Abstract
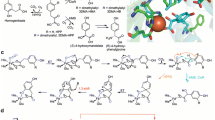
Molecular oxygen is utilized in numerous metabolic pathways fundamental for life. Mononuclear nonheme iron-dependent oxygenase enzymes are well known for their involvement in some of these pathways, activating O2 so that oxygen atoms can be incorporated into their primary substrates. These reactions often initiate pathways that allow organisms to use stable organic molecules as sources of carbon and energy for growth. From the myriad of reactions in which these enzymes are involved, this perspective recounts the general mechanisms of aromatic dihydroxylation and oxidative ring cleavage, both of which are ubiquitous chemical reactions found in life-sustaining processes. The organic substrate provides all four electrons required for oxygen activation and insertion in the reactions mediated by extradiol and intradiol ring-cleaving catechol dioxygenases. In contrast, two of the electrons are provided by NADH in the cis-dihydroxylation mechanism of Rieske dioxygenases. The catalytic nonheme Fe center, with the aid of active site residues, facilitates these electron transfers to O2 as key elements of the activation processes. This review discusses some general questions for the catalytic strategies of oxygen activation and insertion into aromatic compounds employed by mononuclear nonheme iron-dependent dioxygenases. These include: (1) how oxygen is activated, (2) whether there are common intermediates before oxygen transfer to the aromatic substrate, and (3) are these key intermediates unique to mononuclear nonheme iron dioxygenases?
Graphical Abstract



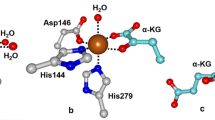
(from 4WHR.pdb)



Similar content being viewed by others
References
Hayaishi O, Katagiri M, Rothberg S (1955) J Am Chem Soc 77:5450–5451
Mason HS, Fowlks WL, Peterson E (1955) J Am Chem Soc 77:2914–2915
Hayaishi O (2008) J Biol Chem 283:19165–19175
Hayaishi O, Rothberg S, Mehler AH, Saito Y (1957) J Biol Chem 229:889–896
Geng J, Liu A (2014) Arch Biochem Biophys 544:18–26
Raven EL (2017) J Biol Inorg Chem. doi:10.1007/s00775-016-1412-5
Kal S, Que L Jr (2017) J Biol Inorg Chem. doi:10.1007/s00775-016-1431-2
Proshlyakov DA, John CW, McKracken C, Hausinger RP (2017) J Biol Inorg Chem. doi:10.1007/s00775-016-1406-3
Gibson DT and Subramanian V (1984) In: Gibson DT (ed). Microbial degradation of organic compounds, Marcel Dekker, Inc., New York
Seo JS, Keum YS, Li QX (2009) Int J Environ Res Public Health 6:278–309
Karlsson A, Parales JV, Parales RE, Gibson DT, Eklund H, Ramaswamy S (2003) Science 299:1039–1042
Wolfe MD, Parales JV, Gibson DT, Lipscomb JD (2001) J Biol Chem 276:1945–1953
Uhlin U, Eklund H (1994) Nature 370:533–539
Nordlund P, Eklund H (1993) J Mol Biol 232:123–164
Kauppi B, Lee K, Carredano E, Parales RE, Gibson DT, Eklund H, Ramaswamy S (1998) Structure 6:571–586
Carredano E, Karlsson A, Kauppi B, Choudhury D, Parales RE, Parales JV, Lee K, Gibson DT, Eklund H, Ramaswamy S (2000) J Mol Biol 296:701–712
Pavel EG, Martins LJ, Ellis WR, Solomon EI (1994) Chem Biol 1:173–183
Yang TC, Wolfe MD, Neibergall MB, Mekmouche Y, Lipscomb JD, Hoffman BM (2003) J Am Chem Soc 125:7056–7066
Martins BM, Svetlitchnaia T, Dobbek H (2005) Structure 13:817–824
Ohta T, Chakrabarty S, Lipscomb JD, Solomon EI (2008) J Am Chem Soc 130:1601–1610
Rivard BS, Rogers MS, Marell DJ, Neibergall MB, Chakrabarty S, Cramer CJ, Lipscomb JD (2015) Biochemistry 54:4652–4664
Ashikawa Y, Fujimoto Z, Usami Y, Inoue K, Noguchi H, Yamane H, Nojiri H (2012) BMC Struct Biol 12:15
