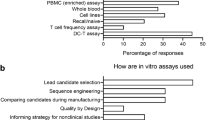
Abstract
Over the past decade, immunotherapy has tremendously augmented the traditional treatment strategies in cancer cure. It has provided newer opportunities to research and development of enormous varieties of lifesaving drugs—each representing its strengths and weaknesses. None-the-less, it has improved treatment protocols, provided simple solutions to complex biological problems, and increased the overall disease-free survival within patients. However, the beneficiaries of these technological advancements are very few. This may be due to the heterogeneity within individuals (which is generally not considered for development of Immunotherapeutics) or due to the fact that it is high risk, high investment, and high returns technology, supplemented with side effects. Some of these manageable factors like cost can be worked upon by newer strategies or improvising existing manufacturing processes—to scale up within regulatory compliances. Process scale up is an integral step in large scale manufacturing that demands for critical understanding of process attribute, analytical tool, and product quality. Designing of scalable technology and manufacturing machineries with emphasis on process invariability, minimized time, and cost of therapy will provide the success of any immunotherapeutic drug. This chapter is an attempt to provide a bird’s eye view on the various opportunities available in the drug designing, scale up and analytics of immunotherapeutic drugs.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ansar W, Ghosh S (2013) Monoclonal antibodies: a tool in clinical research. Indian J Clin Med 4:S11968. https://doi.org/10.4137/ijcm.s11968
Braasch K, Nikolic-Jaric M, Cabel T et al (2013) The changing dielectric properties of CHO cells can be used to determine early apoptotic events in a bioprocess. Biotechnol Bioeng 110(11):2902–2914. https://doi.org/10.1002/bit.24976
Cancer.net (2018) Immunotherapy - An Introduction. https://www.cancer.net/navigating-cancer-care/videos/treatments-tests-and-procedures/immunotherapy-introduction. Published 2018
Cancer.net (2020) Understanding Immunotherapy. https://www.cancer.net/navigating-cancer-care/how-cancer-treated/immunotherapy-and-vaccines/understanding-immunotherapy
Childs RW, Carlsten M (2015) Therapeutic approaches to enhance natural killer cell cytotoxicity against cancer: the force awakens. Nat Rev Drug Discov 14(7):487–498. https://doi.org/10.1038/nrd4506
Collins M, Ling V, Carreno BM (2005) The B7 family of immune-regulatory ligands. Genome Biol 6(6):1–7. https://doi.org/10.1186/gb-2005-6-6-223
Csaszar E, Kirouac DC, Yu M et al (2012) Rapid expansion of human hematopoietic stem cells by automated control of inhibitory feedback signaling. Cell Stem Cell 10(2):218–229. https://doi.org/10.1016/j.stem.2012.01.003
Dougall WC, Kurtulus S, Smyth MJ, Anderson AC (2017) TIGIT and CD96: new checkpoint receptor targets for cancer immunotherapy. Immunol Rev 276(1):112–120. https://doi.org/10.1111/imr.12518
Eaker S, Abraham E, Allickson J et al (2017) Bioreactors for cell therapies: current status and future advances. Cytotherapy 19(1):9–18. https://doi.org/10.1016/j.jcyt.2016.09.011
Gimenez L, Simonet C, Malphettes L (2013) Scale-up considerations for monoclonal antibody production process: an oxygen transfer flux approach. BMC Proc 7(S6):P49. https://doi.org/10.1186/1753-6561-7-s6-p49
Hansmann J, Groeber F, Kahlig A, Kleinhans C, Walles H (2013) Bioreactors in tissue engineering-principles, applications and commercial constraints. Biotechnol J 8(3):298–307. https://doi.org/10.1002/biot.201200162
Iyer RK, Bowles PA, Kim H, Dulgar-Tulloch A (2018) Industrializing autologous adoptive immunotherapies: manufacturing advances and challenges. Front Med 5(MAY):1–9. https://doi.org/10.3389/fmed.2018.00150
