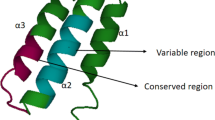
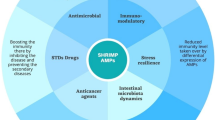
Abstract
Antimicrobial peptides (AMP) belong to the innate arm of immunity and serve as critical defence weapons across the living system. They have a multidimensional role in regulation of immunity which includes their direct antimicrobial response, chemoattraction, immune cell recruitment, stimulation of cytokine secretion and even triggering the adaptive immune responses. AMP-mediated regulation is intricate as these molecules keep a check on proinflammatory cytokines to prevent cytotoxicity in host. These host-derived peptides represent a divergent family in fish and have been tailored to initiate the appropriate response for the pathogen profile encountered by the species. The five classes of fish AMPs—piscidins, β-defensins, hepcidins, cathelicidins and histone-derived peptides—have been investigated for their antimicrobial effects against a wide variety of fish pathogens. The aquaculture industry often resorts to vaccines or antibiotics for evading fish disease; however, the therapeutic dosage of vaccines has the risks of cytotoxicity in host, while the latter promotes the already looming threat of antibiotic resistance. Under these circumstances, the exploitation of the broad-spectrum antimicrobial response of AMPs is not only an effective but also a safe means for combatting pathogenic infections in fish. Interestingly, the fish AMPs have also been found to generate effective immune response against various human pathogens and cancer cells, and this area has been extensively studied for its direct application in human health. However, there is a dearth of reports on their field application for amelioration of fish health. This chapter is focussed on fish AMPs, and it highlights their role in nurturing fish health and their potential for development of therapeutics.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Acosta J et al (2019) In vitro immunomodulatory activities of peptides derived from Salmo salar NK-lysin and cathelicidin in fish cells. Fish Shellfish Immunol 88(March):587–594. https://doi.org/10.1016/j.fsi.2019.03.034
Amparyup P et al (2020) Transcriptome analysis identifies immune-related genes and antimicrobial peptides in Siamese fighting fish (Betta splendens). Fish Shellfish Immunol 99:403–413. https://doi.org/10.1016/j.fsi.2020.02.030
Anooja VV et al (2020) Structural, functional and phylogenetic analysis of a beta defensin gene from the Whipfin silverbiddy, Gerres filamentosus (Cuvier, 1829). Gene Reports 21:100805. https://doi.org/10.1016/j.genrep.2020.100805
Bacalum M et al (2017) Modulating short tryptophan- and arginine-rich peptides activity by substitution with histidine. Biochim Biophys Acta Gen Subj 1861(7):1844–1854. https://doi.org/10.1016/j.bbagen.2017.03.024
Bae JS et al (2018) Gene expressions and biological activities of two novel antimicrobial peptides (AMPs) isolated from leucocytes of the rock bream, Oplegnathus fasciatus. Aquaculture 495(2017):35–43. https://doi.org/10.1016/j.aquaculture.2018.05.021
Bahar AA, Ren D (2013) Antimicrobial peptides. Pharmaceuticals 6(12):1543–1575. https://doi.org/10.3390/ph6121543
Banerjee R et al (2019) ‘Cloning and identification of antimicrobial peptide, hepcidin from freshwater carp. 3 Biotech 9(9):1–12. https://doi.org/10.1007/s13205-019-1874-6
Bao B et al (2005) Catfish hepcidin gene is expressed in a wide range of tissues and exhibits tissue-specific upregulation after bacterial infection. Dev Comp Immunol 29(11):939–950. https://doi.org/10.1016/j.dci.2005.03.006
Bergsson G et al (2005) Isolation and identification of antimicrobial components from the epidermal mucus of Atlantic cod (Gadus morhua). FEBS J 272(19):4960–4969. https://doi.org/10.1111/j.1742-4658.2005.04906.x
