Abstract
Future military special operator selection and education programs will take advantage of state-of-the-art neuroimaging and normative statistical tools in the creation of a customized database of EEG patterns gathered from top performing specialists over their careers. Such a quantitative EEG Normative Database (qEND) will function as the benchmark for screening, assessment, selection and even training of targeted individuals required to work effectively as operators under extreme stresses and for extended periods. This assumption implies that an improved warfighter selection and training pedagogy will embrace the concept of a “model” brain activity pattern (BAP) that represents a warfighter at peak potential and in a highly focused and resilient state of mind. It also implies that this model BAP can be used to: 1) identify biomarkers of positive traits in candidates for specialized training programs, and 2) reduce stress and improve sleep and training performance of program selectees using guided EEG neurofeedback to maintain an optimal BAP. One such statistical qEND (NeuroGuide) is used clinically in the assessment and diagnosis of EEG imbalances specifically related to neurological and behavioral disorders, as well as for guiding individual brain pattern changes through the use of neurofeedback training (NT).
To evaluate qEEG for monitoring an individual’s BAP changes and potentially improving mood and work performance, two military specialists with leadership experience underwent a program of pre- and post-EEG recordings and 20 neurofeedback training (NT) sessions. Here, the NeuroGuide database was used to determine how each participant’s BAP differed from the age-matched group norms, and it was also used during the NT process to inform the software of the differences from the norms at each of the 4 training sites used to adjust the trainees EEG towards the direction of “normal”.
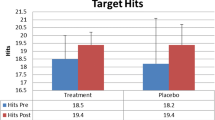
Changes from the NT program were assessed pre- and post-intervention using seven neuropsychological assessments of mood, anxiety, sleep, work performance and life satisfaction. In addition, one subject had a series of blood draws taken over the course of the NT program to evaluate changes in his plasma Cortisol; a reliable biomarker of stress level. Both subjects reported reduced levels of anxiety, impulsivity and anger, and improved mood and life satisfaction after the 20-session NT intervention.
Chapter PDF
Similar content being viewed by others
Keywords
References
Pascual-Leone, A., Freitas, C., Oberman, L., Horvath, J.C., et al.: Characterizing Brain Cortical Plasticity and Network Dynamics Across the Age-Span in Health and Disease with TMS-EEG and TMS-fMRI. Brain Topogr. 24(3-4), 302–315 (2011)
Chu, C.J., Kramer, M.A., Pathmanathan, J., Bianchi, M.T., Westover, M.B., Wizon, L., Cash, S.S.: Emergence of stable functional networks in long-term human electroencephalography. J. Neurosci. 32(8), 2703–2713 (2012)
Thatcher, R.W., Walker, R.A., Biver, C.J., North, D.M., Curtin, R.: Sensitivity and Specificity of an EEG Normative Database: Validation and Clinical Correlation. J. Neurotherapy 7(3/4), 87–121 (2003)
Collura, T.F.: Neuronal Dynamics in Relation to Normative Electroencephalography Assessment and Training. Biofeedback 36(4), 134–139 (2009)
Hoedlmoser, K., Pecherstorfer, T., Gruber, G., Anderer, P., Doppelmayr, M., Klimesch, W., Schabus, M.: Instrumental conditioning of human sensorimotor rhythm (12-15 Hz) and its impact on sleep as well as declarative learning. Sleep 31(10), 1401–1408 (2008)
Michael, A.J., Krishnaswamy, S., Mohamed, J.: An open label study of the use of EEG biofeedback using beta training to reduce anxiety for patients with cardiac events. Neuropsychiatr. Dis. Treat. 1(4), 357–363 (2005)
Giordano, J., DuRousseau, D.R.: Toward Right and Good Use of Brain-Machine Interfacing Neurotechnologies: Ethical Issues, and Implications for Guidelines and Policy. Cog. Technol. 15(2), 5–10 (2011)
Quan, M., Zheng, C., Zhang, N., Han, D., Tian, Y., Zhang, T., Yang, Z.: Impairments of behavior, information flow between thalamus and cortex, and prefrontal cortical synaptic plasticity in an animal model of depression. Brain Res. Bull. 85(3-4), 109–116 (2011)
Flo, E., Steine, I., Blågstad, T., Grønli, J., Pallesen, S., Portas, C.: Transient changes in frontal alpha asymmetry as a measure of emotional and physical distress during sleep. Brain Res. 1367, 234–249 (2011) (Epub October 1, 2010)
Dias-Ferreira, E., Sousa, J.C., Melo, I., Morgado, P., Mesquita, A.R., Cerqueira, J.J., Costa, R.M., Sousa, N.: Chronic stress causes frontostriatal reorganization and affects decision-making. Science 325(5940), 621–625 (2009)
Leuchter, A.F., Cook, I.A., Hunter, A.M., Cai, C., Horvath, S.: Resting-State Quantitative Electroencephalography Reveals Increased Neurophysiologic Connectivity in Depression. PLoS ONE 7(2), e32508 (2012), doi:10.1371/journal.pone.0032508.
