Abstract
Background
This study was designed to provide comparative information on the safety and efficacy of injection with collagenase clostridium histolyticum (CCH) and fasciectomy for patients with Dupuytren’s contracture (DC).
Methods
A single-center, retrospective, observational, longitudinal chart review was conducted of 25 patients treated with CCH injections and 21 patients undergoing fasciectomy. Patients were assessed at 1 week, monthly for 3 months and then yearly for a minimum of 2 years after treatment for changes in contracture and range of motion, time to return to work/normal activities, patient satisfaction, and Disabilities of Arm, Shoulder and Hand (DASH) score.
Results
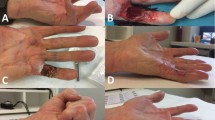
Post-procedure follow-up averaged 32 months for the injection group compared with 39 months for fasciectomy group. For the CCH group, the mean postinjection contracture was 3.6° for the metacarpophalangeal and 17.5° for the proximal interphalangeal joints compared with 3.7° and 8.1° in the fasciectomy group, respectively. Patients treated with injections returned to normal activities after a mean of 1.9 days compared with 37.4 days for fasciectomy patients (p < 0.0001). DASH scores for 13 CCH and 15 fasciectomy patients were obtained. The mean DASH score was significantly lower in the injection group in the first 3 months (p < 0.01). At the 2-year follow-up visit, patients were satisfied with their outcomes following either treatment (92 % and 96 % of CCH and fasciectomy patients, respectively).
Conclusion
CCH injections are safe and effective and may be a viable alternative to fasciectomy for treating DC. It also allows earlier return to work and daily activities.



Similar content being viewed by others
References
Bainbridge C, Dahlin LB, Szczypa PP, Cappelleri JC, Guerin D, Gerber RA. Current trends in the surgical management of Dupuytren’s disease in Europe: an analysis of patient charts. Eur Orthop Traumatol. 2012;3:31–41.
Becker GW, Davis TRC. The outcome of surgical treatments for primary Dupuytren’s disease—a systematic review. J Hand Surg Br. 2010;35:623–6.
Bulstrode NW, Jemec B, Smith PJ. The complications of Dupuytren’s contracture surgery. J Hand Surg Am. 2005;30:1021–5.
Coert JH, Nerin JP, Meek MF. Results of partial fasciectomy for Dupuytren disease in 261 consecutive patients. Ann Plast Surg. 2006;57:13–7.
Crean SM, Gerber RA, Hellio Le Graverand MP, Boyd DM, Cappelleri JC. The efficacy and safety of fasciectomy and fasciotomy for Dupuytren’s contracture in European patients: a structured review of published studies. J Hand Surg Br. 2011;36:396–407.
Denkler K. Surgical complications associated with fasciectomy for Dupuytren’s disease: a 20-year review of the English literature. Eplasty. 2010;10:116–33.
Desai SS, Hentz VR. Collagenase Clostridium histolyticum for Dupuytren’s contracture. Expert Opin Biol Ther. 2010;10:1395–404.
Foucher G, Cornil C, Lenoble E. Open palm technique for Dupuytren’s disease. A five-year follow-up. Ann Chir Main Memb Super. 1992;11:362–6.
Gilpin D, Coleman S, Hall S, Houston A, Karrasch J, Jones N. Injectable collagenase Clostridium histolyticum: a new nonsurgical treatment for Dupuytren’s disease. J Hand Surg Am. 2010;35:2027–38.
Herweijer H, Dijkstra PU, Nicolai JP, Van der Sluis CK. Postoperative hand therapy in Dupuytren’s disease. Disabil Rehabil. 2007;29:1736–41.
Hurst LC, Badalamente MA, Hentz VR, Hotchkiss RN, Kaplan FT, Meals RA, et al. Injectable collagenase clostridium histolyticum for Dupuytren’s contracture. N Engl J Med. 2009;361:968–79.
