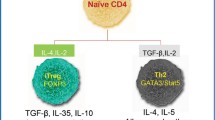
Abstract
Organ transplantation is the optimal treatment for terminal and irreversible organ failure. Achieving transplantation tolerance has long been the ultimate goal in the field of transplantation. Regulatory T cell (Treg)-based therapy is a promising novel approach for inducing donor organ-specific tolerance. Tregs play critical roles in the maintenance of immune homeostasis and self-tolerance, by promoting transplantation tolerance through a variety of mechanisms on different target cells, including anti-inflammatory cytokine production, induction of apoptosis, disruption of metabolic pathways, and mutual interaction with dendritic cells. The continued success of Treg-based therapy in the clinical setting is critically dependent on preclinical studies that support its translational potential. However, although some initial clinical trials of adoptive Treg therapy have successively demonstrated safety and efficacy for immunosuppressant minimization and transplantation tolerance induction, most Treg-based hematopoietic stem cell and solid organ clinical trials are still in their infancy. These clinical trials have not only focused on safety and efficacy but also included optimization and standardization protocols of good manufacturing practice regarding cell isolation, expansion, dosing, timing, specificity, quality control, concomitant immunosuppressants, and post-administration monitoring. We herein report a brief introduction of Tregs, including their phenotypic and functional characterization, and focus on the clinical translation of Treg-based therapeutic applications in the setting of transplantation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Andersson J, Tran DQ, Pesu M et al (2008) CD4+ FoxP3+ regulatory T cells confer infectious tolerance in a TGF-beta-dependent manner. J Exp Med 205:1975–1981. https://doi.org/10.1084/jem.20080308
Antonioli L, Pacher P, Vizi ES, Haskó G (2013) CD39 and CD73 in immunity and inflammation. Trends Mol Med 19:355–367. https://doi.org/10.1016/j.molmed.2013.03.005
Ashoor I, Najafian N, Korin Y et al (2013) Standardization and cross validation of alloreactive IFNγ ELISPOT assays within the clinical trials in organ transplantation consortium. Am J Transplant 13:1871–1879. https://doi.org/10.1111/ajt.12286
Battaglia M, Stabilini A, Roncarolo M-G (2005) Rapamycin selectively expands CD4+CD25+FoxP3+ regulatory T cells. Blood 105:4743–4748. https://doi.org/10.1182/blood-2004-10-3932
Battaglia M, Stabilini A, Migliavacca B et al (2006) Rapamycin promotes expansion of functional CD4+CD25+FOXP3+ regulatory T cells of both healthy subjects and type 1 diabetic patients. J Immunol 177:8338–8347. https://doi.org/10.4049/jimmunol.177.12.8338
Bestard O, Cruzado JM, Mestre M et al (2007) Achieving donor-specific hyporesponsiveness is associated with FOXP3+ regulatory T cell recruitment in human renal allograft infiltrates. J Immunol 179:4901–4909. https://doi.org/10.4049/jimmunol.179.7.4901
Bestard O, Crespo E, Stein M et al (2013) Cross-validation of IFN-γ Elispot assay for measuring alloreactive memory/effector T cell responses in renal transplant recipients. Am J Transplant 13:1880–1890. https://doi.org/10.1111/ajt.12285
Bickerstaff AA, VanBuskirk AM, Wakely E, Orosz CG (2000) Transforming growth factor-beta and interleukin-10 subvert alloreactive delayed type hypersensitivity in cardiac allograft acceptor mice. Transplantation 69:1517–1520. https://doi.org/10.1097/00007890-200004150-00055
