Abstract
Four extra early-heading mutants, named extra early-flowering1 (exe1), exe2, exe3, and exe4, were identified in diploid einkorn wheat (Triticum monococcum L.) following heavy-ion beam mutagenesis. Based on their phenotypes in the field, the four exe mutants were classified into two groups: Type I (moderately extra early-heading type; exe1 and exe3) and Type II (extremely extra early-heading type; exe2 and exe4). Analysis of VERNALIZATION 1 (VRN1), a flowering promoter gene, showed that it was more highly expressed at earlier stages of vegetative growth in Type II mutants than in Type I mutants. Our analyses indicate that the difference in earliness between Type I and Type II mutants is associated with differences in the expression level of VRN1.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Introduction
Improving our understanding of the molecular mechanisms of flowering, the phase transition from vegetative to reproductive growth associated with heading time, is one of the most important goals for wheat breeding at the present time. In bread wheat (Triticum aestivum L.), heading time is genetically determined by three characteristics: vernalization requirement; photoperiod sensitivity; and narrow-sense earliness (earliness per se). Three genes have been identified to determine the requirement of vernalization, namely VERNALIZATION 1 (VRN1), VRN2 and VRN3.
VRN1 encodes an APETALA1/FRUITFULL-like MADS-box transcription factor that is up-regulated by vernalization (Yan et al. 2003; Murai et al. 2003; Trevaskis et al. 2003; Danyluk et al. 2003). Recent studies revealed that expression of VRN1 is epigenetically suppressed in seedlings at an earlier stage of the vegetative growth phase, and that the repressive histone state is modified by the vernalization signal, leading to the up-regulation of VRN1 (Oliver et al. 2009; Diallo et al. 2012). The level of VRN1 transcription gradually increases during the seedling growth stage without the need for further vernalization (Murai et al. 2003; Kitagawa et al. 2012), suggesting that the epigenetic status of VRN1 is also modified by aging. Furthermore, VRN1 shows a diurnal expression pattern that is affected by daylight, with a long photoperiod producing up-regulation of its expression level (Shimada et al. 2009). In summary, these observations indicate that the VRN1 expression is also controlled by autonomous and photoperiodic pathways, as well as the vernalization pathway.
The VRN2 locus consists of two linked ZCCT genes, ZCCT1 and ZCCT2, which encode a protein with a zinc finger motif and a CCT domain (Yan et al. 2004). Natural variations have been identified in the VRN2 locus. Simultaneous deletion or non-functional mutations of these two ZCCT genes result in a plant showing the spring habit (Distelfeld et al. 2009), indicating that VRN2 is a flowering repressor gene. A high level of VRN2 expression is observed in seedlings at the 1-leaf stage, while expression is down-regulated by vernalization and aging; by contrast, VRN1 shows the opposite pattern with low expression in seedlings and up-regulated expression after vernalization (Shimada et al. 2009). It has also been reported that VRN2 shows a diurnal expression pattern and that a long photoperiod up-regulates its expression level (Dubcovsky et al. 2006; Trevaskis et al. 2006), suggesting that the VRN2 expression is affected by photoperiod as well as vernalization.
VRN3 encodes a Raf kinase inhibitor-like protein with a high similarity to the Arabidopsis FLOWERING LOCUS T (FT) protein, which is a florigen (Yan et al. 2006). Transgenic wheat plants overexpressing VRN3 show an extra early-flowering phenotype without the need for vernalization (Yan et al. 2006; Shimada et al. 2009), indicating that VRN3 is a strong flowering promoter. Under long day conditions, VRN3 shows a diurnal expression pattern; however, expression is very low under short day conditions (Shimada et al. 2009; Kitagawa et al. 2012).
Based on data from expression, transgenic and mutant analyses, we developed a gene network model for the interaction of VRN1, VRN2 and VRN3 in leaves (Shimada et al. 2009). In this model, VRN1 is upstream of VRN3 and activates VRN3 expression under long day conditions. Thus, VRN1 is proposed to play a role as an integrator of the vernalization and photoperiodic signals. Trevaskis (2010) put forward an alternative gene network model for VRN1, VRN2 and VRN3; this model was based on the results of investigations using barley. This alternative model postulates that VRN1 and VRN3 mutually up-regulate each other: VRN1 first activates VRN3 expression, and then VRN3 further activates VRN1. The model was referred to as “the flowering model for temperate cereals” in a review paper on flowering in plants (Andres and Coupland 2012). More recently, a third model was suggested by Chen and Dubcovsky (2012). This model proposes that VRN1 is activated by VRN3 and then suppresses VRN2 expression. In this model, VRN1 is not essential for flowering; this conclusion was drawn from an analysis of a VRN1 mutant line. However, it is not certain that the mutant line is a true VRN1 knock-out, because its genotypic alteration is a point mutation and VRN1 mRNA is transcribed.