Kaufman HL, Kohlhapp FJ, Zloza A (2015) Oncolytic viruses: a new class of immunotherapy drugs. Nat Rev Drug Discov 14(9):642–662. https://doi.org/10.1038/nrd4663
Krauss WC, Park JW, Kirpotin DB, Hong K, Benz CC (2000) Emerging antibody-based HER2 (ErbB-2/neu) therapeutics. Breast Dis 11:113–124. https://doi.org/10.3233/BD-1999-11110
Liu L, Sommermeyer D, Cabanov A, Kosasih P, Hill T, Riddell SR (2016) Inclusion of strep-tag II in design of antigen receptors for T-cell immunotherapy. Nat Biotechnol 34(4):430–434. https://doi.org/10.1038/nbt.3461
Lu RM, Hwang YC, Liu IJ et al (2020) Development of therapeutic antibodies for the treatment of diseases. J Biomed Sci 27(1):1–30. https://doi.org/10.1186/s12929-019-0592-z
Mac Cheever MA (2008) Twelve immunotherapy drugs that could cure cancers. Immunol Rev 222(1):357–368. https://doi.org/10.1111/j.1600-065X.2008.00604.x
Mahmuda A, Bande F, Al-Zihiry KJK et al (2017) Monoclonal antibodies: a review of therapeutic applications and future prospects. Trop J Pharm Res 16(3):713–722. https://doi.org/10.4314/tjpr.v16i3.29
Miliotou AN, Papadopoulou LC (2018) CAR T-cell therapy: a new era in Cancer immunotherapy. Curr Pharm Biotechnol 19(1):5–18. https://doi.org/10.2174/1389201019666180418095526
Miller JFAP, Sadelain M (2015) The journey from discoveries in fundamental immunology to cancer immunotherapy. Cancer Cell 27(4):439–449. https://doi.org/10.1016/j.ccell.2015.03.007
Mock U, Nickolay L, Philip B et al (2016) Automated manufacturing of chimeric antigen receptor T cells for adoptive immunotherapy using CliniMACS prodigy. Cytotherapy 18(8):1002–1011. https://doi.org/10.1016/j.jcyt.2016.05.009
Ni L, Dong C (2017) New checkpoints in cancer immunotherapy. Immunol Rev 276(1):52–65. https://doi.org/10.1111/imr.12524
Nickolay L, Mock U, Phillip B et al (2016) Automated Lentiviral transduction of T cells with CARS using the CliniMACS prodigy. Mol Ther 24(May):S180–S181. https://doi.org/10.1016/s1525-0016(16)33264-6
Ribatti D (2014) From the discovery of monoclonal antibodies to their therapeutic application: an historical reappraisal. Immunol Lett 161(1):96–99. https://doi.org/10.1016/j.imlet.2014.05.010
Rita Costa A, Elisa Rodrigues M, Henriques M, Azeredo J, Oliveira R (2010) Guidelines to cell engineering for monoclonal antibody production. Eur J Pharm Biopharm 74(2):127–138. https://doi.org/10.1016/j.ejpb.2009.10.002
Sahin U, Karikó K, Türeci Ö (2014) MRNA-based therapeutics-developing a new class of drugs. Nat Rev Drug Discov 13(10):759–780. https://doi.org/10.1038/nrd4278
Schilsky RL (2018) Tumor-Agnostic Treatment for Cancer: An Expert Perspective. https://www.cancer.net/blog/2018-12/tumor-agnostic-treatment-cancer-expert-perspective
Sharpe AH (2017) Introduction to checkpoint inhibitors and cancer immunotherapy. Immunol Rev 276(1):5–8. https://doi.org/10.1111/imr.12531
U.S. FDA (2017) Resources for Information on Approved Drugs. Fed Drug Adm 1–2. www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/default.htm
Wang X, Rivière I (2016) Clinical manufacturing of CAR T cells: foundation of a promising therapy. Mol Ther Oncol 3(February):16015. https://doi.org/10.1038/mto.2016.15
Zhu F, Shah NN, Xu H et al (2016) CAR-T cell production using the Clinimacs® prodigy system. Blood 128(22):5724–5724. https://doi.org/10.1182/blood.v128.22.5724.5724
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Palahe, P., Pardhi, V. (2021). Discovery, Screening Methods, Design Considerations, and Scale-up Aspects of Immunotherapeutic Drugs. In: Sawarkar, S.P., Nikam, V.S., Syed, S. (eds) Immunotherapy – A Novel Facet of Modern Therapeutics. Springer, Singapore. https://doi.org/10.1007/978-981-15-9038-2_8
Download citation
DOI: https://doi.org/10.1007/978-981-15-9038-2_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-9037-5
Online ISBN: 978-981-15-9038-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)