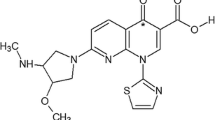
Abstract
In the treatment of hematological malignancies with idarubicin or etoposide two different pharmacological phases can be discriminated. Shortly after i.v. application, a peak with high plasma concentration can be found which lasts shorter than 3 h. As well after i.v. application as after oral application, a long phase with 10–100 fold lower concentration (through level) can be found which lasts for hours or even days. The cellular reaction upon a short exposure of topoisomerase II inhibitors at high concentration is completely different than the cellular reaction upon a long exposure of topoisomerase II inhibitors at very low concentrations. Especially the epipodophyllotoxines have a high capacity to induce cellular differentiation at these conditions which is not the case with anthracyclines. For the induction of apoptosis, a exposure-time which is longer than the cell-cycle-time of the leukemic cells seems to be much more important than especially high concentrations of topoisomerase II inhibitors.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Minderman H, Linssen P, Wessels J, Haanen C (1993) Cell cycle related uptake, retention and toxicity of idarubicin, daunorubicin and doxorubicin. Anticancer Res 13 (4): 1161–1165
Robert J, Gianni L (1993) Pharmacokinetics and metabolism of anthracyclines. Cancer Sury 17 (1): 219–252
Gieseler F, Biersack H, Brieden T, Manderscheid J, Nussler V (1994) Cytotoxicity of anthracydines: correlation with cellular uptake, intracellular distribution and DNA binding. Ann Hematol 69 (1): 13–17
Robert J, Gianni L (1993) Pharmacokinetics and metabolism of anthracyclines. Cancer Sury 17 (1): 219–252
Berman E, McBride M (1992) Comparative cellular pharmacology of daunorubicin and idarubicin in human multidrug-resistant leukemia cells. Blood 79 (12): 3267–3273
Cusack BJ, Young SP, Driskell J, Olson RD (1993) Doxorubicin and doxorubicinol pharmacokinetics and tissue concentrations following bolus injection and continuous infusion of doxorubicin in the rabbit. Cancer Chemother Pharmacol 32 (1): 53–58
Hollingshead LM, Faulds D (1991) Idarubicin. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in the chemotherapy of cancer. Drugs 42 (4): 690–719
Smith DB, Margison JM, Lucas SB (1987) Clinical pharmacology of oral and intravenous idarubicin. Cancer Chemother Pharmacol 19: 138–142
Stewart DJ, Grewaal D, Green RM (1991) Bio-availability and pharmacology of oral idarubicin. Cancer Chemother Pharmacol 27: 308–314
Zanette L, Zuchetti M, Freshi A (1990) Pharmacokinetics of idarubicin in cancer patients. Cancer Chemother Pharmacol 25: 445–448
Camaggi CM, Strocchi E, Carisi P (1992) Idarubicin metabolism and pharmacokinetics after intravenous and oral application in cancer patients–a crossover study. Cancer Chemother Pharmacol 30: 307–316
Cersosimo RJ (1992) Idarubicin: an anthracy- dine antineoplastic agent. Clin Pharm 11 (2): 152–167
Stahelin HF, von Wartburg A (1991) The chemical and biochemical route from podophyllotoxin glucoside to etoposide. Cancer Res 51: 5–15
Dow L, Linkule K, Look A (1983) Comparative cytotoxicity of epipodophyllotoxines and their metabolites on human leukemia lymphoblaste. Cancer Res 43: 5692–5695
Zulian GB, Jotterand BM, Cabrol C, Beris P, Mermillod B, Alberto P (1993) Etoposide and secondary haematological malignancies: coincidence or causality? Ann Oncol 4 (7): 559–566
Chabot GG, Armand JP, Terret C, de Forni M (1996) Etoposide bioavailability after oral administration of the prodrug etoposide phosphate in cancer patients during a phase I study. J Clin Oncol 14 (7): 2020–2030
Gieseler F, Boege F, Biersack H, Spohn B, Clark M, Wilms K (1991) Nuclear topoisomerase II activity changes during HL-60 cell differentiation: alterations of drug sensitivity and pH-dependency. Leukemia and Lymphoma 5: 273–279
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1998 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Gieseler, F. (1998). Pharmacokinetic Basis for an Oral Chemotherapy with Idarubicin and Etoposide: Dose-Dependent Biological Effects of Topoisomerase-II-Inhibitors. In: Hiddemann, W., et al. Acute Leukemias VII. Haematology and Blood Transfusion / Hämatologie und Bluttransfusion, vol 39. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-71960-8_71
Download citation
DOI: https://doi.org/10.1007/978-3-642-71960-8_71
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-71962-2
Online ISBN: 978-3-642-71960-8
eBook Packages: Springer Book Archive