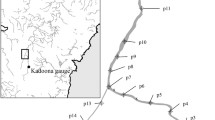
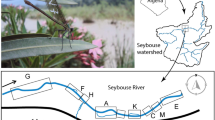
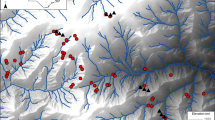
Abstract
Life-history information sets the foundation for our understanding of ecology and conservation requirements. For many species, this information is lacking even for basic demographic rates such as survival and movement. When survival and movement estimates are available, they are often derived from mixed demographic groups and do not consider differences among life stages or sexes, which is critical, because life stages and sexes often contribute differentially to population dynamics. We used hierarchical models informed with spatial capture–mark–recapture data of Ascaphus montanus (Rocky Mountain tailed frog) in five streams and A. truei (coastal tailed frog) in one stream to estimate variation in survival and movement by sex and age, represented by size. By incorporating survival and movement into a single model, we were able to estimate both parameters with limited bias. Annual survival was similar between sexes of A. montanus [females = 0.885 (95% CI 0.614–1), males = 0.901 (0.657–1)], but was slightly higher for female A. truei [0.836 (0.560–0.993)] than for males [0.664 (0.354–0.962)]. Survival of A. montanus peaked at mid-age, suggesting that lower survival of young and actuarial senescence may influence population demographics. Our models suggest that younger A. montanus moved farther than older individuals, and that females moved farther than males in both species. Our results provide uncommon insight into age- and sex-specific rates of survival and movement that are crucial elements of life-history strategies and are important for modeling population growth and prescribing conservation actions.




Similar content being viewed by others
References
Austin JD, Dávila JA, Lougheed SC, Boag PT (2003) Genetic evidence for female-biased dispersal in the bullfrog, Rana catesbeiana (Ranidae). Mol Ecol 12:3165–3172. https://doi.org/10.1046/j.1365-294X.2003.01948.x
Biek R, Funk WC, Maxell BA, Mills LS (2002) What is missing in amphibian decline research: insights from ecological sensitivity analysis. Conserv Biol 16:728–734
Bleu J, Herfindal I, Loison A et al (2015) Age-specific survival and annual variation in survival of female chamois differ between populations. Oecologia 179:1091–1098
Bradford DF (1983) Winterkill, oxygen relations, and energy metabolism of a submerged dormant amphibian, Rana muscosa. Ecology 64:1171–1183
Breden F (1987) The effect of post-metamorphic dispersal on the population genetic structure of Fowler’s toad, Bufo woodhousei fowleri. Copeia 1987:386–395. https://doi.org/10.2307/1445775
Breton AR, Nisbet ICT, Mostello CS, Hatch JJ (2014) Age-dependent breeding dispersal and adult survival within a metapopulation of Common Terns Sterna hirundo. Ibis 156:534–547. https://doi.org/10.1111/ibi.12161
Brown JH, Kodric-Brown A (1977) Turnover rates in insular biogeography: effect of immigration on extinction. Ecology 58:445. https://doi.org/10.2307/1935620
Burkholder LL, Diller LV (2007) Life history of postmetamorphic coastal tailed frogs (Ascaphus truei) in northwestern California. J Herpetol 41:251–262
Bury RB, Corn PS (1987) Evaluation of pitfall trapping in Northwestern forests: trap arrays with drift fences. J Wildl Manag 51:112–119. https://doi.org/10.2307/3801640
Campbell Grant EH, Green LE, Lowe WH (2009) Salamander occupancy in headwater stream networks. Freshw Biol 54:1370–1378. https://doi.org/10.1111/j.1365-2427.2009.02166.x
Caswell H (2001) Matrix population models. Wiley, Hoboken
Caughley G (1966) Mortality patterns in mammals. Ecology 47:906–918
Choquet R, Lebreton J-D, Gimenez O et al (2009) U-CARE: utilities for performing goodness of fit tests and manipulating capture–recapture data. Ecography 32:1071–1074
Clobert J, Baguette M, Benton TG, Bullock JM (2012) Dispersal ecology and evolution. Oxford University Press, Oxford
Colchero F, Clark JS (2012) Bayesian inference on age-specific survival for censored and truncated data. J Anim Ecol 81:139–149
Colchero F, Jones OR, Rebke M (2012) BaSTA: an R package for Bayesian estimation of age-specific survival from incomplete mark–recapture/recovery data with covariates. Methods Ecol Evol 3:466–470
Cole LC (1954) The population consequences of life history phenomena. Q Rev Biol 29:103–137