Bloom DD, Chang Z, Fechner JH et al (2008) CD4+CD25+FOXP3+ regulatory T cells increase de novo in kidney transplant patients after immunodepletion with Campath-1H. Am J Transplant 8:793–802. https://doi.org/10.1111/j.1600-6143.2007.02134.x
Bluestone JA, Liu W, Yabu JM et al (2008) The effect of costimulatory and interleukin 2 receptor blockade on regulatory T cells in renal transplantation. Am J Transplant 8:2086–2096. https://doi.org/10.1111/j.1600-6143.2008.02377.x
Bluestone JA, Buckner JH, Fitch M et al (2015) Type 1 diabetes immunotherapy using polyclonal regulatory T cells. Sci Transl Med 7:315ra189. https://doi.org/10.1126/scitranslmed.aad4134
Boardman DA, Philippeos C, Fruhwirth GO et al (2017) Expression of a chimeric antigen receptor specific for donor HLA class I enhances the potency of human regulatory T cells in preventing human skin transplant rejection. Am J Transplant 17:931–943. https://doi.org/10.1111/ajt.14185
Bocian K, Borysowski J, Wierzbicki P et al (2010) Rapamycin, unlike cyclosporine A, enhances suppressive functions of in vitro-induced CD4+CD25+ Tregs. Nephrol Dial Transplant 25:710–717. https://doi.org/10.1093/ndt/gfp586
Brem-Exner BG, Sattler C, Hutchinson JA et al (2008) Macrophages driven to a novel state of activation have anti-inflammatory properties in mice. J Immunol 180:335–349. https://doi.org/10.4049/jimmunol.180.1.335
Brentjens RJ, Davila ML, Riviere I et al (2013) CD19-targeted T cells rapidly induce molecular remissions in adults with chemotherapy-refractory acute lymphoblastic leukemia. Sci Transl Med 5:177ra38. https://doi.org/10.1126/scitranslmed.3005930
Broichhausen C, Riquelme P, Geissler EK, Hutchinson JA (2012) Regulatory macrophages as therapeutic targets and therapeutic agents in solid organ transplantation. Curr Opin Organ Transplant 17:332–342. https://doi.org/10.1097/MOT.0b013e328355a979
Brunstein CG, Miller JS, Cao Q et al (2011) Infusion of ex vivo expanded T regulatory cells in adults transplanted with umbilical cord blood: safety profile and detection kinetics. Blood 117:1061–1070. https://doi.org/10.1182/blood-2010-07-293795
Brunstein CG, Miller JS, McKenna DH et al (2016) Umbilical cord blood-derived T regulatory cells to prevent GVHD: kinetics, toxicity profile, and clinical effect. Blood 127:1044–1051. https://doi.org/10.1182/blood-2015-06-653667
Brusko TM, Hulme MA, Myhr CB et al (2007) Assessing the in vitro suppressive capacity of regulatory T cells. Immunol Investig 36:607–628. https://doi.org/10.1080/08820130701790368
Calvo-Turrubiartes M, Romano-Moreno S, García-Hernández M et al (2009) Quantitative analysis of regulatory T cells in kidney graft recipients: a relationship with calcineurin inhibitor level. Transpl Immunol 21:43–49. https://doi.org/10.1016/j.trim.2009.02.002
Camirand G, Riella LV (2017) Treg-centric view of immunosuppressive drugs in transplantation: a balancing act. Am J Transplant 17:601–610. https://doi.org/10.1111/ajt.14029
Chandran S, Tang Q, Sarwal M et al (2017) Polyclonal regulatory T cell therapy for control of inflammation in kidney transplants. Am J Transplant 17:2945–2954. https://doi.org/10.1111/ajt.14415
Deaglio S, Dwyer KM, Gao W et al (2007) Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med 204:1257–1265. https://doi.org/10.1084/jem.20062512
Dijke IE, Hoeppli RE, Ellis T et al (2016) Discarded human thymus is a novel source of stable and Long-lived therapeutic regulatory T cells. Am J Transplant 16:58–71. https://doi.org/10.1111/ajt.13456
