Abstract
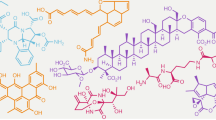
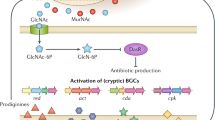
Natural product discovery in the microbial world has historically been biased toward aerobes. Recent in silico analysis demonstrates that genomes of anaerobes encode unexpected biosynthetic potential for natural products, however, chemical data on natural products from the anaerobic world are extremely limited. Here, we review the current body of work on natural products isolated from strictly anaerobic microbes, including recent genome mining efforts to discover polyketides and non-ribosomal peptides from anaerobes. These known natural products of anaerobes have demonstrated interesting molecular scaffolds, biosynthetic logic, and/or biological activities, making anaerobes a promising reservoir for future natural product discovery.


Similar content being viewed by others
References
Abken HJ, Tietze M, Brodersen J et al (1998) Isolation and characterization of methanophenazine and function of phenazines in membrane-bound electron transport of Methanosarcina mazei Gö1. J Bacteriol 180:2027–2032
Alsaker KV, Papoutsakis ET (2005) Transcriptional program of early sporulation and stationary-phase events in Clostridium acetobutylicum. J Bacteriol 187:7103–7118. https://doi.org/10.1128/JB.187.20.7103-7118.2005
Andrianasolo EH, Haramaty L, Rosario-Passapera R et al (2009) Ammonificins A and B, hydroxyethylamine chroman derivatives from a cultured marine hydrothermal vent bacterium, Thermovibrio ammonificans. J Nat Prod 72:1216–1219. https://doi.org/10.1021/np800726d
Andrianasolo EH, Haramaty L, Rosario-Passapera R et al (2012) Ammonificins C and D, hydroxyethylamine chromene derivatives from a cultured marine hydrothermal vent bacterium, Thermovibrio ammonificans. Mar Drugs 10:2300–2311. https://doi.org/10.3390/md10102300
Baltz RH (2017) Gifted microbes for genome mining and natural product discovery. J Ind Microbiol Biotechnol 44:573–588. https://doi.org/10.1007/s10295-016-1815-x
Baltz RH (2017) Molecular beacons to identify gifted microbes for genome mining. J Antibiot 70:639–646. https://doi.org/10.1038/ja.2017.1
Baltz RH (2018) Synthetic biology, genome mining, and combinatorial biosynthesis of NRPS-derived antibiotics: a perspective. J Ind Microbiol Biotechnol 45:635–649. https://doi.org/10.1007/s10295-017-1999-8
Behnken S, Hertweck C (2012) Anaerobic bacteria as producers of antibiotics. Appl Microbiol Biotechnol 96:61–67. https://doi.org/10.1007/s00253-012-4285-8
Behnken S, Lincke T, Kloss F et al (2012) Antiterminator-mediated unveiling of cryptic polythioamides in an anaerobic bacterium. Angew Chemie Int Ed 51:2425–2428. https://doi.org/10.1002/anie.201108214
Beifuss U, Tietze M (2005) Methanophenazine and other natural biologically active phenazines. In: Mulzer J (ed) Natural products synthesis II. Topics in current chemistry, vol 244. Springer, Berlin, pp 77–113. https://doi.org/10.1007/b96889
Beifuss U, Tietze M, Bäumer S, Deppenmeier U (2000) Methanophenazine: structure, total synthesis, and function of a new cofactor from methanogenic archaea. Angew Chemie Int Ed 39:2470–2472. https://doi.org/10.1002/1521-3773(20000717)39:14%3c2470:AID-ANIE2470%3e3.0.CO;2-R
Blankenfeldt W, Parsons JF (2014) The structural biology of phenazine biosynthesis. Curr Opin Struct Biol 29:26–33. https://doi.org/10.1016/J.SBI.2014.08.013
Blin K, Wolf T, Chevrette MG et al (2017) antiSMASH 4.0—improvements in chemistry prediction and gene cluster boundary identification. Nucl Acids Res 45:W36–W41. https://doi.org/10.1093/nar/gkx319
Challinor VL, Bode HB (2015) Bioactive natural products from novel microbial sources. Ann NY Acad Sci 1354:82–97. https://doi.org/10.1111/nyas.12954
Chiriac AI, Kloss F, Krämer J et al (2015) Mode of action of closthioamide: the first member of the polythioamide class of bacterial DNA gyrase inhibitors. J Antimicrob Chemother 70:2576–2588. https://doi.org/10.1093/jac/dkv161
