Abstract
Thirteen Acer species were found in Ooyamazawa riparian forest in Japan. Of these, the shrubby species A. carpinifolium and A. argutum and the tall tree species A. shirasawanum and A. mono were the most abundant. Sprouts of shrubby species were common, but not of tall tree species, which implies that individuals of shrubby species are maintained in the understory layer via sprouts, whereas tall tree species extend upward toward the canopy. All four Acer species were found mainly on upstream sediments; moreover, the density of A. shirasawanum was high, implying that A. shirasawanum may eventually become an important species in the sedimentary upstream area. On the other hand, A. carpinifolium was dominant in the unstable downstream V-shaped valley; this species adapts to the disturbed downstream area by producing more sprouts. These differences in life history promote diversity in the forest structure of Ooyamazawa riparian forest.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Acer argutum
- Acer carpinifolium
- Acer pictim
- Acer shirasawanum
- Distribution
- Riparian forest
- Sprouting traits
1 Introduction
The genus Acer is comprised of tree species that play important roles in forests across the Northern Hemisphere, particularly in late-successional forests dominated by hardwoods such as beech (Fagus) species (Runkle 1990; Poulson and Platt 1996; Cao and Ohkubo 1999). Acer species often coexist with other deciduous species, depending on light conditions and/or disturbance regimes (Sipe and Bazzaz 1995). Some Acer species are found in riparian forests (Ohno 2008), in topographical habitats created by various types of disturbance (Masaki et al. 1992, 2005; Suzuki et al. 2002), suggesting that some Acer species may exhibit pioneer traits.
In Japan, 28 Acer tree species are distributed throughout sub-tropical, warm-temperate, cool-temperate, and sub-alpine forests (Yonekura 2012). Many trees in riparian forests of the cool-temperate zone in Japan are Acer species (Ohno 2008). Acer is therefore an important taxonomic group that contributes to tree species diversity and forest structure in these riparian forests. Acer species typically coexist with other tree species due to their different life histories (Sakio et al. 2002), and represent both early successional species, in response to various riparian disturbances, and late-successional species, which play important roles in riparian forest dynamics.
Acer carpinifolium Siebold et Zucc., Acer shirasawanum Koidz., Acer pictum Thunb., Acer argutum Maxim., Acer nipponicum H.Hara, Acer rufinerve Siebold et Zucc., Acer tenuifolium (Koidz.) Koidz., Acer palmatum Thunb., Acer amoenum Carrière var. amoenum, Acer maximowiczianum Miq., Acer cissifolium (Siebold et Zucc.) K.Koch, Acer distylum Siebold et Zucc., and Acer micranthum Siebold et Zucc. all grow in the Ooyamazawa riparian area of central Japan, and the four dominant Acer species are Acer carpinifolium, A. shirasawanum, A. pictim, and A. argutum. In this chapter, we clarify the life histories of these four major Acer species and discuss the relationship between their life histories and riparian topography in Ooyamazawa.
2 Study Species
A. carpinifolium, A. shirasawanum, A. pictim, and A. argutum are indigenous to Japan. A. carpinifolium and A. pictim are distributed in Honshu, Shikoku, and Kyusyu Islands, and A. shirasawanum and A. argutum are distributed in Honshu and Shikoku Islands. A. carpinifolium and A. argutum generally grow to shrub height (Table 5.1, Fig. 5.1). These four Acer species are deciduous and monophyllous; A. carpinifolium has oval leaves, whereas the remaining three Acer species have palmate leaves.
A. carpinifolium is a dioecious shrub that grows to a height of approximately 10 m and produces shoots around stems. This species is mainly distributed in riparian areas in the mountains, flowering in May and producing seeds during September–October. A. shirasawanum is a monoecious tree that grows to a height of about 20 m and diameter at breast height (DBH) of 30–40 cm, with larger individuals reaching a DBH of 80 cm. A. shirasawanum is mainly found in mountain areas; it flowers in May and produces seeds during September–October. A. pictim is a monoecious tree that grows to a height of 20 m and DBH of 50–60 cm; it is found in riparian and mountain areas, flowers in May, and produces seeds during September–October. A. argutum is a dioecious shrub that grows to a height of about 8 m and produces shoots close to the root system; it is found in riparian and mountain areas, flowers in May, and produces seeds during September–October.