Dijke E, Hoeppli R, Larsen I et al (2017) OR42 expanded thymic regulatory T cells (Tregs) have low class I HLA expression levels and are HLA-DR negative – potential for ‘off-the-shelf’ cellular therapy? Hum Immunol 78:40. https://doi.org/10.1016/j.humimm.2017.06.048
F M, M M, N S et al (2006) Peripheral CD4+ CD25+ Treg cell expansion in lung transplant recipients is not affected by calcineurin inhibitors. Int Immunopharmacol 6:2002–2010. https://doi.org/10.1016/j.intimp.2006.07.019
Fallarino F, Grohmann U, Hwang KW et al (2003) Modulation of tryptophan catabolism by regulatory T cells. Nat Immunol 4:1206–1212. https://doi.org/10.1038/ni1003
Fleming BD, Mosser DM (2011) Regulatory macrophages: setting the threshold for therapy. Eur J Immunol 41:2498–2502. https://doi.org/10.1002/eji.201141717
Fontenot JD, Gavin MA, Rudensky AY (2003) Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol 4:330–336. https://doi.org/10.1038/ni904
Fujio K, Okamoto A, Araki Y et al (2006) Gene therapy of arthritis with TCR isolated from the inflamed paw. J Immunol 177:8140–8147. https://doi.org/10.4049/jimmunol.177.11.8140
Furukawa A, Wisel SA, Tang Q (2016) Impact of immune-modulatory drugs on Treg. Transplantation 100:2288–2300. https://doi.org/10.1097/TP.0000000000001379
Georgiev P, Charbonnier L-M, Chatila TA (2019) Regulatory T cells: the many faces of Foxp3. J Clin Immunol 39(7):623–640. https://doi.org/10.1007/s10875-019-00684-7
Gershon RK, Kondo K (1971) Infectious immunological tolerance. Immunology 21:903–914
Golshayan D, Jiang S, Tsang J et al (2007) In vitro–expanded donor alloantigen–specific CD4+CD25+ regulatory T cells promote experimental transplantation tolerance. Blood 109:827–835. https://doi.org/10.1182/blood-2006-05-025460
Gondek DC, Lu L-F, Quezada SA et al (2005) Cutting edge: contact-mediated suppression by CD4+CD25+ regulatory cells involves a Granzyme B-dependent, perforin-independent mechanism. J Immunol 174:1783–1786. https://doi.org/10.4049/jimmunol.174.4.1783
Gondek DC, DeVries V, Nowak EC et al (2008) Transplantation survival is maintained by Granzyme B+ regulatory cells and adaptive regulatory T cells. J Immunol 181:4752–4760. https://doi.org/10.4049/jimmunol.181.7.4752
Graca L, Cobbold SP, Waldmann H (2002) Identification of regulatory T cells in tolerated allografts. J Exp Med 195:1641–1646. https://doi.org/10.1084/jem.20012097
Grossman WJ, Verbsky JW, Barchet W et al (2004) Human T regulatory cells can use the perforin pathway to cause autologous target cell death. Immunity 21:589–601. https://doi.org/10.1016/j.immuni.2004.09.002
Gurkan S, Luan Y, Dhillon N et al (2010) Immune reconstitution following rabbit antithymocyte globulin. Am J Transplant 10:2132–2141. https://doi.org/10.1111/j.1600-6143.2010.03210.x
Hara M, Kingsley CI, Niimi M et al (2001) IL-10 is required for regulatory T cells to mediate tolerance to alloantigens in vivo. J Immunol 166:3789–3796. https://doi.org/10.4049/jimmunol.166.6.3789
Hasegawa H, Matsumoto T (2018) Mechanisms of tolerance induction by dendritic cells in vivo. Front Immunol 9. https://doi.org/10.3389/fimmu.2018.00350
Hermann-Kleiter N, Baier G (2010) NFAT pulls the strings during CD4+ T helper cell effector functions. Blood 115:2989–2997. https://doi.org/10.1182/blood-2009-10-233585
Hippen KL, Merkel SC, Schirm DK et al (2011a) Generation and large-scale expansion of human inducible regulatory T cells that suppress graft-versus-host disease. Am J Transplant 11:1148–1157. https://doi.org/10.1111/j.1600-6143.2011.03558.x
