Abstract
Levels of radiocesium (134Cs + 137Cs) contamination in lake water, bottom sediment, plankton, and fish were investigated in three geographically separated lakes in Fukushima Prefecture (Lake Hayama, Lake Akimoto, and Lake Tagokura) between June 2012 and November 2013. Levels of contamination differed among the three lakes, with the highest levels in each measured component found in Lake Hayama, followed by Lake Akimoto, and the least contamination in Lake Tagokura. Among the lakes, the magnitude of contamination decreased with distance from the Fukushima Dai-ichi Nuclear Power Plant. Mean radiocesium concentrations were higher in piscivorous fish than in other fish, possibly reflecting differences in trophic levels. Radiocesium concentrations of the lake water, bottom sediment, plankton, and fish were significantly correlated with surface soil radiocesium content near lake sites.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Radiocesium is one of the major radioactive components of the fallout from the Fukushima Dai-ichi Nuclear Power Plant (FNPP). The total amount of 137Cs discharged into the atmosphere between 12 March and 6 April 2011 was estimated as approximately 1.3 × 1016 Bq (Chino et al. 2011). Before 20 April 2011, 18 % of the total fallout settled on Japanese land (Stohl et al. 2012). Radiocesium monitoring of freshwater organisms (Fisheries Agency 2012) showed that trophic level is an important ecological factor affecting bioaccumulation of 137Cs in fish (Mizuno and Kubo 2013). For example, in the Agano River basin in Fukushima, 137Cs contamination of carnivorous salmonids was roughly twice that of the herbivore ayu Plecoglossus altivelis (Mizuno and Kubo 2013). However, levels of radiocesium contamination among individuals within conspecifics have also been found to differ among habitats (Iguchi et al. 2013; Mizuno and Kubo 2013; Yamamoto et al. 2014a), and the causes of these differences are uncertain.
The objective of this study was to investigate factors affecting the differences in concentrations of radiocesium (134Cs + 137Cs) in fish of three geographically separate lakes in Fukushima Prefecture (Fig. 15.1, Table 15.1). These three lakes are located at differing distances from the FNPP and have different air dose rates and radiocesium concentrations in the adjacent surface soil, but have roughly similar retention times (Table 15.1). Sampling of lake water, bottom sediment, plankton, and fish was conducted up to three times per year in spring, summer, and autumn from June 2012 to November 2013.
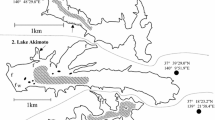
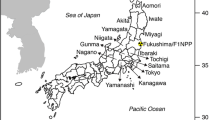
Upper figure shows the locations of the study lakes and the Fukushima Dai-ichi Nuclear Power Plant (FNPP) in Fukushima Prefecture. Lower figure show the sampling sites in each of the lakes (w lake water sampling point, f fish sampling point). Arrows point to bottom sediment sampling points (Modified from Matsuda et al. 2015)
2 Contamination Levels of Lake Water, Bottom Sediment, and Plankton
In each lake, lake water was sampled from one site (n = 1), and plankton was sampled one time along a constant distance of the lake surface (n = 1) (Fig. 15.1). Bottom sediment samples were collected from one point in each lake during one sampling event (n = 1) (Fig. 15.1). Temporal changes in radiocesium concentrations of the lake water, bottom sediment, and plankton are shown in Fig. 15.2. In Lake Tagokura, radiocesium concentrations were not detected in the lake water from November 2012 to October 2013 (detection limits, <2.1 mBq l−1), as was plankton in October 2013 (detection limit, 1,413 Bq kg−1 dry mass).
Among these ecosystem components, only the bottom sediment showed significant temporal changes, with a significant decreasing trend in Lake Hayama (Table 15.2; t test, P < 0.05) and a significant increasing trend in Lake Tagokura (Table 15.2; t test, P < 0.05). Continuing investigation is necessary to determine the patterns and temporal changes of radiocesium contamination in lake water and plankton.