3 Structure and Distribution
3.1 Abundance and Structure of Four Acer Species
In this study, we identified 1204 individual trees (255.4 trees/ha) of 13 Acer species in a 4.71-ha plot within the Ooyamazawa riparian forest (Table 5.2, Fig. 5.2). Thus, Ooyamazawa contains roughly half of the 28 Acer species found in Japan (Yonekura 2012). Acer species comprised 28.9% of a total of 45 tree species and 57.0% of a total of 2111 individuals (448.1/ha) in the study area. Together, A. carpinifolium, A. shirasawanum, A. pictum, and A. argutum comprised 96.3% of all Acer individuals and 55.0% of all individual trees.
Acer species found in the Ooyamazawa riparian site. (a) A. carpinifolium. (b) A. shirasawanum. (c) A. pictim. (d) A. argutum. (e) A. nipponicum. (f) A. rufinerve. (g) A. tenuifolium. (h) A. palmatum. (i) A. amoenum var. amoenum. (j) A. maximowiczianum. (k) A. cissifolium (photo by Takuto Shitara). (l) A. distylum. (m) A. micranthum
Acer species densities were particularly high in the subcanopy and shrub layers (Table 5.2, Fig. 5.3). A. carpinifolium and A. argutum were found in both the subcanopy and shrub layers; however, the vast majority of individuals were found in the shrub layer. In contrast, A. shirasawanum and A. pictum were found in all layers, with most individuals in the subcanopy and shrub layers.
Small Acer individuals (DBH < 40 cm) were numerous; most individual A. carpinifolium and A. argutum shrubs did not exceed a DBH of 20 cm (Fig. 5.4). A. pictum had the largest DBH at 92.0 cm, followed by A. shirasawanum at 62.8 cm (Table 5.2).
3.2 Spatial Distribution of Four Acer Species
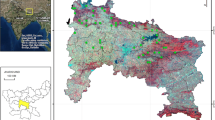
The four Acer species were mainly distributed on upstream sedimentary debris flows ; among these, only A. carpinifolium was also dominant in the downstream V-shaped valley (Fig. 5.5). We divided the study plot into 20 subplots along the stream and compared the densities of the four Acer species and the dominant canopy tree Fraxinus platypoda in the canopy, subcanopy, and shrub layers in each subplot (Fig. 5.6). In the subcanopy layer, A. carpinifolium density was higher in the downstream valley (Wilcoxon rank sum test, P < 0.01), although in the shrub layer A. carpinifolium density was high both upstream and downstream (Wilcoxon rank sum test, P = 0.16). In the subcanopy layer, A. argutum density was low, while in the shrub layer, A. argutum density was significantly higher upstream than downstream in the valley (Wilcoxon rank sum test, P < 0.01). In both the subcanopy and shrub layers, A. shirasawanum density was higher upstream (Wilcoxon rank-sum test, P < 0.01), as was A. pictum density in the shrub layer (Wilcoxon rank sum test, P < 0.01), although A. pictum densities in the canopy and subcanopy layers did not differ significantly between upstream and downstream areas (Wilcoxon rank sum test, canopy layer; P = 0.28, subcanopy layer; P = 0.12).
Density of the four most common Acer species and Fraxinus platypoda. The study plot was divided into 20 subplots at 60-m intervals along the stream, with eight subplots (1–8) in the downstream V-shaped valley and 12 subplots (9–20) in the upstream sedimentary debris flow. Upper, tree density in the canopy layer; middle, tree density in the subcanopy layer; lower, tree density in the shrub layer
The sedimentary upstream area was the best habitat for three of the four Acer species, whereas A. carpinifolium was better suited to the downstream valley (Fig. 5.5). The upstream alluvial fan and terrace debris flows contain rich soil and a considerable litter layer; in contrast, little litter is found on terrace scarps, new landslide sites, old landslide slopes, or talus of the downstream V-shaped valley (Kawanishi et al. 2004). Downstream disturbances including erosion and sedimentation of soil, sand, and/or gravel are frequent due to stream flow and/or steep slopes. Fewer disturbances occur upstream where slopes are gentle (about 12°) and the valley bottom has been filled by large landslides and/or debris flows.
Factors determining the distribution patterns of Acer species can include shade tolerance, the distance from the seed source, and germination site conditions, e.g., areas of soil and litter accumulation. Leaf litter cover also reduces the risk of predation on Acer seeds (Tanaka 1995). Although A. pictum was found in the canopy and subcanopy layers both upstream and downstream, A. shirasawanum clearly occurs in the subcanopy and shrub layers only upstream, where it is dominant (Figs. 5.5 and 5.6). A. pictum saplings can survive even in the forest understory (Abe et al. 1995; Hara 1987; Masaki et al. 1992) by acclimating to deep shade (Kitao et al. 2006). A. shirasawanum can regenerate in smaller gaps than A. pictum, due to its shade tolerance (Sakai 1986). It remains unclear why the range of A. shirasawanum does not extend downstream, since its shade tolerance should allow it to dominate the more stable upstream sediments, where soils are rich, litter accumulation is greater, and the upland forest is a nearby seed source. For these reasons, A. shirasawanum may eventually become an important species in the sedimentary upstream area.