Hippen KL, Merkel SC, Schirm DK et al (2011b) Massive ex vivo expansion of human natural regulatory T cells (T(regs)) with minimal loss of in vivo functional activity. Sci Transl Med 3:83ra41. https://doi.org/10.1126/scitranslmed.3001809
Hippen KL, Riley JL, June CH, Blazar BR (2011c) Clinical perspectives for regulatory T cells in transplantation tolerance. Semin Immunol 23:462–468. https://doi.org/10.1016/j.smim.2011.07.008
Hoffmann P, Eder R, Kunz-Schughart LA et al (2004) Large-scale in vitro expansion of polyclonal human CD4(+)CD25high regulatory T cells. Blood 104:895–903. https://doi.org/10.1182/blood-2004-01-0086
Hoffmann P, Boeld TJ, Eder R et al (2006a) Isolation of CD4+CD25+ regulatory T cells for clinical trials. Biol Blood Marrow Transplant 12:267–274. https://doi.org/10.1016/j.bbmt.2006.01.005
Hoffmann P, Eder R, Boeld TJ et al (2006b) Only the CD45RA+ subpopulation of CD4+CD25high T cells gives rise to homogeneous regulatory T-cell lines upon in vitro expansion. Blood 108:4260–4267. https://doi.org/10.1182/blood-2006-06-027409
Hoffmann P, Boeld TJ, Eder R et al (2009) Loss of FOXP3 expression in natural human CD4+CD25+ regulatory T cells upon repetitive in vitro stimulation. Eur J Immunol 39:1088–1097. https://doi.org/10.1002/eji.200838904
van den Hoogen MWF, Kamburova EG, Baas MC et al (2015) Rituximab as induction therapy after renal transplantation: a randomized, double-blind, placebo-controlled study of efficacy and safety. Am J Transplant 15:407–416. https://doi.org/10.1111/ajt.13052
Hori S, Nomura T, Sakaguchi S (2003) Control of regulatory T cell development by the transcription factor Foxp3. Science 299:1057–1061. https://doi.org/10.1126/science.1079490
Issa F, Hester J, Goto R et al (2010) Ex vivo-expanded human regulatory T cells prevent the rejection of skin allografts in a humanised mouse model. Transplantation 90:1321–1327. https://doi.org/10.1097/TP.0b013e3181ff8772
Jb C, C S, A V et al (2015) Developing in vitro expanded CD45RA+ regulatory T cells as an adoptive cell therapy for Crohn’s disease. Gut 65:584–594. https://doi.org/10.1136/gutjnl-2014-306919
Joffre O, Santolaria T, Calise D et al (2008) Prevention of acute and chronic allograft rejection with CD4+CD25+Foxp3+ regulatory T lymphocytes. Nat Med 14:88–92. https://doi.org/10.1038/nm1688
Jonuleit H, Schmitt E, Kakirman H et al (2002) Infectious tolerance: human CD25(+) regulatory T cells convey suppressor activity to conventional CD4(+) T helper cells. J Exp Med 196:255–260. https://doi.org/10.1084/jem.20020394
Josefowicz SZ, Lu L-F, Rudensky AY (2012) Regulatory T cells: mechanisms of differentiation and function. Annu Rev Immunol 30:531–564. https://doi.org/10.1146/annurev.immunol.25.022106.141623
Kingsley CI, Karim M, Bushell AR, Wood KJ (2002) CD25+CD4+ regulatory T cells prevent graft rejection: CTLA-4- and IL-10-dependent immunoregulation of alloresponses. J Immunol 168:1080–1086. https://doi.org/10.4049/jimmunol.168.3.1080
Kwun J, Knechtle SJ (2009) Overcoming chronic rejection-can it B? Transplantation 88:955–961. https://doi.org/10.1097/TP.0b013e3181b96646
Landwehr-Kenzel S, Issa F, Luu S-H et al (2014) Novel GMP-compatible protocol employing an allogeneic B cell bank for clonal expansion of allospecific natural regulatory T cells. Am J Transplant 14:594–606. https://doi.org/10.1111/ajt.12629