Corn PS, Bury RB (1991) Terrestrial amphibian communities in the Oregon Coast Range. US Department of Agriculture, Forest Service, Pacific Northwest Research Station
Cossel JO, Gaige MG, Sauder JD (2012) Electroshocking as a survey technique for stream-dwelling amphibians. Wildl Soc Bull 36:358–364. https://doi.org/10.1002/wsb.145
Crouse DT, Crowder LB, Caswell H (1987) A stage-based population model for loggerhead sea turtles and implications for conservation. Ecology 68:1412–1423. https://doi.org/10.2307/1939225
Daugherty CH (1979) Population ecology and genetics of Ascaphus truei: an examination of gene flow and natural selection. University of Montana, Montana
Daugherty CH, Sheldon AL (1982a) Age-determination, growth, and life history of a Montana population of the tailed frog (Ascaphus truei). Herpetologica 1982:461–468
Daugherty CH, Sheldon AL (1982b) Age-specific movement patterns of the frog Ascaphus truei. Herpetologica 1982:468–474
Dobson FS (1982) Competition for mates and predominant juvenile male dispersal in mammals. Anim Behav 30:1183–1192
Dodd CK (2013) Frogs of the United States and Canada, vol 2. JHU Press, Baltimore
Dole JW (1971) Dispersal of recently metamorphosed leopard frogs, Rana pipiens. Copeia 1971:221–228. https://doi.org/10.2307/1442821
Duellman WE, Trueb L (1986) Biology of amphibians. JHU press, Baltimore
Frederiksen M, Wanless S, Harris MP (2004) Estimating true age-dependence in survival when only adults can be observed: an example with Black-legged Kittiwakes. Anim Biodivers Conserv 27:541–548
Funk WC, Greene AE, Corn PS, Allendorf FW (2005) High dispersal in a frog species suggests that it is vulnerable to habitat fragmentation. Biol Lett 1:13–16. https://doi.org/10.1098/rsbl.2004.0270
Gaillard J-M, Festa-Bianchet M, Yoccoz NG et al (2000) Temporal variation in fitness components and population dynamics of large herbivores. Annu Rev Ecol Syst 31:367–393
Gaines SD, Bertness M (1993) The dynamics of juvenile dispersal: why field ecologists must integrate. Ecology 74:2430–2435. https://doi.org/10.2307/1939593
Gelman A, Rubin DB (1992) Inference from iterative simulation using multiple sequences. Stat Sci 7:457–472
Grant EHC, Nichols JD, Lowe WH, Fagan WF (2010) Use of multiple dispersal pathways facilitates amphibian persistence in stream networks. Proc Natl Acad Sci 107:6936–6940. https://doi.org/10.1073/pnas.1000266107
Greenwood PJ (1980) Mating systems, philopatry and dispersal in birds and mammals. Anim Behav 28:1140–1162
Gros A, Hovestadt T, Poethke HJ (2008) Evolution of sex-biased dispersal: The role of sex-specific dispersal costs, demographic stochasticity, and inbreeding. Ecol Model 219:226–233. https://doi.org/10.1016/j.ecolmodel.2008.08.014
Halliday TR, Verrell PA (1988) Body size and age in amphibians and reptiles. J Herpetol 22:253. https://doi.org/10.2307/1564148
Hamilton WD, May RM (1977) Dispersal in stable habitats. Nature 269:578–581
Hayes MP, Quinn T (2015) Review and synthesis of literature on tailed frogs (genus Ascaphus) with special reference to managed landscapes. Wash State Dep Nat Resour
Helfer V, Broquet T, Fumagalli L (2012) Sex-specific estimates of dispersal show female philopatry and male dispersal in a promiscuous amphibian, the alpine salamander (Salamandra atra). Mol Ecol 21:4706–4720. https://doi.org/10.1111/j.1365-294X.2012.05742.x
Honeycutt RK, Lowe WH, Hossack BR (2016) Movement and survival of an amphibian in relation to sediment and culvert design. J Wildl Manag 80:761–770
Hossack BR (2016) Amphibian dynamics in constructed ponds on a wildlife refuge: developing expected responses to hydrological restoration. Hydrobiologia. https://doi.org/10.1007/s10750-016-2979-0
Hutchings JA (1993) Adaptive life histories effected by age-specific survival and growth rate. Ecology 74:673–684. https://doi.org/10.2307/1940795
Jones OR, Gaillard J-M, Tuljapurkar S et al (2008) Senescence rates are determined by ranking on the fast–slow life-history continuum. Ecol Lett 11:664–673. https://doi.org/10.1111/j.1461-0248.2008.01187.x
Jones OR, Scheuerlein A, Salguero-Gómez R et al (2014) Diversity of ageing across the tree of life. Nature 505:169–173. https://doi.org/10.1038/nature12789
Kéry M, Schaub M (2012) Bayesian population analysis using WinBUGS: a hierarchical perspective. Academic Press, London