4 Sprouting Traits
The two Acer shrub species, A. carpinifolium and A. argutum, produced large numbers of shoots (Fig. 5.7). Among Acer species, the greatest mean number of shoots was observed in A. carpinifolium (6.0 ± 4.3), exceeded only by Cercidiphyllum japonicum (9.0 ± 12.1) in the Ooyamazawa study site (Chap. 4). A. shirasawanum and A. pictum, both of which are tall tree species, had fewer shoots, suggesting that A. carpinifolium and A. argutum may reproduce in the shrub layer via sprouting.
Shrub species tend to produce many shoots (Midgley 1996); reproduction via sprouting provides an advantage in habitats where environmental conditions are severe (Sakai et al. 1995; Kubo et al. 2010). The low light conditions in the shrub layer prevent A. carpinifolium and A. argutum from consistently receiving direct light. Main shoots tend to die when they grow too large to balance photosynthesis and respiration, allowing a large shoot with large leaf area to become a new main shoot.
The large numbers of shoots observed in the shrub A. carpinifolium are therefore suitable for habitats with significant surface erosion, allowing long-term survival of some individuals in the subcanopy layer within the downstream V-shaped valley, where the density of other Acer species is low (Figs. 5.5 and 5.6). Similarly, Euptelea polyandra has shoots adapted to its unstable habitat conditions (Sakai et al. 1995), with main shoots gradually inclining as they increase in size, facilitating the establishment of younger shoots. The large number of shoots and dominance of A. carpinifolium in the unstable V-shaped valley suggest that this species has sprouting traits adapted to the unstable steep slopes and disturbance regime of this site.
Some species reproduce via spontaneous sprouting (Verwijst 1988; Keeley 1992). In this study, almost all A. argutum were found on stable upstream sediments. Many A. argutum shoots are produced near the soil around the main stem, like root suckers, whereas A. carpinifolium, which was dominant in the unstable V-shaped valley, sprouts from the shoot base. Some tree species extend their roots to the surface to produce new shoots (Gyokusen et al. 1991; Ogawa et al. 1999; Sakio 2015). Therefore A. argutum reproduces through sprouting in the shrub layer on stable upstream sediments, where soil is rich, and its range does not extend to the V-shaped valley, where slopes are steep and soil is poor.
5 Conclusion
In this study, we identified 13 Acer tree species in a 4.71-ha study plot in the Ooyamazawa riparian forest. Among these, four species, A. carpinifolium, A. shirasawanum, A. pictum, and A. argutum constituted 55.0% of all individuals. These four coexisting Acer species had different life history characteristics in terms of size, distribution, and shoot production. A. shirasawanum and A. pictum are tall tree species, whereas A. carpinifolium and A. argutum are shrub species (Table 5.2, Fig. 5.3), which produce more shoots than the taller species (Fig. 5.7). All four Acer species were mainly found on upstream sediments. However, A. shirasawanum and A. argutum were distributed only on stable upstream sediments, A. pictum was distributed in the canopy and subcanopy layers in both areas, and A. carpinifolium was dominant in the unstable downstream V-shaped valley (Figs. 5.5 and 5.6). The two shrub species have different sprouting traits, with A. carpinifolium adapting to the disturbed downstream area by producing more shoots and A. argutum producing a lot of shoots upstream, where the soil is rich and disturbance occurs less frequently. These differences in life history promote diversity in forest structure in this riparian forest.