Dabard J, Bridonneau C, Phillipe C et al (2001) Ruminococcin A, a new lantibiotic produced by a Ruminococcus gnavus strain isolated from human feces. Appl Environ Microbiol 67:4111–4118. https://doi.org/10.1128/AEM.67.9.4111-4118.2001
Ding C, Wu X, Auckloo B et al (2016) An unusual stress metabolite from a hydrothermal vent fungus Aspergillus sp. WU 243 induced by cobalt. Molecules 21:105. https://doi.org/10.3390/molecules21010105
Donia MS, Cimermancic P, Schulze CJ et al (2014) A systematic analysis of biosynthetic gene clusters in the human microbiome reveals a common family of antibiotics. Cell 158:1402–1414. https://doi.org/10.1016/J.CELL.2014.08.032
Dunbar K, Büttner H, Molloy E, Dell M, Kumpfmueller J, Hertweck C (2018) Genome editing reveals novel thiotemplated assembly of polythioamide antibiotics in anaerobic bacteria. Angew Chem Int Ed. https://doi.org/10.1002/anie.201807970
Ezaki M, Muramatsu H, Takase S et al (2008) Naphthalecin, a novel antibiotic produced by the anaerobic bacterium Sporotalea colonica sp. nov. J Antibiot (Tokyo) 61:207–212. https://doi.org/10.1038/ja.2008.30
Fajardo A, Martínez JL (2008) Antibiotics as signals that trigger specific bacterial responses. Curr Opin Microbiol 11:161–167. https://doi.org/10.1016/j.mib.2008.02.006
Gonzalez DJ, Lee SW, Hensler ME et al (2010) Clostridiolysin S, a post-translationally modified biotoxin from Clostridium botulinum. J Biol Chem 285:28220–28228. https://doi.org/10.1074/jbc.M110.118554
Guo C-J, Chang F-Y, Wyche TP et al (2017) Discovery of reactive microbiota-derived metabolites that inhibit host proteases. Cell 168(517–526):e18. https://doi.org/10.1016/J.CELL.2016.12.021
Guttenberger N, Blankenfeldt W, Breinbauer R (2017) Recent developments in the isolation, biological function, biosynthesis, and synthesis of phenazine natural products. Bioorg Med Chem 25:6149–6166. https://doi.org/10.1016/J.BMC.2017.01.002
Hans-Hartwig O, Schirmeister T (1997) Cysteine proteases and their inhibitors. Chem Rev 97:133–172. https://doi.org/10.1021/CR950025U
Herman NA, Kim SJ, Li JS et al (2017) The industrial anaerobe Clostridium acetobutylicum uses polyketides to regulate cellular differentiation. Nat Commun 8:1514. https://doi.org/10.1038/s41467-017-01809-5
Konno H (2012) Synthesis of bioactive natural products as protein inhibitors. Biosci Biotechnol Biochem 76:1257–1261
Letzel A-C, Pidot SJ, Hertweck C (2013) A genomic approach to the cryptic secondary metabolome of the anaerobic world. Nat Prod Rep 30:392–428. https://doi.org/10.1039/C2NP20103H
Letzel A-C, Pidot SJ, Hertweck C (2014) Genome mining for ribosomally synthesized and post-translationally modified peptides (RiPPs) in anaerobic bacteria. BMC Genomics 15:983. https://doi.org/10.1186/1471-2164-15-983
Lincke T, Behnken S, Ishida K et al (2010) Closthioamide: an unprecedented polythioamide antibiotic from the strictly anaerobic bacterium Clostridium cellulolyticum. Angew Chemie 122:2055–2057. https://doi.org/10.1002/ange.200906114
Lütke-Eversloh T, Bahl H (2011) Metabolic engineering of Clostridium acetobutylicum: recent advances to improve butanol production. Curr Opin Biotechnol 22:634–647. https://doi.org/10.1016/J.COPBIO.2011.01.011
Miari VF, Solanki P, Hleba Y et al (2017) In vitro susceptibility to closthioamide among clinical and reference strains of Neisseria gonorrhoeae. Antimicrob Agents Chemother 61:e00929-17. https://doi.org/10.1128/AAC.00929-17
Newman DJ, Cragg GM (2016) Natural products as sources of new drugs from 1981 to 2014. J Nat Prod 79:629–661. https://doi.org/10.1021/acs.jnatprod.5b01055
O’Brien J, Wright GD (2011) An ecological perspective of microbial secondary metabolism. Curr Opin Biotechnol 22:552–558. https://doi.org/10.1016/J.COPBIO.2011.03.010