Broekman DC et al (2011) Functional characterization of codCath, the mature cathelicidin antimicrobial peptide from Atlantic cod (Gadus morhua). Peptides 32(10):2044–2051. https://doi.org/10.1016/j.peptides.2011.09.012
Brunner SR, Varga JFA, Dixon B (2020) Antimicrobial peptides of salmonid fish: from form to function. Biology 9(8):1–17. https://doi.org/10.3390/biology9080233
Cai L et al (2012) Recombinant medaka (Oryzias melastigmus) pro-hepcidin: multifunctional characterization. Comp Biochem Physiol B Biochem Mol Biol 161(2):140–147. https://doi.org/10.1016/j.cbpb.2011.10.006
Cardoso MH et al (2020) Computer-aided design of antimicrobial peptides: are we generating effective drug candidates? Front Microbiol 10:1–15. https://doi.org/10.3389/fmicb.2019.03097
Chaturvedi P, Bhat RAH, Pande A (2020) Antimicrobial peptides of fish: innocuous alternatives to antibiotics. Rev Aquac 12(1):85–106. https://doi.org/10.1111/raq.12306
Chen Y et al (2020) Anti-infective effects of a fish-derived antimicrobial peptide against drug-resistant bacteria and its synergistic effects with antibiotic. Front Microbiol 11:1–12. https://doi.org/10.3389/fmicb.2020.602412
Choi KM et al (2020) Two short antimicrobial peptides derived from prosaposin-like proteins in the starry flounder (Platichthys stellatus). Fish Shellfish Immunol 105:95–103. https://doi.org/10.1016/j.fsi.2020.05.075
Colorni A et al (2008) Activity of the antimicrobial polypeptide piscidin 2 against fish ectoparasites. J Fish Dis 31(6):423–432. https://doi.org/10.1111/j.1365-2761.2008.00922.x
Cuesta A, Meseguer J, Esteban MÁ (2011) Molecular and functional characterization of the gilthead seabream β-defensin demonstrate its chemotactic and antimicrobial activity. Mol Immunol 48(12–13):1432–1438. https://doi.org/10.1016/j.molimm.2011.03.022
D’Este F et al (2016) Antimicrobial and host cell-directed activities of Gly/Ser-rich peptides from salmonid cathelicidins. Fish Shellfish Immunol 59:456–468. https://doi.org/10.1016/j.fsi.2016.11.004
Dai J et al (2020) The effect of dietary cecropin AD on intestinal health, immune response and disease resistance of juvenile turbot (Scophthalmus maximus L.). Fish Shellfish Immunol 100:117–125. https://doi.org/10.1016/j.fsi.2020.02.052
De Bruijn I et al (2012) Immune gene expression in trout cell lines infected with the fish pathogenic oomycete Saprolegnia parasitica. Dev Comp Immunol 38(1):44–54. https://doi.org/10.1016/j.dci.2012.03.018
Dong B et al (2017) High throughput identification of antimicrobial peptides from fish gastrointestinal microbiota. Toxins 9(9):266. https://doi.org/10.3390/toxins9090266
Falco A et al (2009) Antimicrobial peptides as model molecules for the development of novel antiviral agents in aquaculture. Mini-Rev Med Chem 9(10):1159–1164. https://doi.org/10.2174/138955709789055171
Fernandes JMO et al (2002) Anti-microbial properties of histone H2A from skin secretions of rainbow trout, Oncorhynchus mykiss. Biochem J 368(2):611–620. https://doi.org/10.1042/BJ20020980
Ganz T et al (1985) Defensins. Natural peptide antibiotics of human neutrophils. J Clin Investig 76(4):1427–1435. https://doi.org/10.1172/JCI112120
Hancock REW, Chapple DS (1999) Peptide antibiotics. Antimicrob Agents Chemother 43(6):1317–1323. https://doi.org/10.1128/aac.43.6.1317
Hancock REW, Sahl HG (2006) Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat Biotechnol 24(12):1551–1557. https://doi.org/10.1038/nbt1267
Harte A et al (2020) Five subfamilies of β-defensin genes are present in salmonids: evolutionary insights and expression analysis in Atlantic salmon Salmo salar. Dev Comp Immunol 104:103560. https://doi.org/10.1016/j.dci.2019.103560
Hayden RM et al (2015) Complementary effects of host defense peptides piscidin 1 and piscidin 3 on DNA and lipid membranes: biophysical insights into contrasting biological activities. J Phys Chem B 119(49):15235–15246. https://doi.org/10.1021/acs.jpcb.5b09685
