Summary
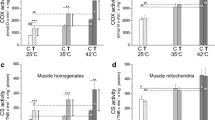
Our purpose was to test the significance of exhaustive training in aerobic or endurance capacity. The extent of adaptations to endurance training was evaluated by assessing the increase in physical performance capability and oxidative markers in the organs of rats trained by various exercise programs. Rats were trained by treadmill running 5 days · week−1 at 30 m · min−1 for 8 weeks by one of three protocols:T 1 — 60 min · day−1;T 2 — 120 min · day−1; andT 3 — 120 min · day−1 (3 days · week−1) and to exhaustion (2 days · week−1). GroupsT 2 andT 3 ran for longer thanT 1 in an endurance exercise test (P<0.05), in which the animals ran at 30 m · min−1 to exhaustion; no difference was observed between groupsT 2 andT 3. All 3 trained groups showed a similar increase (20–27%) in the fast-twitch oxidative-glycolytic (FOG) fibers with a concomitant decrease in the fast-twitch glycolytic (FG) fiber population in gastrocnemius (p<0.05). The capillary supply in gastrocnemius increased with the duration of exercise (p<0.05): no difference was found between groupsT 2 andT 3. Likewise, no distinction was seen between groupsT 2 andT 3 in the increase in succinate dehydrogenase activity in gastrocnemius and the heart. These results suggest that the maximal adaptive response to endurance training does not require daily exhaustive exercise.
Similar content being viewed by others
References
Adolfsson J, Ljungqvist A, Tornling G, Unge G (1981) Capillary increase in the skeletal muscle of trained young and adult rats. J Physiol 310:529–532
Andersen P (1975) Capillary density in skeletal muscle of man. Acta Physiol Scand 95:203–205
Chi MMY, Hintz CS, Coyle EF, Martin WH III, Ivy JL, Nemeth PM, Holloszy JO, Lowry OH (1983) Effects of detraining on enzymes of energy metabolism in individual human muscle fibers. Am J Physiol 244 [Cell Physiol 13]:C276-C287
Costill DL, Fink WJ, Pollock ML (1976) Muscle fiber composition and enzyme activities of elite distance runners. Med Sci Sports 8:96–100
Davies KJA, Packer L, Brooks GA (1981) Biochemical adaptations of mitochondria, muscle and whole-animal respiration to endurance training. Arch Biochem Biophys 209:539–554
Dudley GA, Abraham WM, Terjung RL (1982) Influence of exercise intensity and duration on biochemical adaptations in skeletal muscle. J Appl Physiol 53:844–850
Edgerton VR, Gerchman L, Carrow R (1969) Histochemical changes in rat skeletal muscle after exercise. Exp Neurol 24:110–123
Fitts RH, Booth FW, Winder WW, Holloszy JO (1975) Skeletal muscle respiratory capacity, endurance, and glycogen utilization. Am J Physiol 228:1029–1033
Gollnick PD, Armstrong RB, Saubert CW IV, Piehl K, Saltin B (1972) Enzyme activity and fiber composition in skeletal muscle of untrained and trained men. J Appl Physiol 33:312–319
Green HJ, Klug GA, Reichman H, Seedorf U, Wiehrer W, Pette D (1984) Exercise-induced fibre type transitions with regard to myosin, parvalbumin, and sarcoplasmic reticulum in muscles of the rat. Pflügers Arch 400:432–438
Green HJ, Reichmann H, Pette D (1983) Fibre type specific transformations in the enzyme activity pattern of rat vastus lateralis muscle by prolonged endurance training. Pflügers Arch 399:216–222
Holloszy JO, Coyle EF (1984) Adaptations of skeletal muscle to endurance exercise and their metabolic consequences. J Appl Physiol 56:831–838
Lo S, Russell JC, Taylor AW (1970) Determination of glycogen in small tissue samples. J Appl Physiol 28:234–236
Luginbuhl AJ, Dudley GA, Staron RS (1984) Fiber type changes in rat skeletal muscle after intense interval training. Histochemistry 81:55–58
Mandroukas K, Krotkiewski M, Hedberg M, Wroblewski Z, Björntop P, Grimby G (1984) Physical training in obese women: effects of muscle morphology, biochemistry and function. Fur J Appl Physiol 52:355–361
Maxwell LC, White TP, Faulkner JA (1980) Oxidative capacity, blood flow, and capillarity of skeletal muscles. J Appl Physiol 49:627–633
Novikoff AB, Shin W, Drucker J (1961) Mitochondrial localization of oxidative enzyme. Staining results with two tetrazolium salts. J Biophys Cytol 9:47–61
Padykula HA, Herman E (1955) Factors affecting the activity of adenosine triphosphatase and other phosphatases as measured by histochemical techniques. J Histochem 3:161–169
Peter JB, Barnard RJ, Edgerton YR, Gillespie CA, Stempel KE (1972) Metabolic profiles of three fiber types of skeletal muscle in guinea pigs and rabbits. Biochemistry 11:2627–2633
Pette D (1984) Activity-induced fast to slow transitions in mammalian muscle. Med Sci Sports Exercise 16:517–528
Schantz P, Billeter R, Henriksson J, Jansson E (1982) Training-induced increase in myofibrillar ATPase intermediate fibers in human skeletal muscle. Muscle Nerve 5:628–636
Schantz P, Henriksson J, Jansson E (1983) Adaptations of human skeletal muscle to endurance training of long duration. Clin Physiol 3:141–151
Sembrowich WL, Knudson MB, Gollnick PD (1977) Muscle metabolism and cardiac function of the myopathic hamster following training. J Appl Physiol 43:936–941
Tamaki N (1987) Effect of endurance training on muscle fiber type composition and capillary supply in rat diaphragm. Eur J Appl Physiol 56:127–131
Wada M, Katsuta S (1988) Changes in myoproteins following long-term with high intensity endurance training in rats. Jpn J Physical Educ 32:221–229
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Katsuta, S., Kanao, Y. & Aoyagi, Y. Is exhaustive training adequate preparation for endurance performance?. Europ. J. Appl. Physiol. 58, 68–73 (1988). https://doi.org/10.1007/BF00636605
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00636605