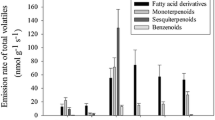
Abstract
Key message
Essential oils have growth regulating properties comparable to the well-documented methyl jasmonate and may be involved in localized and/or airborne plant communication.
Abstract
Aromatic plants employ large amounts of resources to produce essential oils. Some essential oils are known to contain compounds with plant growth regulating activities. However, the potential capacity of essential oils as airborne molecules able to modulate plant growth/development has remained uninvestigated. Here, we demonstrate that essential oils from eight taxonomically diverse plants applied in their airborne state inhibited auxin-induced elongation of Pisum sativum hypocotyls and Avena sativa coleoptiles. This response was also observed using five monoterpenes commonly found in essential oils as well as isoprene, the basic building block of terpenes. Upon transfer to ambient conditions, A. sativa coleoptiles resumed elongation, demonstrating an antagonistic relationship rather than toxicity. Inclusion of essential oils, monoterpenes, or isoprene into the headspace of culture vessels induced abnormal cellular growth along hypocotyls of Arabidopsis thaliana. These responses were also elicited by methyl jasmonate (MeJA); however, where methyl jasmonate inhibited root growth essential oils did not. Gene expression studies in A. thaliana also demonstrated differences between the MeJA and isoprenoid responses. This series of experiments clearly demonstrate that essential oils and their isoprenoid components interact with endogenous plant growth regulators when applied directly or as volatile components in the headspace. The similarities between isoprenoid and MeJA responses suggest that they may act in plant defence signalling. While further studies are needed to determine the ecological and evolutionary significance, the results of this study and the specialized anatomy associated with aromatic plants suggest that essential oils may act as airborne signalling molecules.









Similar content being viewed by others
References
Abdelgadir HA, Johnson SD, Van Staden J (2009) Promoting branching of a potential biofuel crop Jatropha curcas L. by foliar application of plant growth regulators. Plant Growth Regul 58:287–295
Baldwin IT, Schultz JC (1983) Rapid changes in tree leaf chemistry induced by damage: evidence for communication between plants. Science 221:277–279
Başer KHC, Demirci B (2007) Studies on Betula essential oils. Arkivoc 7:335–348
Burström H (1950) Studies on growth and metabolism of roots. IV. Positive and negative auxin effects on cell elongation. Physiol Plant 3:277–292
Chen Q, Sun J, Zhai Q, Zhou W, Qi L, Xu L, Wang B, Chen R, Jiang H, Gi J et al (2011) The basic helix–loop–helix transcription factor MYC2 directly represses PLETHORA expression during jasmonate-mediated modulation of the root stem cell niche in Arabidopsis. Plant Cell 23:3335–3352
Consales F, Schweizer F, Erb M et al (2012) Insect oral secretions suppress wound-induced responses in Arabidopsis. J Exp Bot 63:727–737
Demole E, Lederer E, Mercier D (1962) Isolement et détermination de la structure du jasmonate de méthyle, constituant odorant caractéristique de l’essence de jasmin. Helv Chim Acta 45:675–685
Dun EA, Ferguson BJ, Beveridge CA (2006) Apical dominance and shoot branching. Divergent opinions or divergent mechanisms? Plant Physiol 142:812–819
Edey JM, Byth DE (1970) The influence of 2, 3, 5-triiodobenzoic acid (TIBA) on vegetative and reproductive growth of legumes. Anim Prod Sci 10:732–738
Farmer EE (2001) Surface-to-air signals. Nature 411:854–856
Farmer EE, Ryan CA (1990) Interplant communication: airborne methyl jasmonate induces synthesis of proteinase inhibitors in plant leaves. Proc Natl Acad Sci 87:7713–7716
Godard K-A, White R, Bohlmann J (2008) Monoterpene-induced molecular responses in Arabidopsis thaliana. Phytochemistry 69:1838–1849
