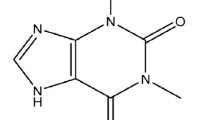
Summary
The acute oral and parenteral LD-50 of beta-cyclodextrin was established on rats, mice and dogs.
The long term toxicity studies were performed on rats and dogs following the two weeks dose finding studies given the test substance by oral route. All of the animals were autopsied and the tissue samples were examined histopathologically in both experiments.
Investigation of the effect of beta-cyclodextrin on hepatic drug metabolizing enzyme system of rats did not show any alteration in the activity.
According to our results the beta-cyclodextrin given orally to rats and dogs did not cause any side effect during the long term toxicity studies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1982 Springer Science+Business Media Dordrecht
About this paper
Cite this paper
Gergely, V., Sebestyén, G., Virág, S. (1982). Toxicity Studies of Beta-Cyclodextrin. In: Szejtli, J. (eds) Proceedings of the First International Symposium on Cyclodextrins. Advances in Inclusion Science, vol 1. Springer, Dordrecht. https://doi.org/10.1007/978-94-009-7855-3_12
Download citation
DOI: https://doi.org/10.1007/978-94-009-7855-3_12
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-009-7857-7
Online ISBN: 978-94-009-7855-3
eBook Packages: Springer Book Archive