To obtain more information about the flowering mechanism in wheat, we are developing a large-scale panel of mutants induced by heavy-ion beam mutagenesis; these mutants are being systematically screened for effects on flowering time (Murai et al. 2013). Heavy-ion beam irradiation is effective at producing gene deletion mutants (null mutations) (Kazama et al. 2011, 2013). Here we describe four newly identified extra early-flowering mutant lines in diploid einkorn wheat, which have been named extra early-flowering 1 (exe1), exe2, exe3, and exe4.
Identification of exe Mutants
Seeds of the diploid einkorn wheat (Triticum monococcum L., 2n = 2x = 14, genome constitution AmAm) strain KU104-1 were given 50 Gy of 50 keV μm−1 LET (linear energy transfer) carbon ion beams and then sown in the field. The spikes of M1 plants were bagged and the harvested selfed seeds of each spike were used to produce the M2 lines. From approximately 1,200 M2 lines, we identified plants showing an abnormal extra early-heading phenotype; we termed these mutants extra early-flowering (exe). The original wild type (WT) strain KU104-1 is a spring habit einkorn wheat having a dominant VRN1 allele and a null VRN2 allele. Therefore, the exe mutants, exe1, exe2, exe3, exe4, identified in this study have no active gene at the VRN2 locus.
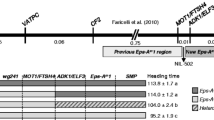
Table 19.1 shows the heading time of the WT and exe mutants in the field. Based on the heading time, the exe mutants were classified into two groups: Type I showed moderately extra early-heading type (exe1 and exe3); and Type II showed extremely extra early-heading type (exe2 and exe4). In the field, Type I and Type II headed about 30 and 45 days earlier than the WT, respectively.
Morphological Characteristics of the exe Mutants
WT plants and exe mutants were grown in the experimental field at Fukui Prefectural University and their morphological phenotypes were characterized during the maturation stage. Three agronomic characters were assessed: internode length, spike length, and spikelet number per spike.
The internode lengths of exe mutants were shorter than those of WT plants (Fig. 19.1). In particular, Type II exe mutants showed a significantly shorter first internode length than WT plants. Compared to WT plants, exe mutant plants produced smaller spikes with fewer spikelets (Fig. 19.2). As a consequence of the smaller numbers of spikelets, spike lengths in exe mutants were shorter than in WT plants. Furthermore, the spikes of Type II exe mutants were smaller than those of Type I mutants. These observations indicate that shortened culm lengths and fewer spikelets per spike are associated with the extra early-flowering phenotype in the exe mutants.
Hypothetical Model for Extra Early-Flowering Phenotype
The expression analysis of VRN1 indicated that VRN1 is highly expressed at earlier stages in Type II mutants than in Type I mutants under both short day (SD) and long day (LD) conditions (data not shown). This clearly indicates that the difference in earliness between Type I and Type II mutants is associated with the level of VRN1 expression. Thus, VRN1 is essential for flowering in wheat, and the level of expression of VRN1 determines flowering time (Fig. 19.3).