Kiffney PM, Richardson JS, Montgomery WL (2001) Interactions among nutrients, periphyton, and invertebrate and vertebrate (Ascaphus truei) grazers in experimental channels. Copeia 2001:422–429
Lawson Handley LJ, Perrin N (2007) Advances in our understanding of mammalian sex-biased dispersal. Mol Ecol 16:1559–1578. https://doi.org/10.1111/j.1365-294X.2006.03152.x
Lebreton J-D, Burnham KP, Clobert J, Anderson DR (1992) Modeling survival and testing biological hypotheses using marked animals: a unified approach with case studies. Ecol Monogr 62:67–118
Levitis DA (2011) Before senescence: the evolutionary demography of ontogenesis. Proc R Soc Lond B Biol Sci 278:801–809. https://doi.org/10.1098/rspb.2010.2190
Liebgold EB, Brodie ED, Cabe PR (2011) Female philopatry and male-biased dispersal in a direct-developing salamander, Plethodon cinereus. Mol Ecol 20:249–257. https://doi.org/10.1111/j.1365-294X.2010.04946.x
Lomnicki A (1988) Population ecology of individuals. Princeton University Press, Princeton
Matsuda BM, Richardson JS (2005) Movement patterns and relative abundance of coastal tailed frogs in clearcuts and mature forest stands. Can J For Res 35:1131–1138
McCaffery RM, Maxell BA (2010) Decreased winter severity increases viability of a montane frog population. Proc Natl Acad Sci 107:8644–8649. https://doi.org/10.1073/pnas.0912945107
Medawar PB (1952) An unsolved problem of biology. H. K, Lewis
Metter DE (1964) A morphological and ecological comparison of two populations of the tailed frog, Ascaphus truei Stejneger. Copeia 1964:181–195
Miller DA, Janzen FJ, Fellers GM et al (2014) Biodemography of ectothermic tetrapods provides insights into the evolution and plasticity of mortality patterns. Sociality Hierarchy Health Comp Biodemography Natl Acad Press Wash
Mills LS, Allendorf FW (1996) The one-migrant-per-generation rule in conservation and management. Conserv Biol 10:1509–1518
Muths E, Scherer RD, Lambert BA (2010) Unbiased survival estimates and evidence for skipped breeding opportunities in females. Methods Ecol Evol 1:123–130. https://doi.org/10.1111/j.2041-210X.2010.00019.x
Nussey DH, Froy H, Lemaitre J-F et al (2013) Senescence in natural populations of animals: widespread evidence and its implications for bio-gerontology. Ageing Res Rev 12:214–225. https://doi.org/10.1016/j.arr.2012.07.004
Palo JU, Lesbarrères D, Schmeller DS et al (2004) Microsatellite marker data suggest sex-biased dispersal in the common frog Rana temporaria. Mol Ecol 13:2865–2869. https://doi.org/10.1111/j.1365-294X.2004.02286.x
Pearson PG (1955) Population ecology of the spadefoot toad, Scaphiopus h. holbrooki (Harlan). Ecol Monogr 25:233–267
Perrin N, Mazalov V (2000) Local competition, inbreeding, and the evolution of sex-biased dispersal. Am Nat 155:116–127
Petranka JW (1985) Does age-specific mortality decrease with age in amphibian larvae? Copeia 1985:1080–1083
Pittman SE, Osbourn MS, Semlitsch RD (2014) Movement ecology of amphibians: a missing component for understanding population declines. Biol Conserv 169:44–53
Pletcher SD (1999) Model fitting and hypothesis testing for age-specific mortality data. J Evol Biol 12:430–439
Plummer M (2003) JAGS: a program for analysis of Bayesian graphical models using Gibbs sampling. In: Hornik K, Leisch F, Zeileis A (eds) Proceedings of the 3rd international workshop on distributed statistical computing (DSC 2003), Vienna, Austria, pp 1–10
Pollock KH (1982) A capture–recapture design robust to unequal probability of capture. J Wildl Manag 46:752–757. https://doi.org/10.2307/3808568
Pradel R, Hines JE, Lebreton J-D, Nichols JD (1997) Capture–recapture survival models taking account of transients. Biometrics 53:60–72. https://doi.org/10.2307/2533097
Pusey AE (1987) Sex-biased dispersal and inbreeding avoidance in birds and mammals. Trends Ecol Evol 2:295–299. https://doi.org/10.1016/0169-5347(87)90081-4
Saether B-E, Coulson T, Grøtan V et al (2013) How life history influences population dynamics in fluctuating environments. Am Nat 182:743–759
Sala OE, Chapin FS, Armesto JJ et al (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774. https://doi.org/10.1126/science.287.5459.1770
Schaub M, Royle JA (2014) Estimating true instead of apparent survival using spatial Cormack–Jolly–Seber models. Methods Ecol Evol 5:1316–1326. https://doi.org/10.1111/2041-210X.12134
