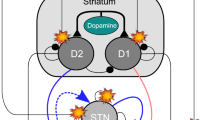
Abstract
This work represents an attempt to elucidate the neurochemical processes in the basal ganglia by mathematical modelling. The correlation between neurochemistry and electrophysiology has been used to construct a dynamical system based on the basal ganglia’s network structure. Mathematical models were constructed for different physical scales to reformulate the neurochemical and electrophysiological behaviour from synapses up to multi-compartment systems. Transformation functions have been developed to transit between the different scales. We show through numerical simulations that this network produces oscillations in the electrical potentials as well as in neurotransmitter concentrations. In agreement with pharmacological experiments, a parameter sensitivity analysis reveals temporary changes in the neurochemical and electrophysiological systems after single exposure to antipsychotic drugs. This behaviour states the structural stability of the system. The correlation between the neurochemical dynamics and drug-induced behaviour provides the perspective for novel neurobiological hypotheses.
Similar content being viewed by others
References
Aharon, S., Parnas, H., Parnas, I., 1994. The magnitude and significance of Ca2+ domains for release of neurotransmitters. Bull. Math. Biol. 56(6), 1095–1119.
Alexander, G.E., DeLong, M.R., Strick, P.L., 1986. Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu. Rev. Neurosci. 9, 357–381.
Betarbet, R., Turner, R., Chockkan, V., DeLong, M.R., Allers, K.A., Walters, J., Levey, A.I., Greenamyre, J.T., 1997. Dopaminergic neurons intrinsic to the primate striatum. J. Neurosci. 17(17), 6761–6768.
Bevan, M., Magill, P.J., Terman, D., Bolam, J.P., Wilson, C.J., 2002. Move to the rhythm: Oscillations in the subthalamic nucleus-external globus pallidus network. Trends Neurosci. 25, 523–531.
Boraud, T., Goldberg, P., Brown, J.A., Graybiel, A.M., Magill, P.J., 2005. Oscillations in the basal ganglia: The good, the bad, and the unexpected. The Basal Ganglia VIII, 3–25.
Carlsson, A., 1988. The current statues of the dopamine hypothesis of schizophrenia. Neuropsychopharm 1, 179–186.
Carlsson, M., Carlsson, A., 1990. Interactions between glutamatergic and monoaminergic systems within the basal ganglia—implications for schizophrenia and Parkinson’s disease. TINS 13(7), 272–276.
Carlsson, A., Waters, N., Carlsson, M.L., 1999. Neurotransmitter interactions in schizophrenia-Therapeutic implications. Biol. Psychiatry 46, 1388–1395.
Cassim, F., Labyt, E., Devos, D., Defebvre, L., Destexhe, A., Derambure, P., 2002. Relationship between oscillations in the basal ganglia and synchronisation of cortical activity. Epileptic Disord. 4, 31–45.
Chesselet, M.F., 2002. Dopamine–GABA Interactions. Dopamine in the CNS 2, 1st edn., pp. 151–172. Springer, Berlin.
Coyle, J.T., 2006. Glutamate and Schizophrenia: Beyond the Dopamine Hypothesis. Cell. Mol. Neurobiol. 26(4–6), 365–384.
Destexhe, A., Mainen, Z.F., Sejnowski, T.J., 1994. Synthesis of models for excitable membranes, synaptic transmission and neuromodulation using a common kinetic formalism. J. Comput. Neurosci. 1, 195–230.
Destexhe, A., Mainen, Z.F., Sejnowski, T.J., 1998. Kinetic models of synaptic transmission. In: Methods in Neuronal Modeling, 2nd edn. MIT Press, Cambridge.
DiFiglia, M., 1987. Synaptic organization of cholinergic neurons in the monkey neostriatum. J. Comput. Neurol. 255(2), 245–258.
DiFiglia, M., Pasik, P., Pasik, T., 1976. A Golgi study of neuronal types in the neostriatum of monkeys. Brain Res. 114(2), 245–256.
E, W., Engquist, B., 2003. The heterogeneous multi-scale methods. Commun. Math. Sci. 1, 87–133.
Frank, M.J., (2004). Dynamic dopamine modulation of striato-cortical circuits in cognition: Converging neuropsychological, psychopharmacological and computational studies. PhD thesis.
Frankle, W.G., Lerma, J., Laruelle, M., 2003. The synaptic hypothesis of schizophrenia. Neuron 39(2), 205–216.
Fujimoto, K., Kita, H., 1993. Response characteristics of subthalamic neurons to the stimulation of the sensorimotor cortex in the rat. Brain Res. 609, 185–192.
Gerfen, C., Wilson, C., 1996. The basal ganglia. In: Swanson, L., Bjorklund, A., Hokfelt, T. (Eds.), Handbook of Chemical Neuroanatomy. Integrated Systems of the CNS, Pt III, vol. 12, pp. 371–468. Elsevier, Amsterdam.
