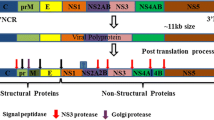
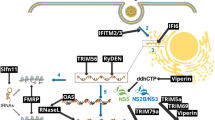
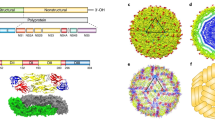
Summary
The importance of viral structural and non-structural antigens in flavivirus pathogenesis has not been satisfactorily assessed. This review examines the antigenic interrelationships of the flaviviruses, considers some of the implications of antigenic cross reactivity in virus virulence and highlights potential consequences of virus-antibody interactions.
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Aaskov JG, Williams L, Fletcher J, Hay R (1988) Failure of a dengue 1 sub-unit vaccine to protect mice against a lethal dengue virus infection. Am J Trop Med Hyg 39: 511–518
Aaskov JG, Geysen HM, Mason TJ (1989) Serologically defined linear epitopes in the envelope protein of dengue 2 (Jamaica strain 1409). Arch Virol 105: 209–221
Barrett ADT, Gould EA (1986) Antibody-mediated early death in vivo after infection with yellow fever virus. J Gen Virol 67: 2539–2542
Bell JR, Kinney RM, Trent DW, Lenches EM, Dalgarno L, Strauss JM (1985) N-terminal amino acid sequences of structural proteins of three flaviviruses. Virology 143: 224–229
Biedrizycka A, Cauchi MR, Darthomoeusz A, Gorman JJ, Wright P (1987) Characterization of protease cleavage sites involved in the formation of the envelope glycoprotein and three nonstructural proteins of dengue virus type 2, New Guinea C strain. J Gen Virol 68: 1317–1326
Blancou J, Andral B, Andral L (1980) A model in mice for the study of the early death phenomenon after vaccination and challenge with rabies virus. J Gen Virol 50: 433–435
Boege U, Heinz PX, Wengler G, Kunz C (1983) Amino acid composition and amino terminal sequences of the structural proteins of a flavivirus, European tick-borne encephalitis virus. Virology 126: 651–657
Brandriss MW, Schlesinger JJ, Walsh EE, Briselli M (1985) Lethal 17D yellow fever encephalitis in mice. I. Passive protection by monoclonal antibodies to the envelope proteins of 17D yellow fever and dengue 2 viruses. J Gen Virol 67: 229–234
Buckley A, Gould EA (1985) Neutralization of yellow fever virus studied using monoclonal and polyclonal antibodies. J Gen Virol 66: 2523–2531
Buckley A, Gould EA (1988) Detection of virus-specific antigen in the nuclei or nucleoli of cells infected with Zika or Langat virus. J Gen Virol 69: 1913–1920
Calisher CH, Karabatsos N, Dalrymple JM, Shope RE, Porterfield J, Westaway EG, Brandt WE (1989) Antigenic relationships between flaviviruses as determined by cross-neutralization tests with polyclonal antisera. J Gen Virol 70: 37–43
Cammack N, Gould EA (1986) Topographical analysis of epitope relationships on the envelope glycoprotein of yellow fever 17D vaccine and the wild type Asibi parent virus. Virology 150: 333–341
Cane PA, Gould EA (1988) Reduction of yellow fever virus mouse neurovirulence by immunization with a bacterially synthesized non-structural protein (NS 1) fragment. J Gen Virol 69: 1241–1246
Cardiff RD, Lund J (1976) Distribution of dengue 2 antigens by electron immunocytochemistry. Infect Immun 13: 1699–1709
Cardosa MJ, Porterfield JS, Gordon S (1983) Complement receptor mediates enhanced flavivirus replication in macrophages. J Exp Med 158: 258–263
Casals J (1963) Relationships among arthropod-borne viruses determined by cross-challenge tests. Am J Trop Med Hyg 12: 587–596
Casals J, Brown LV (1954) Haemagglutination with arthropod borne viruses. J Exp Med 99: 429–449
Castle E, Leidner U, Nowak T, Wengler G, Wengler G (1986) Primary structure of the flavivirus West Nile genome region coding for all nonstructural proteins. Virology 149: 10–26