The order of contamination levels of lake water, bottom sediment, and plankton for the study period (2012–2013) in three lakes was Lake Hayama > Lake Akimoto > Lake Tagokura (Table 15.2).
3 Radiocesium Concentrations in Fish
Fish from Lake Hayama were analyzed individually, except for Japanese smelt (Hypomesus nipponensis); all other fish samples were analyzed from 2 to 103 pooled individuals. Temporal changes in radiocesium concentrations in several fish species in each lake are shown in Fig. 15.3. Significant decreasing trends of radiocesium concentrations in fish from 2012 to 2013 were observed for white-spotted char in Lake Tagokura; smallmouth bass in both Lake Hayama and Lake Akimoto; bluegill, Japanese dace, and crucian carp in Lake Hayama; and Japanese barbell in Lake Akimoto (Table 15.3; t tests for parametric groups or Mann–Whitney tests for nonparametric groups, P < 0.05). Radiocesium concentrations significantly decreased by 33 % to 65 % between 2012 and 2013 in these fish species (Table 15.3).
Time-course of radiocesium (134Cs + 137Cs) concentrations in fish from each lake. Concentrations in Lake Hayama are for individual samples except for Hypomesus nipponensis; all other values were measured from pooled samples. Vertical bars indicate 1 SD. Samples below detection limits are indicated by closed squares on the x-axis
Considering only physical decay of radiocesium, the loss of radiocesium concentration in the fish on 20 June 2013 (the first sampling day on 2013) would have been expected to decrease by 9 % of that on 29 November 2012 (the last sampling day on 2012). Therefore, the radiocesium concentrations of crucian carp in Lake Tagokura might increase during the period between 2012 and 2013 without physical decay (Table 15.3). Fukushima and Arai (2014) also found that radiocesium concentrations in channel catfish (Ictalurus punctatus) and kokanee (Oncorhynchus nerka) increased between 2011 and 2013 in some lakes in northeastern Japan. The order of the contamination level in each fish species for the study period (2012–2013) among the three lakes was also Lake Hayama > Lake Akimoto > Lake Tagokura (Table 15.3).
4 Relationship Between Trophic Level and Radiocesium Concentration
Freshwater fish primarily accumulate radiocesium through the food chain rather than directly from the water (Williams and Pickering 1961; Hewett and Jefferies 1976; Yamamoto et al. 2014b). Species-specific food intake and food availability can cause differences in radiocesium concentrations among fish species. Because metals are concentrated in organisms as they are transferred up trophic levels by consumption, the trophic level of a fish is an important ecological factor affecting its concentration of radiocesium (Rowan and Rasmussen 1994). For example, after the Chernobyl accident in 1987, a higher annual mean concentration of 137Cs has been detected in fish from higher trophic levels in some lakes of Finland, including perch (Perca fluviatilis) and pike (Esox lucius) (Rask et al. 2012).
Okino (2002) showed that fishes classified as piscivorous, including salmonid fishes, the Japanese catfish Silurus asotus, and the largemouth bass Micropterus salmoides, occupy the top of the food chain in temperate lakes in Japan. We also categorized fish into two groups: (1) piscivorous fish (the white-spotted char Salvelinus leucomaenis pluvius, Japanese catfish, the smallmouth bass Micropterus dolomieu, largemouth bass, and masu salmon Oncorhynchus masou); (2) other fish (the bluegill Lepomis macrochirus, the Japanese dace Tribolodon hakonensis, the crucian carp Carassius spp., the common carp Cyprinus carpio, the Japanese barbel Hemibarbus barbus, and Japanese smelt). We found that the mean radiocesium concentration in piscivorous fish was significantly higher than in other types of fish only in Lake Hayama during the study period (2012–2013) (Table 15.4; t test, P < 0.01).