Li Y, Koshiba T, Yoshizawa A et al (2004) Analyses of peripheral blood mononuclear cells in operational tolerance after pediatric living donor liver transplantation. Am J Transplant 4:2118–2125. https://doi.org/10.1111/j.1600-6143.2004.00611.x
Li W, Kuhr CS, Zheng XX et al (2008) New insights into mechanisms of spontaneous liver transplant tolerance: the role of Foxp3-expressing CD25+CD4+ regulatory T cells. Am J Transplant 8:1639–1651. https://doi.org/10.1111/j.1600-6143.2008.02300.x
Lin S-J, Lu C-H, Yan D-C et al (2014) Expansion of regulatory T cells from umbilical cord blood and adult peripheral blood CD4(+)CD25 (+) T cells. Immunol Res 60:105–111. https://doi.org/10.1007/s12026-014-8488-1
Liu W, Putnam AL, Xu-yu Z et al (2006) CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J Exp Med 203:1701–1711. https://doi.org/10.1084/jem.20060772
Long E, Wood KJ (2009) Regulatory T cells in transplantation: transferring mouse studies to the clinic. Transplantation 88:1050–1056. https://doi.org/10.1097/TP.0b013e3181bb7913
Ma A, Qi S, Song L et al (2011) Adoptive transfer of CD4+CD25+ regulatory cells combined with low-dose sirolimus and anti-thymocyte globulin delays acute rejection of renal allografts in Cynomolgus monkeys. Int Immunopharmacol 11:618–629. https://doi.org/10.1016/j.intimp.2010.11.001
MacDonald KG, Hoeppli RE, Huang Q et al (2016) Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor. J Clin Invest 126:1413–1424. https://doi.org/10.1172/JCI82771
Macian F (2005) NFAT proteins: key regulators of T-cell development and function. Nat Rev Immunol 5:472–484. https://doi.org/10.1038/nri1632
Marín E, Cuturi MC, Moreau A (2018) Tolerogenic dendritic cells in solid organ transplantation: where do we stand? Front Immunol 9. https://doi.org/10.3389/fimmu.2018.00274
Maude SL, Frey N, Shaw PA et al (2014) Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med 371:1507–1517. https://doi.org/10.1056/NEJMoa1407222
Min W-P, Zhou D, Ichim TE et al (2003) Inhibitory feedback loop between tolerogenic dendritic cells and regulatory T cells in transplant tolerance. J Immunol 170:1304–1312. https://doi.org/10.4049/jimmunol.170.3.1304
Miroux C, Morales O, Ghazal K et al (2012) In vitro effects of cyclosporine A and tacrolimus on regulatory T-cell proliferation and function. Transplantation 94:123–131. https://doi.org/10.1097/TP.0b013e3182590d8f
Miyara M, Yoshioka Y, Kitoh A et al (2009) Functional delineation and differentiation dynamics of human CD4+ T cells expressing the FoxP3 transcription factor. Immunity 30:899–911. https://doi.org/10.1016/j.immuni.2009.03.019
Nadig SN, Wieckiewicz J, Wu DC et al (2010) In vivo prevention of transplant arteriosclerosis by ex vivo-expanded human regulatory T cells. Nat Med 16:809–813. https://doi.org/10.1038/nm.2154
Nishimura E, Sakihama T, Setoguchi R et al (2004) Induction of antigen-specific immunologic tolerance by in vivo and in vitro antigen-specific expansion of naturally arising Foxp3+CD25+CD4+ regulatory T cells. Int Immunol 16:1189–1201. https://doi.org/10.1093/intimm/dxh122
Noyan F, Zimmermann K, Hardtke-Wolenski M et al (2017) Prevention of allograft rejection by use of regulatory T cells with an MHC-specific chimeric antigen receptor. Am J Transplant 17:917–930. https://doi.org/10.1111/ajt.14175
Ochando J, Ordikhani F, Jordan S et al (2019) Tolerogenic dendritic cells in organ transplantation. Transpl Int 33:113. https://doi.org/10.1111/tri.13504
Ojo AO, Held PJ, Port FK et al (2003) Chronic renal failure after transplantation of a nonrenal organ. N Engl J Med 349:931–940. https://doi.org/10.1056/NEJMoa021744