Globus, M.Y.T., Busto, R., Dietrich, W.D., Martinez, E., Valdes, I., Ginsberg, M.D., 1988. Effect of ischemia on the in vivo release of striatal dopamine, glutamate, and aminobutyric acid studied by intracerebral microdialysis. J. Neurochem. 51(5), 1455–1464.
Graveland, G.A., Williams, R.S., DiFiglia, M., 1985. A Golgi study of the human neostriatum: Neurons and afferent fibers. J. Comput. Neurol. 234(3), 317–333.
Graybiel, A.M., Ragsdale, C.W. Jr., 1978. Histochemically distinct compartments in the striatum of human, monkeys, and cat demonstrated by acetylthiocholinesterase staining. Proc. Natl. Acad. Sci. USA 75(11), 5723–5726.
Graybiel, A.M., Pickel, V.M., Joh, T.H., Reis, D.J., Ragsdale, C.W. Jr., 1981. Direct demonstration of a correspondence between the dopamine islands and acetylcholinesterase patches in the developing striatum. Proc. Natl. Acad. Sci. USA 78(9), 5871–5875.
Haken, H., 2004. Synergetics: Introduction and Advanced Topics (Physics and Astronomy Online Library). Springer, Berlin.
Heine, M., Groc, L., Frischknecht, R., Béïque, J.C., Lounis, B., Rumbaugh, G., Huganir, R.L., Cognet, L., Choquet, D., 2008. Science 320(5873), 201–205.
Humphries, M.D., Stewart, R.D., Gurney, K.N., 2006. A physiologically plausible model of action selection and oscillatory activity in the basal ganglia. J. Neurosci. 26(50), 12921–12942.
Hutchinson, P.J., O’Connell, M.T., Al-Rawi, P.G., Maskell, L.B., Kett-White, R., Gupta, A.K., Kirkpatrick, P.J., Pickard, J.D., 2000. Clinical cerebral microdialysis: a methodological study. J. Neurosurg. 93, 37–43.
Hutchinson, P.J., O’Connell, M.T., Kirkpatrick, P.J., Pickard, J.D., 2002. How can we measure substrate, metabolite and neurotransmitter concentrations in the human brain? Physiol. Meas. 23, R75–R109.
Kanthan, R., Shuaib, A., Griebel, R., Miyashita, H., 1995. Intracerebral human microdialysis. In vivo study of an acute focal ischemic model of the human brain. Stroke 26, 870–873.
Kita, H., Kitai, S.T., 1991. Intracellular study of rat globus pallidus neurons: membrane properties and responses to neostriatal, subthalamic and nigral stimulation. Brain Res. 564, 296–305.
Koch, C., 2004. Biophysics of Computation: Information Processing in Single Neurons (Computational Neuroscience). Oxford University Press, London.
Konradi, C., Cepeda, C., Levine, M.S., 2002. Dopamine–glutamate interactions. In: Dopamine in the CNS 2, 1st edn., pp. 117–133. Springer, Berlin.
Leblois, A., Boraud, T., Meissner, W., Bergman, H., Hansel, D., 2006. Competition between feedback loops underlies normal and pathological dynamics in the basal ganglia. J. Neurosci. 26(13), 3567–3583.
Lee, S.H., Wynn, J.K., Green, M.F., Kim, H., Lee, K.J., Nam, M., Park, J.K., Chung, Y.C., 2006. Quantitative EEG and low resolution electromagnetic tomography (LORETA) imaging of patients with persistent auditory hallucinations. Schizophr. Res. 83(2–3), 111–119.
Levin, S.A., Chao, D., 1999. Herding behaviour: The emergence of large-scale phenomena from local interactions. In: Differ. Equ. with Appl. Biol. Fields Institute Communications, pp. 81–96. AMS, Providence.
Light, G.A., Hsu, J.L., Hsieh, M.H., Meyer-Gomes, K., Sprock, J., Swerdlow, N.R., Braff, D.L., 2006. Gamma band oscillations reveal neural network cortical coherence dysfunction in schizophrenia patients. Biol. Psychiatry 60(11), 1231–1240.
Magill, P.J., Sharott, A., Bolam, J.P., Brown, P., 2004. Brain state-dependency of coherent oscillatory activity in the cerebral cortex and basal ganglia of the rat. J. Neurophysiol. 92, 2122–2136.
Marin, O., Anderson, S.A., Rubenstein, J.L., 2000. Origin and molecular specification of striatal interneurons. J. Neurosci. 20(16), 6063–6076.
Meyerson, B.A., Linderoth, B., Karlsson, H., Ungerstedt, U., 1990. Microdialysis in the human brain: extracellular measurements in the thalamus of Parkinsonian patients. Life Sci. 46, 301–308.