5 Geographic Differences in Levels of Radiocesium Contamination
The FNPP fallout was the source of radiocesium in freshwater fish and in lake water, bottom sediment, and plankton. Therefore, correlations between radiocesium concentrations in each of these lake ecosystem components and concentrations in lakeside surface soil were analyzed. Soil samples were taken at a 0- to 50-mm depth on the lakeside of each lake between 6 June and 8 July 2011 (MEXT 2011). The radiocesium concentrations of surface soil on each lakeside (MEXT 2011) are shown in Table 15.1. Significant correlations were found between surface soil radiocesium content and that of lake water (R 2 = 0.590, P < 0.01), bottom sediment (R 2 = 0.729, P < 0.001), plankton (R 2 = 0.555, P < 0.01), and all fish (R 2 = 0.273–0.971, P < 0.01) (Fig. 15.4).
Correlations between the mean radiocesium concentrations (134Cs + 137Cs) in each lake sample taken during the study period (2012–2013) and those of the surface soil taken at a 0- to 50-mm depth on each lakeshore between 6 June and 8 July 2011 (MEXT 2011). Vertical bars indicate 1 SD. Solid lines show significant fitted regression lines (Cited from Matsuda et al. 2015)
Thus, the differences in radiocesium concentrations in the lake samples likely reflect the quantity of radiocesium from the FNPP that was deposited at each lake. A previous report found a strong relationship between the distance from the FNPP and the radiocesium concentrations in freshwater fish (Mizuno and Kubo 2013). A similar relationship was observed in the present study, where the quantity of radiocesium deposited in lakeside soil decreased with distance from the FNPP. However, this relationship has not been found in all of northeastern Japan because the pollution did not spread concentrically from the FNPP. For example, Lake Chuzenji (located southwest of the FNPP) and Lake Tagokura are both located roughly 160 km from the FNPP in linear distance, but radiocesium concentrations in the muscle of salmonids in 2012 were 142.9–249.2 Bq kg−1 wet mass in Lake Chuzenji and only 12 Bq kg−1 wet mass in Lake Tagokura (Yamamoto et al. 2014a).
In addition, there is some evidence that different levels of radiocesium contamination of fish among lakes can also be caused by variation in retention time (Fukushima and Arai 2014), depth (Broberg et al. 1995), lake water hardness and conductivity (Hakanson et al. 1992; Särkkä et al. 1995), suspended sediment concentration, and temperature (Rowan and Rasmussen 1994).
In addition to continuing to measure levels of radiocesium contamination in these lakes, future studies are needed to determine the factors underlying continued contamination and the retention of radiocesium in these lakes.
References
Broberg A, Malmgren L, Jansson M (1995) Relations between resuspension and the content of 137Cs in freshwater fish in some Swedish lakes. J Aquat Ecosyst Health 4:285–294
Chino M, Nakayama H, Nagai H, Terada H, Katata G, Yamazawa H (2011) Preliminary estimation of release amounts of 131I and 137Cs accidentally discharged from the Fukushima Daiichi Nuclear Power Plant into the atmosphere. J Nucl Sci Technol 48:1129–1134
Fisheries Agency (2012) Results of the inspection on radioactivity materials in fisheries products. http://www.jfa.maff.go.jp/e/inspection/index.html. Accessed 3 June 2014
Fukushima T, Arai H (2014) Radiocesium contamination of lake sediments and fish following the Fukushima nuclear accident and their partition coefficient. Inland Waters 4:204–214
Hakanson L, Andersson T, Nilsson A (1992) Radioactive caesium in fish in Swedish lakes 1986–1988: general pattern related to fallout and lake characteristics. J Environ Radioact 15:207–229