Okada M, Kanamori M, Someya K et al (2017) Stabilization of Foxp3 expression by CRISPR-dCas9-based epigenome editing in mouse primary T cells. Epigenetics Chromatin 10:24. https://doi.org/10.1186/s13072-017-0129-1
Papp G, Boros P, Nakken B et al (2017) Regulatory immune cells and functions in autoimmunity and transplantation immunology. Autoimmun Rev 16:435–444. https://doi.org/10.1016/j.autrev.2017.03.011
Polansky JK, Kretschmer K, Freyer J et al (2008) DNA methylation controls Foxp3 gene expression. Eur J Immunol 38:1654–1663. https://doi.org/10.1002/eji.200838105
Porter DL, Levine BL, Kalos M et al (2011) Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. N Engl J Med 365:725–733. https://doi.org/10.1056/NEJMoa1103849
Putnam AL, Brusko TM, Lee MR et al (2009) Expansion of human regulatory T-cells from patients with type 1 diabetes. Diabetes 58:652–662. https://doi.org/10.2337/db08-1168
Putnam AL, Safinia N, Medvec A et al (2013) Clinical grade manufacturing of human alloantigen-reactive regulatory T cells for use in transplantation. Am J Transplant 13:3010–3020. https://doi.org/10.1111/ajt.12433
Qureshi OS, Zheng Y, Nakamura K et al (2011) Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4. Science 332:600–603. https://doi.org/10.1126/science.1202947
Raffin C, Vo LT, Bluestone JA (2020) Treg cell-based therapies: challenges and perspectives. Nat Rev Immunol 20:158–172. https://doi.org/10.1038/s41577-019-0232-6
Riley JL, June CH, Blazar BR (2009) Human T regulatory cell therapy: take a billion or so and call me in the morning. Immunity 30:656–665. https://doi.org/10.1016/j.immuni.2009.04.006
Roncarolo MG, Gregori S, Bacchetta R, Battaglia M (2014) Tr1 cells and the counter-regulation of immunity: natural mechanisms and therapeutic applications. Curr Top Microbiol Immunol 380:39–68. https://doi.org/10.1007/978-3-662-43492-5_3
Rosen SJ, Harris PE, Hardy MA (2018) State of the art: role of the dendritic cell in induction of allograft tolerance. Transplantation 102:1603–1613. https://doi.org/10.1097/TP.0000000000002239
Sakaguchi S, Sakaguchi N, Asano M et al (1995) Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol 155:1151–1164
Sakaguchi S, Wing K, Onishi Y et al (2009) Regulatory T cells: how do they suppress immune responses? Int Immunol 21:1105–1111. https://doi.org/10.1093/intimm/dxp095
Sakaguchi S, Miyara M, Costantino CM, Hafler DA (2010) FOXP3+ regulatory T cells in the human immune system. Nat Rev Immunol 10:490–500. https://doi.org/10.1038/nri2785
San Segundo D, Fábrega E, López-Hoyos M, Pons F (2007) Reduced numbers of blood natural regulatory T cells in stable liver transplant recipients with high levels of calcineurin inhibitors. Transplant Proc 39:2290–2292. https://doi.org/10.1016/j.transproceed.2007.07.076
Schmidt A, Oberle N, Krammer PH (2012) Molecular mechanisms of Treg-mediated T cell suppression. Front Immunol 3. https://doi.org/10.3389/fimmu.2012.00051
Scottà C, Esposito M, Fazekasova H et al (2013) Differential effects of rapamycin and retinoic acid on expansion, stability and suppressive qualities of human CD4(+)CD25(+)FOXP3(+) T regulatory cell subpopulations. Haematologica 98:1291–1299. https://doi.org/10.3324/haematol.2012.074088
Seay HR, Putnam AL, Cserny J et al (2017) Expansion of human Tregs from cryopreserved umbilical cord blood for GMP-compliant autologous adoptive cell transfer therapy. Mol Ther Methods Clin Dev 4:178–191. https://doi.org/10.1016/j.omtm.2016.12.003