Schmidt BR, Schaub M, Steinfartz S (2007) Apparent survival of the salamander Salamandra salamandra is low because of high migratory activity. Front Zool 4:1
Schmidt BR, Hödl W, Schaub M (2012) From metamorphosis to maturity in complex life cycles: equal performance of different juvenile life history pathways. Ecology 93:657–667
Smith MA, Green DM (2006) Sex, isolation and fidelity: unbiased long-distance dispersal in a terrestrial amphibian. Ecography 29:649–658. https://doi.org/10.1111/j.2006.0906-7590.04584.x
Su Y-S, Yajim M (2014) R2jags: a package for running jags from R. R package version 0.04-03
Tavecchia G, Pradel R, Lebreton J-D et al (2002) Sex-biased survival and breeding dispersal probability in a patchy population of the Rock Sparrow Petronia petronia. Ibis 144:E79–E87. https://doi.org/10.1046/j.1474-919X.2002.00059.x
Toïgo C, Gaillard J-M (2003) Causes of sex-biased adult survival in ungulates: sexual size dimorphism, mating tactic or environment harshness? Oikos 101:376–384
Toïgo C, Gaillard J-M, Festa-Bianchet M et al (2007) Sex- and age-specific survival of the highly dimorphic Alpine ibex: evidence for a conservative life-history tactic. J Anim Ecol 76:679–686. https://doi.org/10.1111/j.1365-2656.2007.01254.x
Vignieri SN (2007) Cryptic behaviours, inverse genetic landscapes, and spatial avoidance of inbreeding in the Pacific jumping mouse. Mol Ecol 16:853–866. https://doi.org/10.1111/j.1365-294X.2006.03182.x
Vitt LJ, Caldwell JP (2013) Herpetology: an introductory biology of amphibians and reptiles. Academic Press, London
Wahbe TR, Bunnell FL, Bury RB (2004) Terrestrial movements of juvenile and adult tailed frogs in relation to timber harvest in coastal British Columbia. Can J For Res 34:2455–2466
Wells KD (2010) The ecology and behavior of amphibians. University of Chicago Press, Chicago
Williams GC (1957) Pleiotropy, natural selection, and the evolution of senescence. Evolution 1957:398–411
Williams BK, Nichols JD, Conroy MJ (2002) Analysis and management of animal populations. Academic Press, London
Wright S (1949) The genetical structure of populations. Ann Hum Genet 15:323–354. https://doi.org/10.1111/j.1469-1809.1949.tb02451.x
Zimmerman GS, Gutiérrez RJ, Lahaye WS (2007) Finite study areas and vital rates: sampling effects on estimates of spotted owl survival and population trends. J Appl Ecol 44:963–971. https://doi.org/10.1111/j.1365-2664.2007.01343.x
Acknowledgements
This manuscript is dedicated to Joshua W. Wilkerson who passed away shortly after the completion of the field portion of this project, to which Mr. Wilkerson made vital contributions. Field technicians R. M. Bourque, M. P. Capozzoli, M. D. Larson, N. E. Proffitt, K. J. Van Atta, and C. A. Wheeler also made valuable contributions to this project. L. A. Eby and P. M. Lukacs provided insight and review on project development. M. Schaub provided assistance with coding Bayesian models. K. A. Smith and many other USFS personnel provided logistical assistance. Comments by T. Chambert, members of W. Lowe’s laboratory, and two anonymous reviewers improved this manuscript. Any use of trade, product, or firm names is for descriptive purposes only and does not imply endorsement by the U.S. Government. This manuscript is contribution number 695 of the U.S. Geological Survey Amphibian Research and Monitoring Initiative (ARMI).
Funding
Funding for this project was provided by the USFS Aquatic Organism Passage program, USFS Pacific Southwest Research Station, and California Department of Fish and Wildlife.
Author information
Authors and Affiliations
Contributions
JMG, RKH, and WHL conceived and designed the field experiments. JMG and RKH performed the field experiments. RKH analyzed the data. BRH and RKH led authorship of the manuscript; JMG and WHL provided extensive editorial input through many revisions of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable institutional and national guidelines for the care and use of animals were followed.
Additional information
Communicated by Howard Whiteman.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Honeycutt, R.K., Garwood, J.M., Lowe, W.H. et al. Spatial capture–recapture reveals age- and sex-specific survival and movement in stream amphibians. Oecologia 190, 821–833 (2019). https://doi.org/10.1007/s00442-019-04464-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-019-04464-3