Jerosch-Herold C, Shepstone L, Chojnowski AJ, Larson D, Barrett E, Vaughan SP. Night-time splinting after fasciectomy or dermo-fasciectomy for Dupuytren’s contracture: a pragmatic, multi-centre, randomised controlled trial. BMC Musculoskelet Disord. 2011;12:136.
Mackin EJ, Skirven TM. Hand therapy. In: Tubiana R, Leclercq C, Hurst LC, et al., editors. Dupuytren’s disease. United Kingdom: Martin Dunitz, Ltd.; 2010:251–63.
Mavrogenis AF, Spyridonos SG, Ignatiadis IA, Antonopoulos D, Papagelopoulos PJ. Partial fasciectomy for Dupuytren’s contractures. J Surg Orthop Adv. 2009;18:106–10.
McGrouther DA. Dupuytren’s contracture. In: Green DP, Hotchkiss RN, Pederson WC, et al., editors. Green’s operative hand surgery. 5th ed. Philadelphia, PA: Elsevier Churchill Livingstone; 2005. p. 159–85.
O’Gorman DB, Vi L, Gan BS. Molecular mechanisms and treatment strategies for Dupuytren’s disease. Ther Clin Risk Manage. 2010;6:383–90.
Auxilium Pharmaceuticals, Inc. Xiaflex® (collagenase clostridium histolyticum) [prescribing information]. Malvern, PA: Auxilium Pharmaceuticals, Inc.; 2012.
Rayan GM. Dupuytren’s disease: anatomy, pathology, presentation, and treatment. J Bone Joint Surg Am. 2007;89:189–98.
Rayan GM. Nonoperative treatment of Dupuytren’s disease. J Hand Surg Am. 2008;33:1208–10.
Skrepnik NV, Waller P, Kushner H, Nguyen D. Collagenase Clostridium histolyticum therapy for Dupuytren's contracture in Europe, Australia and US. Presented at American Academy of Orthopaedic Surgeons 2011 Annual Meeting, February 15–19, 2011, San Diego, CA. Abstract 4601. Available at: http://www3.aaos.org/education/anmeet/anmt2011/proceedings.pdf. Accessed July 3, 2012.
Thomas A, Bayat A. The emerging role of Clostridium histolyticum collagenase in the treatment of Dupuytren disease. Ther Clin Risk Manage. 2010;6:557–72.
Vigroux JP, Valentin P. A natural history of Dupuytren’s contracture treated by surgical fasciectomy: the influence of diathesis (76 hands reviewed at more than 10 years). Ann Chir Main Memb Super. 1992;11:367–74.
Wilson GR. Current surgical treatment of Dupuytren’s disease. Br J Clin Pract. 1997;51:106–10.
Acknowledgement
The author thanks Jennifer Kent, PhD, of MedVal Scientific Information Services, LLC, for providing medical writing and editorial assistance. Independent statistical analysis was provided by Greg Maislin of Biomedical Statistical Consulting. This manuscript was prepared according to the International Society for Medical Publication Professionals’ Good Publication Practice for Communicating Company-Sponsored Medical Research: the GPP2 Guidelines. Study drug was provided by Auxilium Pharmaceuticals, and support for the preparation of this manuscript was provided by Auxilium Pharmaceuticals.
Disclosure
The author of this manuscript is a consultant for Auxilium Pharmaceuticals.
Statement of Human Rights
The procedures described in this manuscript were in accordance with the ethical standards of the local and national responsible committees on human experimentation and with the Helsinki Declaration of 1975 as revised in 2000.
Statement of Informed Consent
All patients included in this study were given detailed informed consent. All patients expressed their understanding of the details of the consents before signing then. Every patient signed the informed consent.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Naam, N.H. Functional outcome of collagenase injections compared with fasciectomy in treatment of Dupuytren’s contracture. HAND 8, 410–416 (2013). https://doi.org/10.1007/s11552-013-9540-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11552-013-9540-7