Paredes CJ, Alsaker KV, Papoutsakis ET (2005) A comparative genomic view of clostridial sporulation and physiology. Nat Rev Microbiol 3:969–978. https://doi.org/10.1038/nrmicro1288
Petitdemange E, Caillet F, Giallo J, Gaudin C (1984) Clostridium cellulolyticum sp. nov., a cellulolytic, mesophilic species from decayed grass. Int J Syst Bacteriol 34:155–159
Pidot S, Ishida K, Cyrulies M, Hertweck C (2014) Discovery of clostrubin, an exceptional polyphenolic polyketide antibiotic from a strictly anaerobic bacterium. Angew Chemie Int Ed 53:7856–7859. https://doi.org/10.1002/anie.201402632
Powers JC, Asgian JL, Asgian Ekici OD, Özlem Doǧan Ekici A, James KE (2002) Irreversible inhibitors of serine, cysteine, and threonine proteases. Chem Rev 102:4639–4750. https://doi.org/10.1021/CR010182V
Price-Whelan A, Dietrich LEP, Newman DK (2006) Rethinking “secondary” metabolism: physiological roles for phenazine antibiotics. Nat Chem Biol 2:71–78. https://doi.org/10.1038/nchembio764
Raaijmakers JM, Mazzola M (2012) Diversity and natural functions of antibiotics produced by beneficial and plant pathogenic bacteria. Annu Rev Phytopathol 50:403–424. https://doi.org/10.1146/annurev-phyto-081211-172908
Rischer M, Raguž L, Guo H et al (2018) Biosynthesis, synthesis, and activities of barnesin A, a NRPS-PKS hybrid produced by an anaerobic epsilonproteobacterium. ACS Chem Biol Article ASAP. https://doi.org/10.1021/acschembio.8b00445
Schneider BA, Balskus EP (2018) Discovery of small molecule protease inhibitors by investigating a widespread human gut bacterial biosynthetic pathway. Tetrahedron 74:3215–3230. https://doi.org/10.1016/J.TET.2018.03.043
Shabuer G, Ishida K, Pidot SJ et al (2015) Plant pathogenic anaerobic bacteria use aromatic polyketides to access aerobic territory. Science 350:670–674. https://doi.org/10.1126/science.aac9990
Shi Y, Pan C, Auckloo BN et al (2017) Stress-driven discovery of a cryptic antibiotic produced by Streptomyces sp. WU20 from Kueishantao hydrothermal vent with an integrated metabolomics strategy. Appl Microbiol Biotechnol 101:1395–1408. https://doi.org/10.1007/s00253-016-7823-y
Siklos M, BenAissa M, Thatcher GRJ (2015) Cysteine proteases as therapeutic targets: does selectivity matter? A systematic review of calpain and cathepsin inhibitors. Acta Pharm Sin B 5:506–519. https://doi.org/10.1016/J.APSB.2015.08.001
Sturgen NO, Casida LE Jr (1962) Antibiotic production by anaerobic bacteria. Appl Microbiol 10:55–59
Vetriani C, Speck MD, Ellor SV et al (2004) Thermovibrio ammonificans sp. nov., a thermophilic, chemolithotrophic, nitrate-ammonifying bacterium from deep-sea hydrothermal vents. Int J Syst Evol Microbiol 54:175–181. https://doi.org/10.1099/ijs.0.02781-0
Westerik JO, Wolfenden R (1972) Aldehydes as inhibitors of papain. J Biol Chem 247:8195–8197
Woo J-T, Sigeizumi S, Yamaguchi K et al (1995) Peptidyl aldehyde derivatives as potent and selective inhibitors of cathepsin L. Bioorg Med Chem Lett 5:1501–1504
Zhu B, Wang X, Li L (2010) Human gut microbiome: the second genome of human body. Protein Cell 1:718–725. https://doi.org/10.1007/s13238-010-0093-z
Ziemert N, Alanjary M, Weber T (2016) The evolution of genome mining in microbes—a review. Nat Prod Rep 33:988–1005. https://doi.org/10.1039/C6NP00025H
Acknowledgements
This work was financially supported by the Energy Biosciences Institute, Alfred P. Sloan Foundation, the National Institutes of Health (DP2AT009148), and the Chan Zuckerberg Biohub Investigator Program.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Special Issue “Natural Product Discovery and Development in the Genomic Era 2019”.
Rights and permissions
About this article
Cite this article
Li, J.S., Barber, C.C. & Zhang, W. Natural products from anaerobes. J Ind Microbiol Biotechnol 46, 375–383 (2019). https://doi.org/10.1007/s10295-018-2086-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-018-2086-5