Sirota, A., Buzsáki, G.: Interaction between neocortical and hippocampal networks via slow oscillations. Thalamus Relat. Syst. 3(4), 245–259 (2005)
Menon, V., Uddin, L.: Saliency, switching, attention and control: a network model of insula function. Brain Struct. Funct. 214(5-6), 655–667 (2010) (Epub 2010 May 29, 2010), doi:10.1007/s00429-010-0262-0.
Brembs, B.: Operant conditioning in invertebrates. Curr. Opin. Neurobiol. 13(6), 710–717 (2003)
Gruzelier, J.: A theory of alpha/theta neurofeedback, creative performance enhancement, long distance functional connectivity and psychological integration. Cogn. Process. 10 (suppl. 1), 101–109 (2009) (Epub December 11, 2008 )
DuRousseau, D.R., Mindlin, G., Insler, J., Levin II: Operational Study to Evaluate Music-Based Neurotraining at Improving Sleep Quality, Mood and Daytime Function in a First Responder Population. Journal Neurotherapy 4, 389–398 (2011)
Collura, T.F., Thatcher, R.W.: Clinical benefit to patients suffering from recurrent migraine headaches and who opted to stop medication and take a neurofeedback treatment series. Clin. EEG Neurosci. 42(2), VIII–IX (2011)
Scharnowski, F., Hutton, C., Josephs, O., Weiskopf, N., Rees, G.: Improving Visual Perception through Neurofeedback. J. of Neuroscience 32(49), 17830–17841 (2012)
Ros, T., Moseley, M.J., Bloom, P.A., Benjamin, L., Parkinson, L.A., Gruzelier, J.H.: Optimizing microsurgical skills with EEG neurofeedback. BMC Neurosci. 10, 87 (2009)
Tacker, M.M., Leach, C.S., Owen, C.A., Rummel, J.: Levels of cortisol, corticosterone, cortisone and 11-deoxycoritsol in the plasma of stressed and unstressed subjects. J. Endocrinol. 76(1), 165–166 (1978)
Swigar, M.E., Kolakowska, T., Quinlan, D.: Plasma cortisol levels in depression and other psychiatric disorders: a study of newly admitted psychiatric patients. Psychol. Med. 9(3), 449–455 (1979)
Plischke, H., DuRousseau, D., Giordano, J.: EEG-based Neurofeedback– The Promise of Neurotechnology and Need for Neuroethically-informed Guidelines and Policies. J. Ethics Biol. Engineer. Med. (July 2012), doi:10.1615/EthicsBiologyEngMed.2012004853
Canli, T., Brandon, S., Casebeer, W., Crowley, P.J., DuRousseau, D., Greely, H., Güzeldere, G., Pascual-Leone, A.: Neuroethics and National Security. The American Journal of Bioethics 7(5), 3–13 (2007)
Gianotti, L.R.R., Figner, B., Ebstein, R.P., Knoch, D.: Why some people discount more than others: baseline activation in the dorsal PFC mediates the link between COMT genotype and impatient choice. Frontiers in Neuroscience, Decision Neuroscience 6, Article 54, 1–12 (2012)
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
DuRousseau, D.R. (2013). QEEG Biomarkers: Assessment and Selection of Special Operators, and Improving Individual Performance. In: Schmorrow, D.D., Fidopiastis, C.M. (eds) Foundations of Augmented Cognition. AC 2013. Lecture Notes in Computer Science(), vol 8027. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-39454-6_60
Download citation
DOI: https://doi.org/10.1007/978-3-642-39454-6_60
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-39453-9
Online ISBN: 978-3-642-39454-6
eBook Packages: Computer ScienceComputer Science (R0)