Castle E, Nowak T, Leidner U, Wengler G, Wengler G (1985) Sequence analysis of the viral core protein and the membrane-associated proteins V1 and NV2 of the flavivirus West Nile virus and of the genome sequence for these proteins. Virology 145: 227–236
Cecilia D, Gadkari DA, Kedarnath N, Ghosh SN (1988) Epitope mapping of Japanese encephalitis virus envelope protein using monoclonal antibodies against an Indian strain. J Gen Virol 69: 2741–2747
Chanas AC, Gould EA, Clegg JCS, Varma MGR (1982) Monoclonal antibodies to Sindbis virus glycoprotein E 1 can neutralize, enhance infectivity and independently inhibit haemagglutination or haemolysis. J Gen Virol 58: 37–46
Clarke DH (1964) Further studies on antigenic relationship among the viruses of the group B tick-borne complex. Bull WHO 31: 45–65
Dales S, Fujinami RS, Oldstone MBA (1983) Infection with vaccinia favors the selection of hybridomas synthesising autoantibodies against intermediate filaments, one of them cross-reacting with the virus haemagglutinin. J Immunol 131: 1546–1553
Dalgarno L, Trent DW, Strauss JH, Rice CM (1986) Partial nucleotide sequence of the Murray Valley encephalitis virus genome. Comparison of the encoded polypeptides with Yellow Fever virus structural and non-structural proteins. J Mol Biol 187: 309–323
Deubel V, Kinney RM, Trent DW (1986) Nucleotide sequence and deduced amino acid sequence of the structural proteins of dengue type 2 virus, Jamaican genotype. Virology 155: 365–377
Deubel V, Kinney RM, Trent DW (1988) Nucleotide sequence and deduced amino acid sequence of the nonstructural proteins of Dengue type 2 virus, Jamaica genotype: comparative analysis of the full length genome. Virology 165: 234–244
de Madrid AT, Porterfield JS (1974) The Flaviviruses (group B arboviruses) a cross-neutralization study. J Gen Virol 23: 91–96
Dittmar D, Haines HG, Castro A (1980) Monoclonal antibodies specific for dengue virus type 3. J Clin Microbiol 12: 74–78
Fujinami RS, Oldstone MBA, Wroblewska Z, Frankel ME, Koprowski H (1983) Molecular mimicry in virus infection: cross reaction of measles virus phosphoprotein or of herpes simplex virus protein with human intermediate filaments. Proc Natl Acad Sci USA 80: 2346–2350
Gentry MK, Henchal EA, McCown JM, Brandt WE, Dalrymple JM (1982) Identification of distinct antigenic determinants on dengue 2 virus using monoclonal antibodies. Am J Trop Med Hyg 31: 548–555
Gould EA, Buckley A (1989) Antibody dependent enhancement of yellow fever and Japanese encephalitis virus neurovirulence. J Gen Virol 70: 1605–1608
Gould EA, Chanas AC, Buckley A, Clegg CS (1983) Monoclonal immunoglobulin M antibody to Japanese encephalitis virus that can react with a nuclear antigen in mammalian cells. Infect Immun 41: 774–779
Gould EA, Buckley A, Cammack N, Barrett ADT, Clegg JCS, Ishak R, Varma MGR (1985) Examination of the immunological relationships between flaviviruses using yellow fever virus monoclonal antibodies. J Gen Virol 66: 1369–1382
Gould EA, Buckley A, Barrett ADT, Cammack N (1986) Neutralizing (54K) and non-neutralizing (54K and 48K) monoclonal antibodies against structural and nonstructural yellow fever virus proteins confer immunity in mice. J Gen Virol 67: 591–595
Gould EA, Buckley A, Groeger BK, Cane PA, Doenhoff M (1987) Immune enhancement of yellow fever virus neurovirulence for mice: Studies of mechanism involved. J Gen Virol 68: 3105–3112
Halstead SB, Chow JS, Marchette MJ (1973) Immunological enhancement of dengue virus replication. Nature 243: 24–25
Halstead SB, O’Rourke EJ (1977) Dengue viruses and mononuclear phagocytes. I. Infection enhancement by non-neutralizing antibody. J Exp Med 146: 201–217
Halstead SB (1981) The pathogenesis of dengue: molecular epidemiology in infectious disease. Am J Epidemiol 114: 632–648
