Abstract
The properties of Pluronic F127 aqueous solutions with the variation of concentration and temperature were studied by cyclic voltammetry at the presence of sodium bromide. By this method, the relationship between the peak currents of electroactive probe, 2, 2, 6, 6-tetramethyl-1-piperidinyloxy (TEMPO), and the viscosity of F127 aqueous solutions within some temperature ranges was discussed, and the diffusion coefficient (D) of TEMPO in F127 aqueous solutions at various concentrations under different temperatures was determined. The critical micellar temperature (CMT) of F127 aqueous solution at various concentrations can be obtained from the curve of the peak current i p against the square root of reciprocal viscosity (1/η)1/2 and from the curve of logarithm diffusion coefficient (logD) of TEMPO as a function of reciprocal temperature (1/T). All the results are in good agreement with the results from 1H NMR measurements. Besides, the thermodynamic parameters such as diffusion activation energy (E D) and hydrodynamic radius (R H) can be also obtained from the above two curves. A new method was introduced to determine the CMT of copolymer systems, which is simple, quick, and accurate.

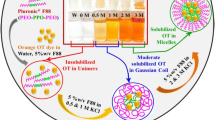
For Pluronic F127 in aqueous solutions containing NaBr, the critical micellar temperature (CMT) can be determined by the plot of anodic peak currents of electroactive probe TEMPO against the square root of reciprocal viscosities.









Similar content being viewed by others
References
Malmsten’ M, Lindman B (1992) Macromolecules 25:5440–5445
Miyazaki S, Tobiyama T, Takada M, Attwood D (1995) J Pharm Pharmacol 47:455–457
Ho HO, Chen CN, Sheu MT (2000) J Control Release 68:433–440
Oh KS, Lee KE, Han SS, Cho SH, Kim D, Yuk SH (2005) Biomacromolecules 6:1062–1067
Wang YQ, Yang CM, Zibrowius B, Spliethoff B, Linden M, Schuth F (2003) Chem Mater 15:5029–5035
Flodstrom K, Wennerstrom H, Alfredsson V (2004) Langmuir 20:680–688
Sun DJ, Zhang CG, Chen YJ, Guo BY, Liu SY, Sun MB (2004) Drill Fluid Complet Fluid 21:8–10
Mortensen K, Pedersen JS (1993) Macromolecules 26:805–812
Yang L, Alexandridis P, Steytler DC, Kositza MJ, Holzwarth JF (2000) Langmuir 16:8555–8561
Patel T, Bahadur P, Mata J (2010) J Colloid Interface Sci 345:346–350
Jia LW, Guo C, Yang LR, Xiang JF, Tang YL, Liu CZ et al (2010) J Colloid Interface Sci 345:332–337
Su YL, Wang J, Liu HZ (2002) Macronolecules 35:6426–6431
Ganguly R, Aswal VK, Hassana PA (2007) J Colloid Interface Sci 315:693–700
Sardar N, Ali MS, Kamil M, Kabir-ud-Din (2010) J Chem Eng Data 55:4990–4994
Bharatiya B, Guo C, Ma JH, Kubota O, Nakashima K, Bahadur P (2009) Colloid Polym Sci 287:63–71
Mata JP, Majhi PR, Guo C, Liu HZ, Bahadur P (2005) J Colloid Interface Sci 292:548–556
Naskar B, Ghosh S, Moulik SP (2012) Langmuir 28:7134–7146
Li Y, Xu R, Couderc S, Bloor M, Wyn-Jones E, Holzwarth JF (2001) Langmuir 17:183–188
O’Lenick TG, Jiang XM, Zhao B (2009) Polymer 50:4363–4371
Bakshi MS, Kaur N, Mahajan RK (2007) J Photochem Photobiol A 186:349–356
Bakshi MS, Bhandari P (2007) J Photochem Photobiol A 186:166–172
Grant CD, DeRitter MR, Steege KE, Fadeeva TA, Castner JEW (2005) Langmuir 21:1745–1752
Grant CD, Steege KE, Bunagan MR, Castner JEW (2005) J Phys Chem B 109:22273–22284
Sony G, Manoj K, Prabhat Kr. S, Rajib G, Sukhendu N et al (2009) J Phys Chem B 113:5117–5127
Mali KS, Dutt GB, Mukherjee T (2007) Langmuir 23:1041–1046
Patel K, Bahadur P, Guo C, Ma JH, Liu HZ, Yamashita Y et al (2007) Eur Polym J 43:1699–1708
Kadama Y, Singh K, Marangoni DG, Ma JH, Aswal VK, Bahadur P (2010) Colloids Surf A: Physicochem Eng Asp 369:121–127
Ma JH, Guo C, Tang YL, Xiang JF, Chen S, Wang J et al (2007) J Colloid Interface Sci 312:390–396
Ma JH, Guo C, Tang YL, Wang JL, Zheng L, Liang XF et al (2006) J Colloid Interface Sci 299:953–961
Desai PR, Jain NJ, Sharma RK, Bahadur P (2001) Colloids Surf A Physicochem Eng Asp 178:57–69
álvarez-Ramírez JG, Fernández VVA, Macías ER, Rharbi Y, Taboada P et al (2009) J Colloid Interface Sci 333:655–662
Wen SY, Knoll D, Knoll H (2005) J Colloid Interface Sci 291:244–250
Li GB, Li HG, Hao JC (2013) J Colloid Polym Sci 291:1479–1486
Nandni DK, Vohra K, Mahajan RK (2009) J Colloid Interface Sci 338:420–427
Mahajan RK, Vohra KK, Shaheen A, Aswal VK (2008) J Colloid Interface Sci 326:89–95
Mahajan RK, Kaur N, Bakshi MS (2005) Colloids Surf A 255:33–39
Ding YH, Wang Y, Guo R (2004) J Surfactant Deterg 7:379–385
Cai C, Gao ZN (2004) Chem Res Appl 16:643–645
Zhang XL, Wang AJ, Zhang XS, Xu P (2007) Chin J Appl Chem 24:374–377
Kissinger PT, Heineman WR (1983) J Chem Educ 60:702–706
Stackellberg MV, Pilgram M, Toome V (1953) Z Elektrochem 57:342–350
Baur JE, Wang S, Brandt MC (1996) Anal Chem 68:3815–3821
Wu DG, Malec AD, Head-Gordon M, Majda M (2005) J Am Chem Soc 27:4490–4996
Krzyczmonik PHS (1992) J Electroanal Chem 335:233–251
Bird RB, Stewart WE, Lightfoot EN (1960) Transport phenomena. Wiley, New York
Waldeck AR, Kuchel PW, Lennon AJ, Chapman BE (1997) Prog Nucl Magn Reson Spectrosc 30:39–68
Dean JA (1999) Langep’s handbook of chemistry [M]. 15th ed. Mc Graw-Hill, New York
Yang JZ, Jin Y, Pan W (2004) Acta Chim Sin 2035–2039:62
Alexandridis P, Holzwarth JFT, Hatton A (1994) Macromolecules 27:2414–2425
Acknowledgments
This work was financially supported by the International Science Cooperation Project of Shandong Province, China (2012GHZ20205), the Hundred Talents Program of Chinese Academy of Sciences (Y20245YBR1), and the Doctor Foundation of University of Jinan (No. XBS1204).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 2097 kb)
Rights and permissions
About this article
Cite this article
Li, G., Hao, J., Li, H. et al. Determination of the critical micellar temperature of F127 aqueous solutions at the presence of sodium bromide by cyclic voltammetry. Colloid Polym Sci 293, 787–796 (2015). https://doi.org/10.1007/s00396-014-3461-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-014-3461-z