Neibergall MB, Stubna A, Mekmouche Y, Münck E, Lipscomb JD (2007) Biochemistry 46:8004–8016
Bugg TDH, Ramaswamy S (2008) Curr Opin Chem Biol 12:134–140
Tarasev M, Rhames F, Ballou DP (2004) Biochemistry 43:12799–12808
Tarasev M, Ballou DP (2005) Biochemistry 44:6197–6207
Fu R, Gupta R, Geng J, Dornevil K, Wang S, Zhang Y, Hendrich MP, Liu A (2011) J Biol Chem 286:26541–26554
Geng J, Dornevil K, Liu A (2012) J Am Chem Soc 134:12209–12218
Chen K, Que L Jr (2001) J Am Chem Soc 123:6327–6337
Chen K, Costas M, Kim J, Tipton AK, Que L Jr (2002) J Am Chem Soc 124:3026–3035
Oloo WN, Fielding AJ, Que L Jr (2013) J Am Chem Soc 135:6438–6441
Prat I, Mathieson JS, Guell M, Ribas X, Luis JM, Cronin L, Costas M (2011) Nat Chem 3:788–793
Lyakin OY, Prat I, Bryliakov KP, Costas M, Talsi EP (2012) Catal Commun 29:105–108
Olivo G, Cusso O, Borrell M, Costas M (2017) J Biol Inorg Chem. doi:10.1007/s00775-016-1434-z
Leonowicz A, Matuszewska A, Luterek J, Ziegenhagen D, Wojtas-Wasilewska M, Cho NS, Hofrichter M, Rogalski J (1999) B. Fungal Genet Biol 27:175–185
Ruiz-Duenas FJ, Martinez AT (2009) Microb Biotechnol 2:164–177
Hayaishi O, Hashimoto K (1950) J Biochem (Tokyo) 37:71
Hayaishi O, Katagiri M, Rothberg S (1957) J Biol Chem 229:905–920
Stanier RY, Ingraham JL (1954) J Biol Chem 210:799–808
Hayaishi O (1966) Bacteriol Rev 30:720–731
Dagley S, Evans WC, Ribbons DW (1960) Nature 188:560–566
Kojima Y, Itada N, Hayaishi O (1961) J Biol Chem 236:2223–2228
Adachi K, Takeda Y, Senoh S, Kita H (1964) Biochim Biophys Acta 93:483–493
Lipscomb JD (2008) Curr Opin Struct Biol 18:644–649
Kovaleva EG, Neibergall MB, Chakrabarty S, Lipscomb JD (2007) Acc Chem Res 40:475–483
Vaillancourt FH, Bolin JT, Eltis LD (2006) Crit Rev Biochem Mol Biol 41:241–267
Abu-Omar MM, Loaiza A, Hontzeas N (2005) Chem Rev 105:2227–2252
Bugg TDH (2003) Tetrahedron 59:7075–7101
Solomon EI, Brunold TC, Davis MI, Kemsley JN, Lee S-K, Lehnert N, Neese F, Skulan AJ, Yang Y-S, Zhou J (2000) Chem Rev 100:235–349
Broderick JB (1999) Essays Biochem 34:173–189
Que L Jr, Ho RYN (1996) Chem Rev 96:2607–2624
Spence EL, Kawamukai M, Sanvoisin J, Braven H, Bugg TD (1996) J Bacteriol 178:5249–5256
Eltis LD, Bolin JT (1996) J Bacteriol 178:5930–5937
Han S, Eltis LD, Timmis KN, Muchmore SW, Bolin JT (1995) Science 270:976–980
Senda T, Sugiyama K, Narita H, Yamamoto T, Kimbara K, Fukuda M, Sato M, Yano K, Mitsui Y (1996) J Mol Biol 255:735–752
Dunwell JM (1998) Biotechnol Genet Eng Rev 15:1–32
Khuri S, Bakker FT, Dunwell JM (2001) Mol Biol Evol 18:593–605
Dunwell JM, Culham A, Carter CE, Sosa-Aguirre CR, Goodenough PW (2001) Trends Biochem Sci 26:740–746
Fetzner S (2012) Appl Environ Microbiol 78:2505–2514
Colabroy KL, Zhai H, Li T, Ge Y, Zhang Y, Liu A, Ealick SE, McLafferty FW, Begley TP (2005) Biochemistry 44:7623–7631
Liu F, Geng J, Gumpper RH, Barman A, Davis I, Ozarowski A, Hamelberg D, Liu A (2015) J Biol Chem 290:15621–15634
Zhang Y, Colabroy KL, Begley TP, Ealick SE (2005) Biochemistry 44:7632–7643
Bugg TDH, Lin G (2001) Chem Commun 11:941–952
Kovaleva EG, Lipscomb JD (2007) Science 316:453–457
Kovaleva EG, Lipscomb JD (2008) Biochemistry 47:11168–11170
Mbughuni MM, Chakrabarti M, Hayden JA, Bominaar EL, Hendrich MP, Münck E, Lipscomb JD (2010) Proc Natl Acad Sci USA 107:16788–16793
Groce SL, Lipscomb JD (2005) Biochemistry 44:7175–7188
Mbughuni MM, Chakrabarti M, Hayden JA, Meier KK, Dalluge JJ, Hendrich MP, Münck E, Lipscomb JD (2011) Biochemistry 50:10262–10274
Kovaleva EG, Lipscomb JD (2012) Biochemistry 51:8755–8763
Mbughuni MM, Meier KK, Münck E, Lipscomb JD (2012) Biochemistry 51:8743–8754