Haspel MV, Onodera T, Prabhakar BS, Horita M, Suzuki H, Notkins AL (1983) Virus induced autoimmunity: monoclonal antibodies that react with endocrine tissues. Science 220: 304–306
Haynes BF, Robert-Gurhoff M, Metzgar RS, Franchini G, Kalyanamaran VS, Palker TJ, Gallo RC (1983) Monoclonal antibody against human T cell leukaemia virus p19 defines a human thymic epithelial antigen acquired during ontogeny. J Exp Med 157: 907–920
Hawkes RA, Lafferty KJ (1967) The enhancement of virus infectivity by antibody. Virology 33: 250–261
Heinz FX, Berger R, Tuma W, Kunz C (1983) Location of immunodominant antigenic determinants on fragments of the tick-borne encephalitis virus glycoprotein: evidence for two different mechanisms by which antibodies mediate neutralization and haemagglutination—inhibition. Virology 126: 525–537
Heinz FX, Mandl C, Berger R, Tuma W, Kunz C (1984) Antibody-induced conformational changes result in enhanced avidity of antibodies to different antigenic sites on the tick-borne encephalitis virus glycoprotein. Virology 133: 25–34
Henchal EA, Gentry MK, McCown JM, Brandt WE (1982) Dengue virus-specific and flavivirus group determinants identified with monoclonal antibodies by indirect immunofluorescence. Am J Trop Med Hyg 31: 830–836
Henchal EA, McCown JM, Seguin MC, Gentry MK, Brandt WE (1983) Rapid identification of dengue virus isolates by using monoclonal antibodies in an indirect immunofluorescence assay. Am J Trop Med Hyg 32: 164
Henchal EA, McCown JM, Burke DS, Seguin MC, Brandt WE (1985) Epitope analysis of antigenic determinants on the surface of dengue 2 virions using monoclonal antibodies. Am J Trop Med Hyg 34: 162–169
Hotta H, Wiharta AS, Hotta S (1984) Antibody-mediated enhancement of dengue virus infection in mouse macrophage cell lines Mkt and Mml. Proc Soc Exp Biol Med 175: 320–327
Innis BL, Thirawuth V, Hemachuda C (1989) Identification of continuous epitopes of the envelope glycoprotein of dengue type 2 virus. Am J Trop Med Hyg 40: 676–687
Kaufman BM, Summers PL, Dubois DR, Cohen WH, Gentry MK, Timchak RL, Burke DS, Eckels KH (1989) Monoclonal antibodies for dengue virus prM glycoprotein protect mice against lethal dengue infection. Am J Trop Med Hyg 41: 576–580
Kimura-Kuroda J, Yasui K (1983) Topographical analysis of antigenic determinants on envelope glycoprotein V3 ( E) of Japanese encephalitis virus, using monoclonal antibodies. J Virol 45: 124–132
Kobayashi Y, Hasegawa H, Oyama T, Tamai T, Kusaba T (1984) Antigenic analysis of Japanese encephalitis virus by using monoclonal antibodies. Infect Immun 44: 117–123
Lane DP, Hoeffler WK (1980) SV40 large T shares an antigenic determinant with a cellular protein of molecular weight 68,000. Nature 288: 167–170
McAda PC, Mason PW, Schmaljohn CS, Dalrymple JM, Mason TL, Fournier MJ (1987) Partial nucleotide sequence of the Japanese encephalitis virus genome. Virology 158: 348–360
Mandl CW, Heinz FX, Kunz C (1988) Sequence of the structural proteins of tick-borne encephalitis virus ( Western subtype) and comparative analysis with other flaviviruses. Virology 166: 197–205
Mandl CW, Guirhakoo F, Holzman H, Heinz FX, Kunz C (1989) Antigenic structure of the flavivirus envelope protein E at molecular level, using tick-borne encephalitis virus as a model. J Virol 63: 564–571
Mathews JM, Roehrig JT (1984) Elucidation of the topography and determination of the protective epitopes on the E glycoprotein of St Louis encephalitis virus by passive transfer with monoclonal antibodies. J Immun 132: 1533–1537
Monath TP (1986) Pathobiology of the flaviviruses. In: Schlesinger S, Schlesinger MJ (eds) The Togaviridae and Flaviviridae. Academic Press, New York, pp 375–440