Moghaddam, B., 2003. Bringing order to the glutamate chaos in schizophrenia. Neuron 40, 881–884.
Moghaddam, B., Adams, B.W., 1998. Reversal of phencyclidine effects by a group II metabotropic glutamate receptor agonist in rats. Science 281, 1349–1352.
Moghaddam, B., Krystal, J.H., 2003. The neurochemistry of schizophrenia In: Schizophrenia, 2nd edn., pp. 349–364. Blackwell Science, Oxford.
Nakanishi, H., Kita, H., Kitai, S.T., 1987. Intracellular study of rat substantia nigra pars reticulata neurons in an in vitro slice preparation: electrical membrane properties and response characteristics to subthalamic stimulation. Brain Res. 437, 45–55.
Noori, H.R., 2008. The predominance of electric transport in synaptic transmission. In: Nature Neuroscience Proceedings.
Noori, H.R., 2009. Averaging transformations of synaptic potentials on networks. Preprint.
Parnas, H., Hovav, G., Parnas, I., 1989. Effect of Ca2+ diffusion on the time course of neurotransmitter release. Biophys. J. 55, 859–874.
Parnas, H., Segel, L., Dudel, J., Parnas, I., 2000. Autoreceptors, membrane potential and the regulation of transmitter release. Trends Neurosci. 23, 60–68.
Pavliotis, G.A., Stuart, A.M., 2008. Multiscale Methods: Averaging and Homogenization. Springer, Berlin.
Pediaditakis, N., 2006. Considering the major mental disorders as clinical expressions of periodic pathological oscillations of the overall operating mode of brain function. Med. Hypotheses 67(2), 395–400.
Ragsdale, C.W. Jr., Graybiel, A.M., 1990. A simple ordering of neocortical areas established by the compartmental organization of their striatal projections. Proc. Natl. Acad. Sci. USA 87(16), 6196–6199.
Ronne-Engstrom, E., Hillered, L., Flink, R., Spannare, B., Ungerstedt, U., Carlson, H., 1992. Intracerebral microdialysis of extracellular amino acids in the human epileptic focus. J. Cereb. Blood Flow Metab. 12, 873–876.
Rubin, J., Terman, D., 2002. Geometric singular perturbation analysis of neuronal dynamics. In: Fiedler, B. (Ed.), Handbook of Dynamical Systems II: Toward Applications, pp. 93–146. Elsevier, Amsterdam.
Rubin, J., Terman, D., 2004. High frequency stimulation of the subthalamic nucleus eliminates pathological thalamic rhythmicity in a computational model. J. Comput. Neurosci. 16, 211–235.
Sela, R., Segel, L., Parnas, I., Parnas, H., 2005. Release of neurotransmitter induced by Ca2+-uncaging: Reexamination of the Ca-voltage hypothesis for release. J. Comput. Neurosci. 19, 5–20.
Shenton, M.E., Dickey, C.C., Frumin, M., McCarley, R.W., 2001. A review of MRI findings in schizophrenia. Schizophr. Res. 49(1–2), 1–52.
Sunol, C., Tusell, J.M., Gelpi, E., Rodriguez-Farre, E., 1988. Convulsant effect of lindane and regional brain concentration of GABA and dopamine. Toxicology 49, 247–252.
Ungerstedt, U., 1984. Measurement of neurotransmitter release by intracranial dialysis. In: Measurement of Neurotransmitter Release in Vivo, pp. 81–105. Wiley, Chichester.
Van der Stelt, O., Belger, A., Lieberman, J.A., 2004. Macroscopic fast neuronal oscillations and synchrony in schizophrenia. Proc. Natl. Acad. Sci. USA 101(51), 17567–17568.
Wassle, H., Chun, M.H., 1988. Dopaminergic and indoleamine-accumulating amacrine cell express GABA-like immunoreactivity in the cat retina. J. Neurosci. 8, 3383–3394.
Winograd, M., Destexhe, A., Sanchez-Vives, M.V., 2008. Hyperpolarization-activated graded persistent activity in the prefrontal cortex. PNAS 105(20), 7298–7303.
Yelnik, J., Francois, C., Percheron, G., Tande, D., 1991. Morphological taxonomy of the neurons of the primate striatum. J. Comput. Neurol. 313(2), 273–294.
Yusim, K., Parnas, H., Segel, L., 1999. Theory of fast neurotransmitter release control based on voltage-dependent interaction between autoreceptors and proteins of the exocytotic machinery. Bull. Math. Biol. 61, 701–725.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Noori, H.R., Jäger, W. Neurochemical Oscillations in the Basal Ganglia. Bull. Math. Biol. 72, 133–147 (2010). https://doi.org/10.1007/s11538-009-9441-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-009-9441-7