Hewett CJ, Jefferies DF (1976) The accumulation of radioactive caesium from water by the brown trout (Salmo trutta) and its comparison with plaice and rays. J Fish Biol 9:479–489
Iguchi K, Fujimoto K, Kaeriyama H, Tomiya A, Enomoto M, Abe S, Ishida T (2013) Cesium-137 discharge into the freshwater fishery ground of grazing fish, ayu Plecoglossus altivelis, after the March 2011 Fukushima nuclear accident. Fish Sci 79:983–988
Matsuda K, Takagi K, Tomiya A, Enomoto M, Tsuboi J, Kaeriyama H, Ambe D, Fujimoto K, Ono T, Uchida K, Morita T, Yamamoto S (2015) Fisheries Science. doi:10.1007/s12562-015-0874-7
Ministry of Education, Culture, Sports, Science and Technology (2011) Nuclide analysis of the soil. http://www.mext.go.jp/b_menu/shingi/chousa/gijyutu/017/shiryo/icsFiles/afieldfile/2011/09/02/1310688_1.pdf. Accessed 3 June 2014 (in Japanese)
Mizuno T, Kubo H (2013) Overview of active cesium contamination of freshwater fish in Fukushima and Eastern Japan. Sci Rep 3:1742
Okino T (2002) Ecosystem of lake. Kyoritsu Shuppan, Tokyo (in Japanese)
Rask M, Saxen R, Ruuhijarvi J, Arvola L, Jarvinen M, Koskelainen U, Outola I, Vuorinen PJ (2012) Short- and long-term patterns of Cs-137 in fish and other aquatic organisms of small forest lakes in southern Finland since the Chernobyl accident. J Environ Radioact 103:41–47
Rowan DJ, Rasmussen JB (1994) Bioaccumulation of radiocesium by fish: the influence of physicochemical factors and trophic structure. Can J Fish Aquat Sci 51:2388–2410
Särkkä J, Jämsä A, Luukko A (1995) Chernobyl-derived radiocaesium in fish as dependent on water quality and lake morphometry. J Fish Biol 46:227–240
Stohl A, Seibert P, Wotawa G, Arnold D, Burkhart JF, Eckhardt S, Tapia C, Vargas A, Yasunari TJ (2012) Xenon-133 and caesium-137 releases into the atmosphere from the Fukushima Dai-ichi nuclear power plant: determination of the source term, atmospheric dispersion, and deposition. Atmos Chem Phys 12:2313–2343
Williams LG, Pickering Q (1961) Direct and food-chain uptake of cesium 137 and strontium 85 in bluegill fingerlings. Ecology 42:205–206
Yamamoto S, Yokoduka T, Fujimoto K, Takagi K, Ono T (2014a) Radiocesium concentrations in the muscle and eggs of salmonids from Lake Chuzenji, Japan, after the Fukushima fallout. J Fish Biol 84:1607–1613
Yamamoto S, Mutou K, Nakamura H, Miyamoto K, Uchida K, Takagi K, Fujimoto K, Kaeriyama H, Ono T (2014b) Assessment of radiocaesium accumulation by hatchery-reared salmonids after the Fukushima nuclear accident. Can J Fish Aquat Sci 71:1772–1775
Acknowledgments
This chapter was adapted from a paper published by Matsuda et al. (2015). The authors are grateful to Masato Murakami, Tomoko Okazaki, and Maki Yoshida for their assistance with the sample assays and data analyses. They also thank Kaoru Nakata for critical review of the manuscript. This study was supported by the Fisheries Agency, Ministry of Agriculture, Forestry and Fisheries, Japan.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is distributed under the terms of the Creative Commons Attribution Noncommercial License, which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Copyright information
© 2015 The Author(s)
About this chapter
Cite this chapter
Matsuda, K. et al. (2015). Comparison of the Radioactive Cesium Contamination Level of Fish and their Habitat Among Three Lakes in Fukushima Prefecture, Japan, After the Fukushima Fallout. In: Nakata, K., Sugisaki, H. (eds) Impacts of the Fukushima Nuclear Accident on Fish and Fishing Grounds. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55537-7_15
Download citation
DOI: https://doi.org/10.1007/978-4-431-55537-7_15
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55536-0
Online ISBN: 978-4-431-55537-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)