Hegg EL, Que L Jr (1997) Eur J Biochem 250:625–629
Vetting MW, Wackett LP, Que L Jr, Lipscomb JD, Ohlendorf DH (2004) J Bacteriol 186:1945–1958
Arciero DM, Lipscomb JD (1986) J Biol Chem 261:2170–2178
Jeoung JH, Bommer M, Lin TY, Dobbek H (2013) Proc Natl Acad Sci USA 110:12625–12630
Siegbahn PEM, Haeffner F (2004) J Am Chem Soc 126:8919–8932
Deeth RJ, Bugg TDH (2003) J Biol Inorg Chem 8:409–418
Christian GJ, Ye S, Neese F (2012) Chem Sci 3:1600–1611
Dong G, Shaik S, Lai W (2013) Chem Sci 4:3624–3635
Sanvoisin J, Langley GJ, Bugg TDH (1995) J Am Chem Soc 117:7836–7837
Xin M, Bugg TD (2008) J Am Chem Soc 130:10422–10430
Borowski T, Wojcik A, Milaczewska A, Georgiev V, Blomberg MRA, Siegbahn PEM (2012) J Biol Inorg Chem 17:881–890
Mendel S, Arndt A, Bugg TDH (2004) Biochemistry 43:13390–13396
Ohlendorf DH, Lipscomb JD, Weber PC (1988) Nature 336:403–405
Orville AM, Elango N, Lipscomb JD, Ohlendorf DH (1997) Biochemistry 36:10039–10051
Orville AM, Lipscomb JD, Ohlendorf DH (1997) Biochemistry 36:10052–10066
Davis MI, Wasinger EC, Decker A, Pau MYM, Vaillancourt FH, Bolin JT, Eltis LD, Hedman B, Hodgson KO, Solomon EI (2003) J Am Chem Soc 125:11214–11227
Knoot CJ, Purpero VM, Lipscomb JD (2015) Proc Natl Acad Sci USA 112:388–393
Pau MY, Lipscomb JD, Solomon EI (2007) Proc Natl Acad Sci USA 104:18355–18362
Que L Jr, Lipscomb JD, Münck E, Wood JM (1977) Biochim Biophys Acta 485:60–74
Schlosrich J, Eley KL, Crowley PJ, Bugg TDH (2006) ChemBioChem 7:1899–1908
Groce SL, Lipscomb JD (2003) J Am Chem Soc 125:11780–11781
Simmons CR, Krishnamoorthy K, Granett SL, Schuller DJ, Dominy JE Jr, Begley TP, Stipanuk MH, Karplus PA (2008) Biochemistry 47:11390–11392
Dominy JE Jr, Simmons CR, Hirschberger LL, Hwang J, Coloso RM, Stipanuk MH (2007) J Biol Chem 282:25189–25198
Richerson RB, Ziegler DM (1987) Methods Enzymol 143:410–415
Bruland N, Wubbeler JH, Steinbuchel A (2009) J Biol Chem 284:660–672
Tchesnokov EP, Fellner M, Siakkou E, Kleffmann T, Martin LW, Aloi S, Lamont IL, Wilbanks SM, Jameson GN (2015) J Biol Chem 290:24424–24437
Pierce BS, Subedi BP, Sardar S, Crowell JK (2015) Biochemistry 54:7477–7490
Driggers CM, Cooley RB, Sankaran B, Hirschberger LL, Stipanuk MH, Karplus PA (2013) J Mol Biol 425:3121–3136
Kumar D, Thiel W, de Visser SP (2011) J Am Chem Soc 133:3869–3882
Souness RJ, Kleffmann T, Tchesnokov EP, Wilbanks SM, Jameson GB, Jameson GN (2013) Biochemistry 52:7606–7617
Hohenberger J, Ray K, Meyer K (2012) Nat Commun 3:720
Bassan A, Blomberg MR, Siegbahn PE, Que L Jr (2005) Angew Chem Int Ed Engl 44:2939–2941
Costas M, Tipton AK, Chen K, Jo DH, Que L Jr (2001) J Am Chem Soc 123:6722–6723
Harder D, Hirschi S, Ucurum Z, Goers R, Meier W, Muller DJ, Fotiadis D (2016) Angew Chem Int Ed Engl 55:8846–8849
Yang A, Ha S, Ahn J, Kim R, Kim S, Lee Y, Kim J, Soll D, Lee HY, Park HS (2016) Science 354:623–626
Bose JL (2016) Methods Mol Biol 1373:111–115
Acknowledgements
The research in our laboratory is supported by the National Science Foundation Grants CHE-1623856 and MCB-0843537, the National Institutes of Health Grants GM107529, GM108988, and MH107985, and the Lutcher Brown Distinguished Chair Endowment fund. We thank Professors Lawrence Que, Jr. and John Lipscomb for helpful discussions of the catalytic mechanisms and editing of the text.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Li, J. & Liu, A. Oxygen activation by mononuclear nonheme iron dioxygenases involved in the degradation of aromatics. J Biol Inorg Chem 22, 395–405 (2017). https://doi.org/10.1007/s00775-017-1436-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-017-1436-5