Schematic outline of the proposed mechanism for the extra early-flowering phenotype in exe mutants. This is based on the model of Shimada et al. (2009) in which VRN1 acts as an integrator of the vernalization and photoperiodic pathways that are coordinated with the autonomous pathway. VRN1 acts by up-regulating the florigen gene VRN3. In exe mutants, the mechanism for suppressing expression of VRN1 under SD conditions must be disrupted. Levels of accumulation of VRN1 transcripts induce VRN3 expression, resulting in the extra early-flowering phenotype. Note that the exe mutant has no VRN2 gene, because the original strain KU104-1 lacks VRN2 locus. Arrows and T-bars indicate promotion and suppression effect, respectively. Arrows indicated by bold lines show stronger effects
References
Andres F, Coupland G (2012) The genetic basis of flowering responses to seasonal cues. Nat Rev Genet 13:627–639
Chen A, Dubcovsky J (2012) Wheat TILLING mutants show that the vernalization gene VRN1 down-regulates the flowering repressor VRN2 in leaves but is not essential for flowering. PLoS Genet 8:e1003134
Danyluk J, Kane NA, Breton G et al (2003) TaVRT-1, a putative transcription factor associated with vegetative to reproductive transition in cereals. Plant Physiol 132:1849–1860
Diallo AO, Ali-Benali MA, Badawi M et al (2012) Expression of vernalization responsive genes in wheat is associated with histone H3 trimethylation. Mol Genet Genomics 287:575–590
Distelfeld A, Tranquilli G, Li C et al (2009) Genetic and molecular characterization of the VRN2 loci in tetraploid wheat. Plant Physiol 149:245–257
Dubcovsky J, Loukoianov A, Fu D et al (2006) Effect of photoperiod on the regulation of wheat vernalization genes VRN1 and VRN2. Plant Mol Biol 60:469–480
Kazama Y, Hirano T, Saito H et al (2011) Characterization of highly efficient heavy-ion mutagenesis in Arabidopsis thaliana. BMC Plant Biol 11:161
Kazama Y, Fujiwara MT, Hirano T et al (2013) Characterization of a heavy-ion induced white flower mutant of allotetraploid Nicotiana tabacum. Plant Cell Rep 32:11–19
Kitagawa S, Shimada S, Murai K (2012) Effect of Ppd-1 on the expression of flowering-time genes in vegetative and reproductive growth stages of wheat. Genes Genet Syst 87:161–168
Murai K, Miyamae M, Kato H et al (2003) WAP1, a wheat APETALA1 homolog, plays a central role in the phase transition from vegetative to reproductive growth. Plant Cell Physiol 44:1255–1265
Murai K, Nishiura A, Kazama Y, Abe T (2013) A large-scale mutant panel in wheat developed using heavy-ion beam mutagenesis and its application to genetic research. Nucl Inst Methods Phys Res B 314:59–62
Oliver SN, Finnegan EJ, Dennis ES et al (2009) Vernalization-induced flowering in cereals is associated with changes in histone methylation at the VERNALIZATION1 gene. Proc Natl Acad Sci U S A 106:88386–88391
Shimada S, Ogawa T, Kitagawa S et al (2009) A genetic network of flowering time genes in wheat leaves, in which an APETALA1/FRUITFULL-like gene, VRN1, is upstream of FLOWERING LOCUS T. Plant J 58:668–681
Trevaskis B (2010) The central role of the VERNALIZATION1 gene in the vernalization response of cereals. Funct Plant Biol 37:479–487
Trevaskis B, Bagnall DJ, Ellis MH et al (2003) MADS box genes control vernalization-induced flowering in cereals. Proc Natl Acad Sci U S A 100:13099–13104
Trevaskis B, Hemming MN, Peacock WJ, Dennis ES (2006) HvVRN2 responds to daylength, whereas HvVRN1 is regulated by vernalization and developmental status. Plant Physiol 140:1397–1405
Yan L, Loukoianov A, Tranquilli G et al (2003) Positional cloning of the wheat vernalization gene VRN1. Proc Natl Acad Sci U S A 100:6263–6268
Yan L, Loukoianov A, Blechl A, Tranquilli G et al (2004) The wheat VRN2 gene is a flowering repressor down-regulated by vernalization. Science 303:1640–1644
Yan L, Fu D, Li C, Blechl A et al (2006) The wheat and barley vernalization gene VRN3 is an orthologue of FT. Proc Natl Acad Sci U S A 103:19581–19586
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is distributed under the terms of the Creative Commons Attribution Noncommercial License, which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Copyright information
© 2015 The Author(s)
About this paper
Cite this paper
Nishiura, A., Kazama, Y., Abe, T., Mizuno, N., Nasuda, S., Murai, K. (2015). Extra Early-Flowering (exe) Mutants in Einkorn Wheat Generated by Heavy-Ion Beam Irradiation. In: Ogihara, Y., Takumi, S., Handa, H. (eds) Advances in Wheat Genetics: From Genome to Field. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55675-6_19
Download citation
DOI: https://doi.org/10.1007/978-4-431-55675-6_19
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55674-9
Online ISBN: 978-4-431-55675-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)