Hemshekhar M, Anaparti V, Mookherjee N (2016) Functions of cationic host defense peptides in immunity. Pharmaceuticals 9(3):40. https://doi.org/10.3390/ph9030040
Hsieh JC, Pan CY, Chen JY (2010) Tilapia hepcidin (TH)2-3 as a transgene in transgenic fish enhances resistance to Vibrio vulnificus infection and causes variations in immune-related genes after infection by different bacterial species. Fish Shellfish Immunol 29(3):430–439. https://doi.org/10.1016/j.fsi.2010.05.001
Huan Y et al (2020) Antimicrobial peptides: classification, design, application and research progress in multiple fields. Front Microbiol 11:1–21. https://doi.org/10.3389/fmicb.2020.582779
Huang PH, Chen JY, Kuo CM (2007) Three different hepcidins from tilapia, Oreochromis mossambicus: analysis of their expressions and biological functions. Mol Immunol 44(8):1922–1934. https://doi.org/10.1016/j.molimm.2006.09.031
Huang HN, Pan CY, Chen JY (2018) Grouper (Epinephelus coioides) antimicrobial peptide epinecidin-1 exhibits antiviral activity against foot-and-mouth disease virus in vitro. Peptides 106:91–95. https://doi.org/10.1016/j.peptides.2018.07.003
Huang HN et al (2020) Dietary supplementation of recombinant tilapia piscidin 4-expressing yeast enhances growth and immune response in Lates calcarifer. Aquac Rep 16:100254. https://doi.org/10.1016/j.aqrep.2019.100254
Jin JY et al (2010) Antibacterial and antiviral roles of a fish β-defensin expressed both in pituitary and testis. PLoS One 5(12):e12883. https://doi.org/10.1371/journal.pone.0012883
Kang X et al (2019) DRAMP 2.0, an updated data repository of antimicrobial peptides. Sci Data 6(1):1–10. https://doi.org/10.1038/s41597-019-0154-y
Kim HS et al (1996) cDNA cloning and characterization of buforin I, an antimicrobial peptide: a cleavage product of histone H2A pathogenic microorganisms, are widely distributed in nature and often show marked inter-species diversity in structure and spectrum of activity. Biochem Biophys Res Commun 387(2):381–387. http://pojagi.com/ParkChanBae/publication_files/publications/BBRC_96.pdf
Kim CH, Kim EJ, Nam YK (2019) Chondrostean sturgeon hepcidin: an evolutionary link between teleost and tetrapod hepcidins. Fish Shellfish Immunol 88:117–125. https://doi.org/10.1016/j.fsi.2019.02.045
Kuan S et al (2012) Binding kinetics of grouper nervous necrosis viruses with functionalized antimicrobial peptides by nanomechanical detection. Biosens Bioelectron 31(1):116–123. https://doi.org/10.1016/j.bios.2011.10.003
Kumar P, Kizhakkedathu JN, Straus SK (2018) Antimicrobial peptides: diversity, mechanism of action and strategies to improve the activity and biocompatibility in vivo. Biomol Ther 8(1). https://doi.org/10.3390/biom8010004
Lai R et al (2002) An anionic antimicrobial peptide from toad Bombina maxima. Biochem Biophys Res Commun 295(4):796–799. https://doi.org/10.1016/S0006-291X(02)00762-3
Lee E et al (2014) Role of phenylalanine and valine10 residues in the antimicrobial activity and cytotoxicity of piscidin-1. PLoS One 9(12):1–30. https://doi.org/10.1371/journal.pone.0114453
Lei J et al (2019) The antimicrobial peptides and their potential clinical applications. Am J Transl Res 11(7):3919–3931
Li J et al (2017) Membrane active antimicrobial peptides: translating mechanistic insights to design. Front Neurosci 11:1–18. https://doi.org/10.3389/fnins.2017.00073
Lyu Y, Fitriyanti M, Narsimhan G (2019) Nucleation and growth of pores in 1,2-Dimyristoyl-sn-glycero-3-phosphocholine (DMPC)/cholesterol bilayer by antimicrobial peptides melittin, its mutants and cecropin P1. Colloids Surf B: Biointerfaces 173:121–127. https://doi.org/10.1016/j.colsurfb.2018.09.049
Mahlapuu M et al (2016) Antimicrobial peptides: an emerging category of therapeutic agents. Front Cell Infect Microbiol 6:1–12. https://doi.org/10.3389/fcimb.2016.00194