Seddiki N, Santner-Nanan B, Martinson J et al (2006) Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J Exp Med 203:1693–1700. https://doi.org/10.1084/jem.20060468
Segundo DS, Ruiz JC, Izquierdo M et al (2006) Calcineurin inhibitors, but not rapamycin, reduce percentages of CD4+CD25+FOXP3+ regulatory T cells in renal transplant recipients. Transplantation 82:550–557. https://doi.org/10.1097/01.tp.0000229473.95202.50
Shalev I, Schmelzle M, Robson SC, Levy G (2011) Making sense of regulatory T cell suppressive function. Semin Immunol 23:282–292. https://doi.org/10.1016/j.smim.2011.04.003
Shevach EM, Thornton AM (2014) tTregs, pTregs, and iTregs: similarities and differences. Immunol Rev 259:88–102. https://doi.org/10.1111/imr.12160
Shimony O, Nagler A, Gellman YN et al (2012) Anti-T lymphocyte globulin (ATG) induces generation of regulatory T cells, at least part of them express activated CD44. J Clin Immunol 32:173–188. https://doi.org/10.1007/s10875-011-9599-2
Shultz LD, Ishikawa F, Greiner DL (2007) Humanized mice in translational biomedical research. Nat Rev Immunol 7:118–130. https://doi.org/10.1038/nri2017
Sicard A, Koenig A, Morelon E et al (2015) Cell therapy to induce allograft tolerance: time to switch to plan B? Front Immunol 6. https://doi.org/10.3389/fimmu.2015.00149
Stephens LA, Malpass KH, Anderton SM (2009) Curing CNS autoimmune disease with myelin-reactive Foxp3+ Treg. Eur J Immunol 39:1108–1117. https://doi.org/10.1002/eji.200839073
Strauss L, Whiteside TL, Knights A et al (2007) Selective survival of naturally occurring human CD4+CD25+Foxp3+ regulatory T cells cultured with rapamycin. J Immunol 178:320–329. https://doi.org/10.4049/jimmunol.178.1.320
Streitz M, Miloud T, Kapinsky M et al (2013) Standardization of whole blood immune phenotype monitoring for clinical trials: panels and methods from the ONE study. Transplant Res 2:17. https://doi.org/10.1186/2047-1440-2-17
Tang Q, Lee K (2012) Regulatory T-cell therapy for transplantation: how many cells do we need? Curr Opin Organ Transplant 17:349–354. https://doi.org/10.1097/MOT.0b013e328355a992
Tang Q, Henriksen KJ, Bi M et al (2004) In vitro-expanded antigen-specific regulatory T cells suppress autoimmune diabetes. J Exp Med 199:1455–1465. https://doi.org/10.1084/jem.20040139
Tarbell KV, Petit L, Zuo X et al (2007) Dendritic cell–expanded, islet-specific CD4+ CD25+ CD62L+ regulatory T cells restore normoglycemia in diabetic NOD mice. J Exp Med 204:191–201. https://doi.org/10.1084/jem.20061631
Tey S-K (2014) Adoptive T-cell therapy: adverse events and safety switches. Clin Transl Immunol 3:e17. https://doi.org/10.1038/cti.2014.11
Thomson AW, Turnquist HR, Raimondi G (2009) Immunoregulatory functions of mTOR inhibition. Nat Rev Immunol 9:324–337. https://doi.org/10.1038/nri2546
Thomson AW, Metes DM, Ezzelarab MB, Raïch-Regué D (2019) Regulatory dendritic cells for human organ transplantation. Transplant Rev 33:130–136. https://doi.org/10.1016/j.trre.2019.05.001
Thornton AM, Shevach EM (2000) Suppressor effector function of CD4+CD25+ immunoregulatory T cells is antigen nonspecific. J Immunol 164:183–190. https://doi.org/10.4049/jimmunol.164.1.183
Todo S, Yamashita K, Goto R et al (2016) A pilot study of operational tolerance with a regulatory T-cell-based cell therapy in living donor liver transplantation. Hepatology 64:632–643. https://doi.org/10.1002/hep.28459
Tresoldi E, Dell’Albani I, Stabilini A et al (2011) Stability of human rapamycin-expanded CD4+CD25+ T regulatory cells. Haematologica 96:1357–1365. https://doi.org/10.3324/haematol.2011.041483
