Abstract
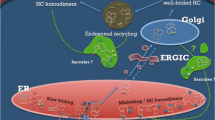
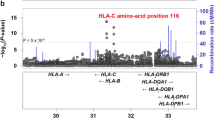
Major Histocompatibility Complex (MHC), firstly identified as the main factor determining the fate of organ transplantation, is involved in antigen presentation to T cells. Among the ever-increasing list of genes identified to be related to different autoimmune-inflammatory diseases, HLA loci have shown the strongest association. This is because of their role in antigen presentation to T lymphocytes and their involvement in the negative selection of auto-reactive thymocytes. One of the most striking associations between an autoimmune disorder and an HLA allele is for Ankylosing Spondylitis (AS) and HLA-B27. Several mechanisms have been proposed to explain how HLA-B27 can participate in the pathogenesis of AS. However, the association of other HLA loci and also HLA-related genes with AS has been evidenced. Epigenetics is another important player in the etiopathology of AS through affecting the expression of the genes involved in different physiologic and immunologic processes. In this chapter, we will explain the cutting-edge knowledge about the probable mechanisms through which, HLA-B27 plays a role in AS development. Then, we discuss other HLA and HLA-related genes shown to be associated with AS in different populations. At the end of the chapter, we describe the impact of epigenetics as another predisposing factor.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Colbert RA, et al. HLA-B27 misfolding and spondyloarthropathies. Prion. 2009;3(1):15–26.
Akassou A, Bakri Y. Does HLA-B27 status influence ankylosing spondylitis phenotype? Clin Med Insights: Arthritis and Musculoskeletal Disorders. 2018;11:1179544117751627.
Chen B, et al. Role of HLA-B27 in the pathogenesis of ankylosing spondylitis. Mol Med Rep. 2017;15(4):1943–51.
Breban M, et al. Revisiting MHC genes in spondyloarthritis. Curr Rheumatol Rep. 2015;17(6):40.
Nicknam MH, et al. Determination of HLA-B27 subtypes in Iranian patients with ankylosing spondylitis. Iran J Allergy Asthma Immunol. 2008;7(1):19–24.
Colbert RA, et al. From HLA-B27 to spondyloarthritis: a journey through the ER. Immunol Rev. 2010;233(1):181–202.
Gran JT, Husby G. HLA-B27 and spondyloarthropathy: value for early diagnosis? J Med Genet. 1995;32(7):497.
Reveille JD. An update on the contribution of the MHC to as susceptibility. Clin Rheumatol. 2014;33(6):749–57.
Nicknam M, et al. Association between HLA-B27 antigen and ankylosing spondylitis in Iranian patients. Hakim. 2005;8(1):29–34.
Nicknam M, et al. Comparison of val idity of microlymphocytotoxicity and flowcytometry methods with PCR for HLA-B27 antigen typing. Med J Islamic Republic Iran (MJIRI). 2003;17(1):75–9.
Brown M. Progress in studies of the genetics of ankylosing spondylitis. in clinical and experimental rheumatology. In: Clinical & exper rheumatology via santa maria, vol. 31. Italy: PISA; 2010. p. 56126.
Hanson A, Brown MA. Genetics and the causes of ankylosing spondylitis. Rheum Dis Clin. 2017;43(3):401–14.
Burney R, et al. Analysis of the MHC class II encoded components of the HLA class I antigen processing pathway in ankylosing spondylitis. Ann Rheum Dis. 1994;53(1):58–60.
Colbert RA, Tran TM, Layh-Schmitt G. HLA-B27 misfolding and ankylosing spondylitis. Mol Immunol. 2014;57(1):44–51.
Ganjalikhani Hakemi M, et al. Optimization of human Th17 cell differentiation in vitro: evaluating different polarizing factors. In Vitro Cell Dev Biol Animal. 2011;47(8):581.
Ringrose J. HLA-B27 associated spondyloarthropathy, an autoimmune disease based on crossreactivity between bacteria and HLA-B27? Ann Rheum Dis. 1999;58(10):598–610.
Robinson PC, Brown MA. Genetics of ankylosing spondylitis. Mol Immunol. 2014;57(1):2–11.
Mahmoudi M, et al. New insights toward the pathogenesis of ankylosing spondylitis; genetic variations and epigenetic modifications. Mod Rheumatol. 2017;27(2):198–209.
Fechtenbaum M, et al. MICA and NKG2D variants as risk factors in spondyloarthritis: a case–control study. Genes Immun. 2019;20:599–605.