Maier VH et al (2008) Characterisation of cathelicidin gene family members in divergent fish species. Mol Immunol 45(14):3723–3730. https://doi.org/10.1016/j.molimm.2008.06.002
Mardirossian M et al (2019) Proline-rich peptides with improved antimicrobial activity against E. coli, K. pneumoniae, and A. baumannii. ChemMedChem 14(24):2025–2033. https://doi.org/10.1002/cmdc.201900465
Masso-Silva JA, Diamond G (2014) Antimicrobial peptides from fish. Pharmaceuticals 7(3):265–310. https://doi.org/10.3390/ph7030265
Mehrnejad F, Zarei M (2010) Molecular dynamics simulation study of the interaction of piscidin 1 with DPPC bilayers: structure-activity relationship. J Biomol Struct Dyn 27(4):551–559. https://doi.org/10.1080/07391102.2010.10507338
Menousek J et al (2012) Database screening and in vivo efficacy of antimicrobial peptides against methicillin-resistant Staphylococcus aureus USA300. Int J Antimicrob Agents 39(5):402–406. https://doi.org/10.1016/j.ijantimicag.2012.02.003
Muncaster S et al (2018) Antimicrobial peptides within the yellowtail kingfish (Seriola lalandi). Dev Comp Immunol 80:67–80. https://doi.org/10.1016/j.dci.2017.04.014
Münch D, Sahl HG (2015) Structural variations of the cell wall precursor lipid II in gram-positive bacteria—impact on binding and efficacy of antimicrobial peptides. Biochim Biophys Acta Biomembr 1848(11):3062–3071. https://doi.org/10.1016/j.bbamem.2015.04.014
Murrell B et al (2018) Evolution of recognition of ligands from gram-positive bacteria: similarities and differences in the TLR2-mediated response between mammalian vertebrates and teleost fish. Fish Shellfish Immunol 35(1):2355–2368. https://doi.org/10.4049/jimmunol.0900990
Mylonakis E et al (2016) Diversity, evolution and medical applications of insect antimicrobial peptides. Philos Trans R Soc B Biol Sci 371(1695):20150290. https://doi.org/10.1098/rstb.2015.0290
Nam BH et al (2010) Multiple β-defensin isoforms identified in early developmental stages of the teleost Paralichthys olivaceus. Fish Shellfish Immunol 28(2):267–274. https://doi.org/10.1016/j.fsi.2009.11.004
Neves JV et al (2017) Hamp1 but not Hamp2 regulates ferroportin in fish with two functionally distinct hepcidin types. Sci Rep 7(1):1–14. https://doi.org/10.1038/s41598-017-14933-5
Nguyen LT, Haney EF, Vogel HJ (2011) The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol 29(9):464–472. https://doi.org/10.1016/j.tibtech.2011.05.001
Noga EJ et al (2009) Piscidin 4, a novel member of the piscidin family of antimicrobial peptides. Comp Biochem Physiol B Biochem Mol Biol 152(4):299–305. https://doi.org/10.1016/j.cbpb.2008.12.018
Omardien S et al (2018) Synthetic antimicrobial peptides delocalize membrane bound proteins thereby inducing a cell envelope stress response. Biochim Biophys Acta Biomembr 1860(11):2416–2427. https://doi.org/10.1016/j.bbamem.2018.06.005
Oren Z, Shai Y (1996) A class of highly potent antibacterial peptides derived from pardaxin, a pore-forming peptide isolated from Moses sole fish Pardachirus marmoratus. Eur J Biochem 237(1):303–310. https://doi.org/10.1111/j.1432-1033.1996.0303n.x
Pan CY et al (2009) In vitro activities of three synthetic peptides derived from epinecidin-1 and an anti-lipopolysaccharide factor against Propionibacterium acnes, Candida albicans, and Trichomonas vaginalis. Peptides 30(6):1058–1068. https://doi.org/10.1016/j.peptides.2009.02.006
Pan CY et al (2017) Study of the antimicrobial activity of tilapia piscidin 3 (TP3) and TP4 and their effects on immune functions in hybrid tilapia (Oreochromis spp.). PLoS One 12(1):1–23. https://doi.org/10.1371/journal.pone.0169678
Park CB et al (1997) A novel antimicrobial peptide from the loach, Misgurnus anguillicaudatus. FEBS Lett 411:173–178
Park NG et al (2011) Structure-activity relationships of piscidin 4, a piscine antimicrobial peptide. Biochemistry 50(16):3288–3299. https://doi.org/10.1021/bi101395j