Heil M, Karban R (2010) Explaining evolution of plant communication by airborne signals. Trends Ecol Evol 25:137–144
Heil M, Silva Bueno JC (2007) From the Cover: within-plant signaling by volatiles leads to induction and priming of an indirect plant defense in nature. Sci Signal 104:5467
Ishizaki S, Shiojiri K, Karban R, Ohara M (2011) Effect of genetic relatedness on volatile communication of sagebrush (Artemisia tridentata). J Plant Interactions 6:193
Jones AMP, Klun JA, Cantrell CL et al (2012) Isolation and identification of mosquito (Aedes aegypti) biting deterrent fatty acids from male inflorescences of breadfruit (Artocarpus altilis (Parkinson) Fosberg). J Agric Food Chem 60:3867–3873
Karban R (2008) Plant behaviour and communication. Ecol Lett 11:727–739. doi:10.1111/j.1461-0248.2008.01183.x
Karban R, Baldwin IT, Baxter KJ et al (2000) Communication between plants: induced resistance in wild tobacco plants following clipping of neighboring sagebrush. Oecologia 125:66–71
Karban R, Shiojiri K, Ishizaki S, Wetzel WC, Evans RY (2013) Kin recognition affects plant communication and defence. Proc R Soc B Biol, Sci 280
Kim S-H, Arnold D, Lloyd A, Roux SJ (2001) Antisense expression of an Arabidopsis ran binding protein renders transgenic roots hypersensitive to auxin and alters auxin-induced root growth and development by arresting mitotic progress. Plant Cell Online 13:2619–2630
Lindigkeit R, Biller A, Buch M, Schiebel HM, Boppre M, Hartmann T (1997) The two faces of pyrrolizidine alkaloids: the role of the tertiary amine and its N-oxide in chemical defense of insects with acquired plant alkaloids. Eur J Biochem 245:626–636
Little DB, Croteau RB (1999) Biochemistry of essential oil terpenes. Flavor chemistry. Springer, New York, pp 239–253
Machado RA, Ferrieri AP, Robert CA, Glauser G, Kallenbach M, Baldwin IT, Erb M (2013) Leaf-herbivore attack reduces carbon reserves and regrowth from the roots via jasmonate and auxin signaling. New Phytol 200:1234–1246
MacRae E (2007) Extraction of plant RNA. Protocols for nucleic acid analysis by nonradioactive probes. Springer, New York, pp 15–24
Mao W, Rupasinghe S, Zangerl AR, Schuler MA, Berenbaum MR (2006) Remarkable substrate-specificity of CYP6AB3 in Depressaria pastinacella a highly specialized caterpillar. Insect Mol Biol 15:169–179
McNair JB (1932) The interrelation between substances in plants: essential oils and resins, cyanogen and oxalate. Am J Bot 19:255–272
Miller CO (1954) The influence of cobalt and sugars upon the elongation of etiolated pea stem segments. Plant Physiol 29:79
Moloney MM, Elliott MC, Cleland RE (1981) Acid growth effects in maize roots: evidence for a link between auxin-economy and proton extrusion in the control of root growth. Planta 152:285–291
Murch SJ, Rupasinghe HP, Saxena PK (2002) An in vitro and hydroponic growing system for hypericin, pseudohypericin, and hyperforin production of St. John’s wort (Hypericum perforatum CV new stem). Planta Med 68:1108
Navarro L, Dunoyer P, Jay F et al (2006) A plant miRNA contributes to antibacterial resistance by repressing auxin signaling. Sci Signal 312:436
Noodén LD, Thimann KV (1963) Evidence for a requirement for protein synthesis for auxin-induced cell enlargement. Proc Natl Acad Sci USA 50:194
Pichersky E, Gershenzon J (2002) The formation and function of plant volatiles: perfumes for pollinator attraction and defense. Curr Opin Plant Biol 5:237–243
Preston CA, Laue G, Baldwin IT (2004) Plant–plant signaling: application of trans-or cis-methyl jasmonate equivalent to sagebrush releases does not elicit direct defenses in native tobacco. J Chem Ecol 30:2193–2214
Salinas J, Sánchez-Serrano JJ (2006) Arabidopsis protocols. Springer, New York
Schwachtje J, Baldwin IT (2008) Why does herbivore attack reconfigure primary metabolism? Plant Physiol 146:845–851