O’rielly DD, Uddin M, Rahman P. Ankylosing spondylitis: beyond genome-wide association studies. Curr Opin Rheumatol. 2016;28(4):337–45.
Tsui FW, et al. The genetic basis of ankylosing spondylitis: new insights into disease pathogenesis. Appl Clin Genet. 2014;7:105.
Qian Y, et al. Genetic association between TAP1 and TAP2 polymorphisms and ankylosing spondylitis: a systematic review and meta-analysis. Inflamm Res. 2017;66(8):653–61.
Feng M, et al. TAP1 and TAP2 polymorphisms associated with ankylosing spondylitis in genetically homogenous Chinese Han population. Hum Immunol. 2009;70(4):257–61.
Agrawal N, Brown M. Genetic associations and functional characterization of M1 aminopeptidases and immune-mediated diseases. Genes Immun. 2014;15(8):521.
Alvarez-Navarro C, de Castro JAL. ERAP1 structure, function and pathogenetic role in ankylosing spondylitis and other MHC-associated diseases. Mol Immunol. 2014;57(1):12–21.
Zhai J, et al. Immunogenetic study in Chinese population with ankylosing spondylitis: are there specific genes recently disclosed? Clin Dev Immunol. 2013;2013:419357.
de Castro JAL. How ERAP1 and ERAP2 shape the peptidomes of disease-associated MHC-I proteins. Front Immunol. 2018;9:2463.
Driscoll J, et al. MHC-linked LMP gene products specifically alter peptidase activities of the proteasome. Nature. 1993;365(6443):262.
Gaczynska M, Rock KL, Goldberg AL. γ-Interferon and expression of MHC genes regulate peptide hydrolysis by proteasomes. Nature. 1993;365(6443):264.
Westman P, et al. TAP1 and TAP2 polymorphism in HLA-B27-positive subpopulations: no allelic differences in ankylosing spondylitis and reactive arthritis. Hum Immunol. 1995;44(4):236–42.
Soleimanifar N, et al. Effect of food intake and ambient air pollution exposure on ankylosing spondylitis disease activity. Adv Rheumatol. 2019;59(1):9.
Roberts S, et al. Epigenetics of osteoarticular diseases: recent developments. Rheumatol Int. 2015;35(8):1293–305.
Lai N-S, et al. Association between cytokines and methylation of SOCS-1 in serum of patients with ankylosing spondylitis. Mol Biol Rep. 2014;41(6):3773–80.
Aslani S, et al. Evaluation of DNMT1 gene expression profile and methylation of its promoter region in patients with ankylosing spondylitis. Clin Rheumatol. 2016;35(11):2723–31.
Karami J, et al. Promoter hypermethylation of BCL11B gene correlates with downregulation of gene transcription in ankylosing spondylitis patients. Genes Immun. 2017;18(3):170.
Hao J, et al. Genome-wide DNA methylation profile analysis identifies differentially methylated loci associated with ankylosis spondylitis. Arthritis Res Ther. 2017;19(1):177.
Mohammadi H, et al. MicroRNA implications in the etiopathogenesis of ankylosing spondylitis. J Cell Physiol. 2018;233(8):5564–73.
Perez-Sanchez C, et al. Circulating microRNAs as potential biomarkers of disease activity and structural damage in ankylosing spondylitis patients. Hum Mol Genet. 2018;27(5):875–90.
Li Z, et al. The role of MicroRNAS in ankylosing spondylitis. Medicine. 2016;95(14):e3325.
Wang M, et al. Overexpression of miR-31 in peripheral blood mononuclear cells (PBMC) from patients with ankylosing spondylitis. Medical Science Monitor: International Medical Journal of Experimental and Clinical Research. 2017;23:5488.
Yang L, et al. miRNA copy number variants confer susceptibility to acute anterior uveitis with or without ankylosing spondylitis. Invest Ophthalmol Vis Sci. 2017;58(4):1991–2001.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Ganjalikhani Hakemi, M. (2022). Association of HLA and HLA-Related Genes with Ankylosing Spondylitis. In: Nicknam, M.H. (eds) Ankylosing Spondylitis - Axial Spondyloarthritis. Springer, Singapore. https://doi.org/10.1007/978-981-16-4733-8_4
Download citation
DOI: https://doi.org/10.1007/978-981-16-4733-8_4
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-4732-1
Online ISBN: 978-981-16-4733-8
eBook Packages: MedicineMedicine (R0)