Perrin BS et al (2014) High-resolution structures and orientations of antimicrobial peptides piscidin 1 and piscidin 3 in fluid bilayers reveal tilting, kinking, and bilayer immersion. J Am Chem Soc 136(9):3491–3504. https://doi.org/10.1021/ja411119m
Pouny Y et al (1992) Interaction of antimicrobial dermaseptin and its fluorescently labeled analogs with phospholipid membranes. Biochemistry 31(49):12416–12423. https://doi.org/10.1021/bi00164a017
Preena PG et al (2020) Antimicrobial resistance in aquaculture: a crisis for concern. Biologia 75(9):1497–1517. https://doi.org/10.2478/s11756-020-00456-4
Raju SV et al (2021) Piscidin, fish antimicrobial peptide: structure, classification, properties, mechanism, gene regulation and therapeutical importance. Int J Pept Res Ther 27(1):91–107. https://doi.org/10.1007/s10989-020-10068-w
Ramirez-Acuña JM et al (2019) Diabetic foot ulcers: current advances in antimicrobial therapies and emerging treatments. Antibiotics 8(4):1–32. https://doi.org/10.3390/antibiotics8040193
Röhrl J et al (2010) Specific binding and chemotactic activity of mBD4 and its functional orthologue hBD2 to CCR6-expressing cells. J Biol Chem 285(10):7028–7034. https://doi.org/10.1074/jbc.M109.091090
Ruangsri J et al (2013) A novel beta-defensin antimicrobial peptide in Atlantic cod with stimulatory effect on phagocytic activity. PLoS One 8(4):e62302. https://doi.org/10.1371/journal.pone.0062302
Salger SA et al (2016) A diverse family of host-defense peptides (piscidins) exhibit specialized anti-bacterial and anti-protozoal activities in fishes. PLoS One 11(8):1–25. https://doi.org/10.1371/journal.pone.0159423
Schmitt P et al (2015) Immunomodulatory effect of cathelicidins in response to a β-glucan in intestinal epithelial cells from rainbow trout. Dev Comp Immunol 51(1):160–169. https://doi.org/10.1016/j.dci.2015.03.007
Scocchi M et al (2009) The salmonid cathelicidins: a gene family with highly varied C-terminal antimicrobial domains. Comp Biochem PhysiolB Biochem Mol Biol 152(4):376–381. https://doi.org/10.1016/j.cbpb.2009.01.003
Scocchi M et al (2016) Cathelicidins: an ancient family of fish antimicrobial peptides. Lessons in immunity: from single-cell organisms to mammals. Elsevier Inc. https://doi.org/10.1016/B978-0-12-803252-7.00017-5
Shabir U et al (2018) Fish antimicrobial peptides (AMP’s) as essential and promising molecular therapeutic agents: a review’. Microb Pathog 114:50–56. https://doi.org/10.1016/j.micpath.2017.11.039
Silphaduang U, Colorni A, Noga EJ (2006) Evidence for widespread distribution of piscidin antimicrobial peptides in teleost fish. Dis Aquat Org 72(3):241–252. https://doi.org/10.3354/dao072241
Song R et al (2012) Isolation and characterization of an antibacterial peptide fraction from the pepsin hydrolysate of half-fin anchovy (Setipinna taty). Molecules 17(3):2980–2991. https://doi.org/10.3390/molecules17032980
Steiner H, Hultmark D, Engström A, Bennich H, Boman HG (1981) Sequence and specificity of two antibacterial proteins involved in insect immunity. Nature 820:246–248. https://doi.org/10.1038/292246a0. PMID: 7019715
Su YL et al (2019) Effects of antimicrobial peptides on serum biochemical parameters, antioxidant activity and non-specific immune responses in Epinephelus coioides. Fish Shellfish Immunol 86:1081–1087. https://doi.org/10.1016/j.fsi.2018.12.056
Sung WS, Lee J, Lee DG (2008) Fungicidal effect and the mode of action of piscidin 2 derived from hybrid striped bass. Biochem Biophys Res Commun 371(3):551–555. https://doi.org/10.1016/j.bbrc.2008.04.107
Tan SY, Tatsumura Y (2015) Alexander Fleming (1881–1955): discoverer of penicillin. Singapore Med J 56(7):366–367. https://doi.org/10.11622/smedj.2015105
Tsai PW et al (2011) Human antimicrobial peptide LL-37 inhibits adhesion of Candida albicans by interacting with yeast cell-wall carbohydrates. PLoS One 6(3):e1775. https://doi.org/10.1371/journal.pone.0017755