Scott TK (1972) Auxins and roots. Annu Rev Plant Physiol 23:235–258
Sharkey TD, Wiberley AE, Donohue AR (2008) Isoprene emission from plants: why and how. Ann Bot 101:5–18
Shukla MR, Jones AMP, Sullivan JA et al (2012) In vitro conservation of American elm (Ulmus americana): potential role of auxin metabolism in sustained plant proliferation. Can J For Res 42:686–697
Shulaev V, Silverman P, Raskin I (1997) Airborne signaling by methyl salicylate in plant pathogen resistance. Nature 385:718–721. doi:10.1038/385718a0
Sirois JC (1966) Studies on growth regulators. I. Improved Avena coleoptile elongation test for auxin. Plant Physiol 41:1308–1312
Strauss SY, Agrawal AA (1999) The ecology and evolution of plant tolerance to herbivory. Trends Ecol Evol 14:179–185
Svoboda KP, Svoboda TG, Syred AD, Syred PM (2000) Secretory structures of aromatic and medicinal plants: a review and atlas of micrographs. Microscopix Publications, London
Tanimoto E (2005) Regulation of root growth by plant hormones—roles for auxin and gibberellin. Crit Rev Plant Sci 24:249–265
Tanimoto E, Watanabe J (1986) Automated recording of lettuce root elongation as affected by auxin and acid pH in a new rhizometer with minimum mechanical contact to roots. Plant Cell Physiol 27:1475–1487
Terry GM, Stokes NJ, Hewitt CN, Mansfield TA (1995) Exposure to isoprene promotes flowering in plants. J Exp Bot 46:1629–1631
Tiffin P (2000) Mechanisms of tolerance to herbivory damage: what do we know? Evol Ecol 14:523–536
Tiryaki I, Staswick PE (2002) An Arabidopsis mutant defective in jasmonate response is allelic to the auxin-signaling mutant axr1. Plant Physiol 130:887–894
Ueda J, Miyamoto K, Aoki M (1994) Jasmonic acid inhibits the IAA-induced elongation of oat coleoptile segments: a possible mechanism involving the metabolism of cell wall polysaccharides. Plant Cell Physiol 35:1065–1070
Ulmasov T, Murfett J, Hagen G, Guilfoyle TJ (1997) Aux/IAA proteins repress expression of reporter genes containing natural and highly active synthetic auxin response elements. Plant Cell 9:1963–1971
Unlu M, Ergene E, Unlu GV et al (2010) Composition, antimicrobial activity and in vitro cytotoxicity of essential oil from Cinnamomum zeylanicum Blume Lauraceae. Food Chem Toxicol 48:3274–3280
Van Overbeek J, Blondeau R, Horne V (1951) Trans-cinnamic acid as an anti-auxin. Am J Bot 38:589–595
Wang L, Wu J (2013) The essential role of jasmonic acid in plant-herbivore interactions–using the wild tobacco Nicotiana attenuata as a model. J Genet Genomics 40:597–606
Welch BL, McArthur ED (1981) Variation of monoterpenoid content among subspecies and accessions of Artemisia tridentata grown in a uniform garden. J Range Manag 34:380–384
Werker E (1993) Function of essential oil-secreting glandular hairs in aromatic plans of Lamiacea—a review. Flavour Fragr J 8:249–255
Yang DJ, Yao J, Mei C-S, Tong X-H, Zeng L-J, Li Q, Xiao L-T, Sun T, Li J, Deng X-W et al (2012) Plant hormone jasmonate prioritizes defense over growth by interfering with gibberellin signaling cascade. Proc Nat Acad Sci USA 109:E1192–E1200
Acknowledgments
The authors thank the Gosling foundation and the National Science and Engineering Research Council for supporting this work. Additionally, the authors acknowledge the contributions of Dr. Jaideep Mathur, Abhishek Chattopadhyay, and Shuping Li for their assistance in various aspects of the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests.
Additional information
Communicated by A. Dhingra.
A. M. P. Jones and M. R. Shukla contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jones, A.M.P., Shukla, M.R., Sherif, S.M. et al. Growth regulating properties of isoprene and isoprenoid-based essential oils. Plant Cell Rep 35, 91–102 (2016). https://doi.org/10.1007/s00299-015-1870-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-015-1870-1