Monath TP, Schlesinger JJ, Brandriss MW, Cropp CB, Prange WB (1984) Yellow fever monoclonal antibodies: type-specific and cross-reactive determinants identified by immunofluorescence. Am J Trop Med Hyg 33: 695–698
Murphy FA (1980) Morphology and morphogenesis. In: Monath TP (ed) St. Louis encephalitis. American Public Health Association, Washington, DC, pp 65–103
Nowak T, Wengler G (1987) Analysis of disulphides present in the membrane proteins of the West Nile flavivirus. Virology 156: 127–137
Peiris JSM, Porterfield JS (1979) Antibody-mediated enhancement of flavivirus replication in macrophage-like cell lines. Nature 282: 509–511
Peiris JSM, Gordon S, Unkeless JC, Porterfield JS (1981) Monoclonal anti Fc receptor IgG blocks enhancement of viral replication in macrophages by antiviral antibody. Nature 289: 189–191
Peiris JSM, Porterfield JS, Roehrig JT (1982) Monoclonal antibodies against the flavivirus West Nile. J Gen Virol 58: 283–289
Phillpotts RJ, Stephenson JR, Porterfield JS (1985) Antibody-dependent enhancement of tick-borne encephalitis virus infectivity. J Gen Virol 66: 1831–1837
Porterfield JS (1980) Antigenic characteristics and classification of Togaviridae. In: Schlesinger RW (ed) The Togaviruses. Academic Press, New York, pp 13–46
Porterfield JS (1982) Immunological enhancement and the pathogenesis of dengue haemorrhagic fever. J Hyg 89: 355–364
Porterfield JS (1986) Antibody-dependent enhancement of viral infectivity. Adv Virus Res 31: 335–352
Prabhakar BS, Nathanson N (1981) Acute rabies death mediated by antibody. Nature 290: 590–591
Rice CM, Lenches EM, Eddy SR, Shin SJ, Sheets RL, Strauss JH (1985) Nucleotide sequence of yellow fever virus: Implications for flavivirus gene expression and evolution. Science 229: 726–733
Rice CM, Strauss EG, Strauss JH (1986) Structure of the flavivirus genome. In: Schlesinger S, Schlesinger MJ (eds) The Togaviridae and Flaviviridae. Plenum, New York, pp 279–326
Roehrig JT, Hunt AR, Johnson AJ, Hawkes RA (1989) Synthetic peptides derived from the deduced amino acid sequences of the E glycoprotein of Murray Valley encephalitis virus elicit antiviral antibody. Virology 171: 49–60
Roehrig JT, Mathews JH, Trent DW (1983) Identification of epitopes on the E glycoprotein of Saint Louis encephalitis virus using monoclonal antibodies. Virology 128: 118–126
Schlesinger JJ, Brandriss MW (1981) Growth of 17D yellow fever virus in a macrophage-like cell line, U937: role of Fc and viral receptors in antibody-mediated infection. J Immunol 127: 659–665
Schlesinger JJ, Brandriss MW, Monath TP (1983) Monoclonal antibodies distinguish between wild and vaccine strains of yellow fever virus by neutralization, haemagglutination—inhibition and immunoprecipitation of virus envelope protein. Virology 125: 8–17
Schlesinger JJ, Brandriss MW, Walsh EE (1985) Protection against 17D yellow fever encephalitis in mice by passive transfer of monoclonal antibodies to the non-structural glycoprotein gp48 and by active immunization with gp48. J Immunol 135: 2805–2809
Schlesinger JJ, Brandriss MW, Cropp CB, Monath TP (1986) Protection against yellow fever in monkeys by immunization with yellow fever virus nonstructural NS1. J Virol 60: 1153–1155
Shapiro D, Brandt WE, Russell PK (1972) Changes involving a viral membrane glycoprotein during morphogenesis of group B arboviruses. Virology 50: 906–911
Sheshberadaran H, Norrby E (1984) Three monoclonal antibodies against measles virus F protein cross-react with cellular stress proteins. J Virol 52: 995–999
Sikes RK, Cleary WF, Koprowski H, Wiktor TJ, Kaplan MM (1971) Effective protection of monkeys against death from street rabies by post-exposure administration of tissue culture rabies vaccine. Bull WHO 45: 1–11