References
Abe S, Masaki T, Nakashizuka T (1995) Factors influencing sapling composition in canopy gaps of a temperate deciduous forest. Vegetatio 120:21–32
Cao K-F, Ohkubo T (1999) Suppression and release during canopy recruitment in Fagus crenata and Acer mono in two old-growth beech forests in Japan. Plant Ecol 145:281–290
Gyokusen K, Iijima Y, Yahata H (1991) Spacial distribution and morphological features of root sprouts in Niseakasia (Robinia pseudo-acasia L.) growing under a coastal black pine forest. Bull Kyushu Univ For 64:13–28 (Japanese with English summary)
Hara M (1987) Analysis of seedling banks of a climax beech forest: ecological importance of seedling sprouts. Vegetatio 71:67–74
Kawanishi M, Sakio H, Ohno K (2004) Forest floor vegetation of Fraxinus platypoda—Pterocarya rhoifolia forest along Ooyamazawa valley in Chichibu, Kanto district, Japan, with a special reference to ground disturbance. Veg Sci 21:15–26. (In Japanese with English abstract)
Keeley JE (1992) Recruitment of seedlings and vegetative sprouts in unburned chaparral. Ecology 73:1194–1208
Kitao M, Lei TT, Koike T, Tobita H, Maruyama Y (2006) Tradeoff between shade adaptation and mitigation of photoinhibition in leaves of Quercus mongolica and Acer mono acclimated to deep shade. Tree Physio 26:441–448
Kubo M, Shimano K, Sakio H, Isagi Y, Ohno K (2010) Difference between sprouting traits of Cercidiphyllum japonicum and C. magnificum. J For Res 15:337–340
Masaki T, Osumi K, Takahashi K, Hoshizaki K (2005) Seedling dynamics of Acer mono and Fagus crenata: an environmental filter limiting their adult distributions. Plant Ecol 177:189–199
Masaki T, Suzuki W, Niiyama K, Iida S, Tanaka H, Nakashizuka T (1992) Community structure of a species-rich temperate forest, Ogawa Forest Reserve, central Japan. Vegetatio 98:97–111
Midgley JJ (1996) Why the world’s vegetation is not totally dominated by resprouting plants; because resprouters are shorter than reseeders. Ecography 19:92–95
Ogawa M, Aiba Y, Watanabe N (1999) Morphological and anatomical characteristics of sprouting root of Prunus ssiori. J Jpn For Soc 81:36–41
Ohno K (2008) Vegetation-geographic evaluation of the syntaxonomic system of valley-bottom forests occurring in the cool-temperate zone of the Japanese Archipelago. In: Sakio H, Tamura T (eds) Ecology of riparian forests in Japan. Springer, Tokyo, pp 49–72
Poulson TL, Platt WJ (1996) Replacement patterns of beech and sugar maple in warren woods, Michigan. Ecology 77:1234–1253
Runkle JR (1990) Gap dynamics in an Ohio Acer-Fagus forest and speculations on the geography of disturbance. Can J For Res 20:632–641
Sakai A, Ohsawa T, Ohsawa M (1995) Adaptive significance of sprouting of Euptelea polyandra, a deciduous tree growing on steep slopes with shallow soil. J Plant Res 108:377–386
Sakai S (1986) Patterns of branching and extension growth of vigorous saplings of Japanese Acer species in relation to their regeneration strategies. Can J Bot 65:1578–1585
Sakio H, Kubo M, Shimano K, Ohno K (2002) Coexistence of three canopy tree species in a riparian forest in the Chichibu mountains, central Japan. Folia Geobotanica 37:45–61
Sakio H (2015) Why did the black locust expand broadly at the river basin in Japan? Jpn Soc Reveg Technol 40(3):465–471 (Japanese with English abstract)
Sipe TW, Bazzaz FA (1995) Gap partitioning among maples (Acer) in central New England: survival and growth. Ecology 76:1587–1602
Suzuki W, Osumi K, Masaki T, Takahashi K, Daimaru H, Hoshizaki K (2002) Disturbance regimes and community structures of a riparian and an adjacent terrace stand in the Kanumazawa Riparian Research Forest, northern Japan. For Ecol Manage 157:285–301
Tanaka H (1995) Seed demography of three co-occurring Acer species in a Japanese temperate deciduous forest. J Veg Sci 6:887–896
Verwijst T (1988) Environmental correlates of multiple-stem formation in Betula pubescens ssp. tortuosa. Vegetatio 76:29–36
Yonekura K (2012) An enumeration of the vascular plants of Japan. Hokuryukan, Tokyo
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2020 The Author(s)
About this chapter
Cite this chapter
Kubo, M., Sakio, H., Kawanishi, M., Higa, M. (2020). Acer Tree Species. In: Sakio, H. (eds) Long-Term Ecosystem Changes in Riparian Forests. Ecological Research Monographs. Springer, Singapore. https://doi.org/10.1007/978-981-15-3009-8_5
Download citation
DOI: https://doi.org/10.1007/978-981-15-3009-8_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-3008-1
Online ISBN: 978-981-15-3009-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)