Trzonkowski P, Bieniaszewska M, Juścińska J et al (2009) First-in-man clinical results of the treatment of patients with graft versus host disease with human ex vivo expanded CD4+CD25+CD127- T regulatory cells. Clin Immunol 133:22–26. https://doi.org/10.1016/j.clim.2009.06.001
Tsang JY-S, Tanriver Y, Jiang S et al (2008) Conferring indirect allospecificity on CD4+CD25+ Tregs by TCR gene transfer favors transplantation tolerance in mice. J Clin Invest 118:3619–3628. https://doi.org/10.1172/JCI33185
Valenzuela NM, Reed EF (2017) Antibody-mediated rejection across solid organ transplants: manifestations, mechanisms, and therapies. J Clin Invest 127:2492–2504. https://doi.org/10.1172/JCI90597
Venken K, Thewissen M, Hellings N et al (2007) A CFSE based assay for measuring CD4+CD25+ regulatory T cell mediated suppression of auto-antigen specific and polyclonal T cell responses. J Immunol Methods 322:1–11. https://doi.org/10.1016/j.jim.2007.01.025
Watanabe T, Masuyama J, Sohma Y et al (2006) CD52 is a novel costimulatory molecule for induction of CD4+ regulatory T cells. Clin Immunol 120:247–259. https://doi.org/10.1016/j.clim.2006.05.006
Watt KDS, Pedersen RA, Kremers WK et al (2010) Evolution of causes and risk factors for mortality post-liver transplant: results of the NIDDK long-term follow-up study. Am J Transplant 10:1420–1427. https://doi.org/10.1111/j.1600-6143.2010.03126.x
Wing K, Sakaguchi S (2010) Regulatory T cells exert checks and balances on self tolerance and autoimmunity. Nat Immunol 11:7–13. https://doi.org/10.1038/ni.1818
Wood KJ, Goto R (2012) Mechanisms of rejection: current perspectives. Transplantation 93:1–10. https://doi.org/10.1097/TP.0b013e31823cab44
Wood KJ, Bushell A, Hester J (2012) Regulatory immune cells in transplantation. Nat Rev Immunol 12:417–430. https://doi.org/10.1038/nri3227
Wu DC, Hester J, Nadig SN et al (2013) Ex vivo expanded human regulatory T cells can prolong survival of a human islet allograft in a humanized mouse model. Transplantation 96:707–716. https://doi.org/10.1097/TP.0b013e31829fa271
Xia G, He J, Leventhal JR (2008) Ex vivo-expanded natural CD4+CD25+ regulatory T cells synergize with host T-cell depletion to promote long-term survival of allografts. Am J Transplant 8:298–306. https://doi.org/10.1111/j.1600-6143.2007.02088.x
Zeiser R, Leveson-Gower DB, Zambricki EA et al (2008) Differential impact of mammalian target of rapamycin inhibition on CD4+CD25+Foxp3+ regulatory T cells compared with conventional CD4+ T cells. Blood 111:453–462. https://doi.org/10.1182/blood-2007-06-094482
Zhou P, Borojevic R, Streutker C et al (2004) Expression of dual TCR on DO11.10 T cells allows for ovalbumin-induced oral tolerance to prevent T cell-mediated colitis directed against unrelated enteric bacterial antigens. J Immunol 172:1515–1523. https://doi.org/10.4049/jimmunol.172.3.1515
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Que, W., Li, XK. (2021). Regulatory T Cells for the Induction of Transplantation Tolerance. In: Zheng, SG. (eds) T Regulatory Cells in Human Health and Diseases. Advances in Experimental Medicine and Biology, vol 1278. Springer, Singapore. https://doi.org/10.1007/978-981-15-6407-9_15
Download citation
DOI: https://doi.org/10.1007/978-981-15-6407-9_15
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-6406-2
Online ISBN: 978-981-15-6407-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)