Abstract
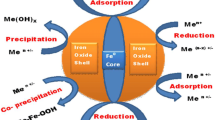
We report herein the near complete reduction of perchlorate (ClO \(_{4}^{-}\)) to chloride by nanoscale iron particles. The nanoparticles also reduce chlorate (ClO \(_{3}^-\)), chlorite (ClO \(_{2}^-\)) and hypochlorite (ClO \(^{-}\)) to chloride. No reaction was observed with microscale iron powder under identical conditions. The temperature sensitivity of the perchlorate-nanoparticle reaction is evidenced by progressively increasing rate constant values of 0.013, 0.10, 0.64 and 1.52 mg perchlorate per g nanoparticles per hour (mg-g-1-hr-1), respectively, at temperatures of 25, 40, 60 and 75°C. The activation energy of perchlorate-iron reaction was calculated to be 79.02 ± 7.75 kJ/mole. Despite favorable thermodynamics, the relatively large activation energy for this reaction suggests that perchlorate reduction is limited by the slow kinetics. The nanoscale iron particles may represent a potential treatment method for perchlorate-contaminated water.
Similar content being viewed by others
References
B.V. Aken J.L. Schnoor (2002) ArticleTitleEvidence of Perchlorate (ClO −4 ) Reduction in Plant Tissues (Poplar Tree) Using Radio-Labeled 36ClO −4 Environ. Sci. Technol., 36 IssueID(12) 2783–2788
California Department of Health Services. 1997. Perchlorate in␣California Drinking Water
J.D. Coates R.A. Bruce J.D. Haddock (1998) ArticleTitleAnoxic bioremediation of hydrocarbons Nature 396 730 Occurrence Handle10.1038/25470 Occurrence Handle9874368
F.A. Cotton G. Wilkinson C.A. Murillo M. Bochumann (1999) Advanced Inorganic Chemistry EditionNumber6 Wiley New York 560–563
F.R. Duke P.R. Quinney (1954) ArticleTitleThe kinetics of the reduction of perchlorate ion by Ti(III) in dilute solution J. Am. Chem. Soc. 76 3800–3803
D.W. Elliott W. Zhang (2001) ArticleTitleField assessment of nanoscale bimetallic particles for groundwater treatment Environ. Sci. Technol. 35 IssueID(24) 4922–4926 Occurrence Handle10.1021/es0108584 Occurrence Handle11775172
Espenson J., 1997. In Proceedings of the Symposium on Biological and Chemical Reduction of Perchlorate and Chlorate. Cincinnati, OH: US EPA National Risk Management Research Laboratory.
W.R. King C.S. Garner (1954) ArticleTitleKinetics of the oxidation of vanadium(II) and vanadium(III) ions by perchlorate J. Phys. Chem. 58 29–33 Occurrence Handle10.1021/j150511a007
T.W. Kallen J.E. Earley (1971) ArticleTitleReduction of Perchlorate in by aquoruthenium(II) Inorg. Chem. 10 1152–1155 Occurrence Handle10.1021/ic50100a010
H. Lien W. Zhang (1999) ArticleTitleTransformation of chlorinated methanes by nanoscale iron particles J. Environ. Eng. 125 1042–1047 Occurrence Handle10.1061/(ASCE)0733-9372(1999)125:11(1042)
H. Lien (2000) Nanoscale bimetallic particles for dehalogenation of halogenated aliphatic compounds Lehigh University Bethlehem, PA
H. Lien W. Zhang (2005) ArticleTitleReactions of Chlorinated Methanes with Nanoscale Metal Particles J. of Environ. Eng. 131 IssueID(1) 4–10 Occurrence Handle10.1061/(ASCE)0733-9372(2005)131:1(4)
E.B. Logan (2001) ArticleTitleAssessing the outlook for perchlorate remediation Environ. Eng. 35 482A–487A
A.G. Moore C.H. De Leon T.M. Young (2003) ArticleTitleRate and extent of aqueous perchlorate removal by iron surface Environ. Sci. Technol. 37 IssueID(14) 3189–3198 Occurrence Handle10.1021/es026007t Occurrence Handle12901669
V.A. Nzengung Ch-H. Wang G. Harvey (1999) ArticleTitlePlant-Mediated transformation of perchlorate into chloride Environ. Sci. Technol. 33 IssueID(9) 1470–1478 Occurrence Handle10.1021/es981234+
E. Stokstad (2005) ArticleTitleDebate Continues Over Safety of Water Spiked With Rocket Fuel Science. 307 507 Occurrence Handle10.1126/science.307.5709.507 Occurrence Handle15681361
Ch-M. Su R.W. Puls (1999) ArticleTitleKinetics of trichloroethene reduction by zero-valent iron and Tin: pretreatment of effect, apparent activation energy, and intermediate products Environ. Sci. Technol. 33 IssueID(1) 163–168 Occurrence Handle10.1021/es980481a
U. S. Environmental Protection Agency. 1998. Drinking Water Contaminant List. EPA Document No. 815-F-98–002; GPO: Washington, DC
E.T. Urbansky M.R. Schock (1999) ArticleTitleIssues in managing the risks associated with perchlorate in drinking water J. Environ. Manage. 56 IssueID(2) 79–95 Occurrence Handle10.1006/jema.1999.0274
E.T. Urbansky (2000) Perchlorate in the Environment Kluwer Academic/Plenum Publishers New York
C. Wang W. Zhang (1997) ArticleTitleSynthesis nanoscale iron particles for rapid and complete dechlorination of TCE and PCBs Environ. Sci. Technol. 31 IssueID(7) 2154–2156 Occurrence Handle10.1021/es970039c
W. Zhang C. Wang H. Lien (1998) ArticleTitleTreatment of chlorinated organic contaminants with nanoscale bimetallic particles Catal. Today. 40 387–395 Occurrence Handle10.1016/S0920-5861(98)00067-4
W. Zhang (2003) ArticleTitleNanoscale Iron Particles for Environmental Remediation: An Overview J. of Nanoparticle Research 5 323–332 Occurrence Handle10.1023/A:1025520116015
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cao, J., Elliott, D. & Zhang, Wx. Perchlorate Reduction by Nanoscale Iron Particles. J Nanopart Res 7, 499–506 (2005). https://doi.org/10.1007/s11051-005-4412-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11051-005-4412-x