Speight S, Coia G, Parker MD, Westaway EG (1988) Gene mapping and positive identification of the nonstructural proteins NS2A, NS2B, NS3 and NS5 of the flavivirus Kunjin and their cleavage sites. J Gen Virol 69: 23–34
Srinivasappa J, Saegusa J, Prabhakar BS, Gentry MK, Buchmeier MJ, Wiktor TJ, Koprowski H, Oldstone MBA, Notkins AL (1986) Molecular mimicry: frequency of reactivity of monoclonal antiviral antibodies with normal tissues. J Virol 57: 397–401
Stephenson JR, Lee JM, Wilton-Smith PD (1984) Antigenic variation among members of the tick-borne encephalitis complex. J Gen Virol 65: 81–89
Sumiyoshi H, Mori C, Morita K, Kuhara S, Kondou J, Kukushi Y, Nagamatu H, Igarashi A (1987) Complete nucleotide sequence of the Japanese encephalitis virus genome RNA. Virology 161: 497–510
Tadano M, Makino Y, Fukunaga T, Okuno Y, Fukai K (1989) Detection of Dengue 4 virus core protein in the nucleus. I. A monoclonal antibody to dengue 4 virus reacts with the antigen in the nucleus and cytoplasm. J Gen Virol 70: 1409–1415
Tikhomirova TI, Karpovich LG, Reingold VN, Levkovich EN, Shestopalova NM (1968) Comparative study of Langat and tick-borne encephalitis viruses in cultured cells. Acta Virol 12: 529–534
Trent DW (1977) Antigenic characterization of flavivirus structural proteins separated by isoelectric focusing. J Virol 22: 608–618
Trent DW, Kinney RM, Johnson BJB, Vordnam AV, Grant JA, Deubel V, Rice CM, Hahn C (1987) Partial nucleotide sequence of St Louis encephalitis virus RNA: structural proteins, NS1, ns2a and ns2b. Virology 156: 293–304
Webb HE, Wight DGD, Platt GS, Smith CEG (1968) Langat virus encephalitis in mice. I. The effect of the administration of specific antiserum. J Hyg 66: 343–354
Weiss RC, Scott FW (1981) Antibody mediated enhancement of disease in feline infectious peritonitis: comparisons with dengue haemorrhagic fever. Comp Immunol Microbiol Infect Dis 4: 175–289
Wengler G, Castle E, Leidner U, Nowak T, Wengler G (1985) Sequence analysis of the membrane protein V3 of the flavivirus West Nile virus and of its gene. Virology 147: 264–274
Wengler G, Wengler G, Nowak T, Wahn K (1987) Analysis of the influence of proteolytic cleavage on the structural organization of the surface of the West Nile flavivirus leads to the isolation of a protease-resistant E protein oligomer from the viral surface. Virology 160: 210–219
Westaway EG (1973) Proteins specified by group B Togaviruses in mammalian cells during productive infection. Virology 51: 454–465
Westaway EG (1980) Replication of flaviviruses. In: Schlesinger RW (ed) The Toga-viruses. Academic Press, New York, pp 531–577
Westaway EG, Gaidamovich SYa, Horzinek MS, Igarashi A, Kääriäinen L, Lvov DK, Porterfield JS, Russell PK, Trent DW (1985) Flaviviridae. Intervirology 24: 183–192
Yasui K (1984) The antigenic structure of Japanese encephalitis virus glycoprotein VB(E). In: Proceedings of the International Workshop on the Molecular Biology of Flaviviruses, Fort Detrick, pp 16–17
Yasuzumi G, Tsubo I (1965) Analysis of the development of Japanese B encephalitis (JBE) virus. III. Electron microscope studies on inclusion bodies appearing in neurons and microglial cells infected with JBE virus. J Ultrastruc Res 12: 317–327
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1990 Springer-Verlag
About this paper
Cite this paper
Gould, E.A., Buckley, A., Higgs, S., Gaidamovich, S. (1990). Antigenicity of flaviviruses. In: Calisher, C.H. (eds) Hemorrhagic Fever with Renal Syndrome, Tick- and Mosquito-Borne Viruses. Archives of Virology Supplementum, vol 1. Springer, Vienna. https://doi.org/10.1007/978-3-7091-9091-3_17
Download citation
DOI: https://doi.org/10.1007/978-3-7091-9091-3_17
Publisher Name: Springer, Vienna
Print ISBN: 978-3-211-82217-3
Online ISBN: 978-3-7091-9091-3
eBook Packages: Springer Book Archive