Umasuthan N et al (2016) Molecular, genomic, and expressional delineation of a piscidin from rock bream (Oplegnathus fasciatus) with evidence for the potent antimicrobial activities of Of-Pis1 peptide. Fish Shellfish Immunol 48:154–168. https://doi.org/10.1016/j.fsi.2015.11.005
Valero Y, Chaves-Pozo E, Meseguer J, Esteban MA, Cuesta A (2013) Antimicrobial peptides. In: Biological role of fish antimicrobial peptides. Nova Science Publishers, New York, United States, pp 31–60
Valero Y et al (2020) Antimicrobial peptides from fish: beyond the fight against pathogens. Rev Aquac 12(1):224–253. https://doi.org/10.1111/raq.12314
Varga JFA, Bui-Marinos MP, Katzenback BA (2019) Frog skin innate immune defences: sensing and surviving pathogens. Front Immunol 10:1–21. https://doi.org/10.3389/fimmu.2018.03128
Vollmer W, Blanot D, De Pedro MA (2008) Peptidoglycan structure and architecture. FEMS Microbiol Rev 32(2):149–167. https://doi.org/10.1111/j.1574-6976.2007.00094.x
Wang KJ et al (2009) Cloning and expression of a hepcidin gene from a marine fish (Pseudosciaena crocea) and the antimicrobial activity of its synthetic peptide. Peptides 30(4):638–646. https://doi.org/10.1016/j.peptides.2008.12.014
Wang YA, Kung CW, Chen JY (2010) Antiviral activity by fish antimicrobial peptides of epinecidin-1 and hepcidin 1-5 against nervous necrosis virus in medaka. Peptides 31(6):1026–1033. https://doi.org/10.1016/j.peptides.2010.02.025
Wang G, Li X, Wang Z (2016) APD3: the antimicrobial peptide database as a tool for research and education. Nucleic Acids Res 44(D1):D1087–D1093. https://doi.org/10.1093/nar/gkv1278
Wang S et al (2021) Effects of mixed antimicrobial peptide on the growth performance, antioxidant and immune responses and disease resistance of Pengze crucian carp (Carassius auratus var. Pengze). Fish Shellfish Immunol 114:112–118. https://doi.org/10.1016/j.fsi.2021.04.017
Wangkahart E, Secombes CJ, Wang T (2019) Dissecting the immune pathways stimulated following injection vaccination of rainbow trout (Oncorhynchus mykiss) against enteric redmouth disease (ERM). Fish Shellfish Immunol 85:18–30. https://doi.org/10.1016/j.fsi.2017.07.056
Wimley WC, Hristova K (2011) Antimicrobial peptides: successes, challenges and unanswered questions. J Membr Biol 239(1–2):27–34. https://doi.org/10.1007/s00232-011-9343-0
Wu XM et al (2019) Histone H2A cooperates with RIP2 to induce the expression of antibacterial genes and MHC related genes. Dev Comp Immunol 101:103455. https://doi.org/10.1016/j.dci.2019.103455
Xu T et al (2012) Miiuy croaker hepcidin gene and comparative analyses reveal evidence for positive selection. PLoS One 7(4):e35449. https://doi.org/10.1371/journal.pone.0035449
Zasloff M (1987) Magainins, a class of antimicrobial peptides from Xenopus skin: isolation, characterization of two active forms, and partial cDNA sequence of a precursor. Proc Natl Acad Sci U S A 84(15):5449–5453. https://doi.org/10.1073/pnas.84.15.5449
Zheng L et al (2021) Molecular characterization and antiparasitic activity analysis of a novel piscidin 5-like type 4 from Larimichthys crocea. Mol Immunol 129:12–20. https://doi.org/10.1016/j.molimm.2020.11.015
Zhou Y et al (2019) A β-defensin gene of Trachinotus ovatus might be involved in the antimicrobial and antiviral immune response. Dev Comp Immunol 92:105–115. https://doi.org/10.1016/j.dci.2018.11.011
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Priyam, M., Bhat, R.A., Kumar, N. (2021). Recent Advances in Antimicrobial Peptides to Improve Fish Health. In: Gupta, S.K., Giri, S.S. (eds) Biotechnological Advances in Aquaculture Health Management . Springer, Singapore. https://doi.org/10.1007/978-981-16-5195-3_8
Download citation
DOI: https://doi.org/10.1007/978-981-16-5195-3_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-5194-6
Online ISBN: 978-981-16-5195-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)