Layered Double Hydroxides as Chloride Nanotraps in Active Protective Coatings.” Corros. Sci., 55 1–4 (2012)
Deflorian, F, Rossi, S, “An EIS Study of Ion Diffusion Through Organic Coatings.” Electrochim. Acta, 51 1736–1744 (2006)
Leng, A, Streckel, H, Stratmann, M, “The Delamination of Polymeric Coatings from Steel. Part 1: Calibration of the Kelvin Probe and Basic Delamination Mechanism.” Corros. Sci., 41 547–578 (1999)
Kittel, J, Celati, N, Keddam, M, Takenouti, H, “Influence of the Coating–Substrate Interactions on the Corrosion Protection: Characterisation by Impedance Spectroscopy of the Inner and Outer Parts of a Coating.” Prog. Org. Coat., 46 135–147 (2003)
Bierwagen, GP, Tallman, D, Li, J, He, L, Jeffcoate, C, “EIS Studies of Coated Metals in Accelerated Exposure.” Prog. Org. Coat., 46 148–158 (2003)
De Rosa, RL, Earl, DA, Bierwagen, GP, “Statistical Evaluation of EIS and ENM Data Collected for Monitoring Corrosion Barrier Properties of Organic Coatings on Al-2024-T3.” Corros. Sci., 44 1607–1620 (2002)
Hinderliter, BR, Croll, SG, Tallman, DE, Su, Q, Bierwagen, GP, “Interpretation of EIS Data from Accelerated Exposure of Coated Metals Based on Modeling of Coating Physical Properties.” Electrochim. Acta, 51 4505–4515 (2006)
Acknowledgments
This research was funded by the Vietnam National Foundation for Science and Technology Development (NAFOSTED) under Grant number 104.01-2012.15. The authors gratefully acknowledge the support of Vietnam Academy of Science and Technology and Thai-French Innovation Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hang, T.T.X., Duong, N.T., Truc, T.A. et al. Effects of hydrotalcite intercalated with corrosion inhibitor on cathodic disbonding of epoxy coatings. J Coat Technol Res 12, 375–383 (2015). https://doi.org/10.1007/s11998-014-9642-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11998-014-9642-3