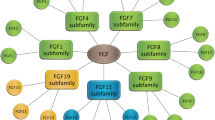
Abstract
In the fibroblast growth factor (FGF) system, sulfated glycosaminoglycans (GAGs) play very important roles. They interact with most FGF ligands and their receptors (FGFRs) and enable formation of active and stable signaling complexes composed of FGF-FGFR or FGF-FGFR/coreceptor. The fine structures of GAGs, particularly position-specific O-sulfation patterns, determine the GAGs’ regulatory activity toward specific FGFs and FGFRs.
Similar content being viewed by others
References
Asada M, Shinomiya M, Suzuki M, Honda E, Sugimoto R, Ikekita M, Imamura T (2009) Glycosaminoglycan affinity of the complete fibroblast growth factor family. Biochim Biophys Acta 1790(1):40–48
Imamura T, Mitsui Y (1987) Heparan sulfate and heparin as a potentiator or a suppressor of growth of normal and transformed vascular endothelial cells. Exp Cell Res 172(1):92–100
Kan M, Wang F, Xu J, Crabb JW, Hou J, McKeehan WL (1993) An essential heparin-binding domain in the fibroblast growth factor receptor kinase. Science 259(5103):1918–1921
Maciag T, Mehlman T, Friesel R, Schreiber AB (1984) Heparin binds endothelial cell growth factor, the principal endothelial cell mitogen in bovine brain. Science 225(4665):932–935
Nakamura M, Uehara Y, Asada M, Honda E, Nagai N, Kimata K, Suzuki M, Imamura T (2011) Sulfated glycosaminoglycans are required for specific and sensitive fibroblast growth factor (FGF) 19 signaling via FGF receptor 4 and betaKlotho. J Biol Chem 286(30):26418–26423
Ornitz DM, Itoh N (2001) Fibroblast growth factors. Genome Biol. 2(3):REVIEWS3005
Sugaya N, Habuchi H, Nagai N, Ashikari-Hada S, Kimata K (2008) 6-O-sulfation of heparan sulfate differentially regulates various fibroblast growth factor-dependent signalings in culture. J Biol Chem 283(16):10366–10376
Thompson LD, Pantoliano MW, Springer BA (1994) Energetic characterization of the basic fibroblast growth factor-heparin interaction: identification of the heparin binding domain. Biochemistry 33(13):3831–3840
Thornton SC, Mueller SN, Levine EM (1983) Human endothelial cells: use of heparin in cloning and long-term serial cultivation. Science 222(4624):623–625
Yoneda A, Asada M, Oda Y, Suzuki M, Imamura T (2000) Engineering of an FGF-proteoglycan fusion protein with heparin-independent, mitogenic activity. Nat Biotechnol 18(6):641–644
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Japan
About this entry
Cite this entry
Imamura, T. (2014). FGF System and GAGs. In: Endo, T., Seeberger, P., Hart, G., Wong, CH., Taniguchi, N. (eds) Glycoscience: Biology and Medicine. Springer, Tokyo. https://doi.org/10.1007/978-4-431-54836-2_57-1
Download citation
DOI: https://doi.org/10.1007/978-4-431-54836-2_57-1
Received:
Accepted:
Published:
Publisher Name: Springer, Tokyo
Online ISBN: 978-4-431-54836-2
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences