Abstract
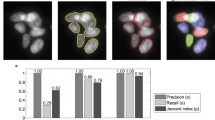
Accurate and efficient segmentation of cells in fluorescence microscopy images is of central importance for the quantification of protein expression in high-throughput screening applications. We propose a new approach for segmenting cell nuclei which is based on active contours and convex energy functionals. Compared to previous work, our approach determines the global solution. Thus, the approach does not suffer from local minima and the segmentation result does not depend on the initialization. We also suggest a numeric approach for efficiently computing the solution. The performance of our approach has been evaluated using fluorescence microscopy images of different cell types. We have also performed a quantitative comparison with previous segmentation approaches.
Chapter PDF
Similar content being viewed by others
Keywords
- Segmentation Result
- Active Contour
- Fluorescence Microscopy Image
- Energy Functional
- Intensity Inhomogeneity
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Chan, T.F., Esedoglu, S., Nikolova, M.: Algorithms for finding global minimizers of image segmentation and denoising models. SIAM J. Appl. Math. 66, 1632–1648 (2006)
Chan, T.F., Vese, L.A.: Active contours without edges. IEEE Trans. Image Process 10(2), 266–277 (2001)
Coelho, L.P., Shariff, A., Murphy, R.F.: Nuclear segmentation in microscope cell images: a hand-segmented dataset and comparison of algorithms. In: ISBI 2009, pp. 518–521 (2009)
Dufour, A., Shinin, V., Tajbakhsh, S., Guillen-Aghion, N., Olivo-Marin, J.C., Zimmer, C.: Segmenting and Tracking Fluorescent Cells in Dynamic 3-D Microscopy With Coupled Active Surfaces. IEEE Trans. Image Process 14(9), 1396–1410 (2005)
Dzyubachyk, O., van Cappellen, W.A., Essers, J., Niessen, W.J., Meijering, E.H.W.: Advanced Level-Set-Based Cell Tracking in Time-Lapse Fluorescence Microscopy. IEEE Trans. Med. Imag. 29(3), 852–867 (2010)
Goldstein, T., Bresson, X., Osher, S.: Geometric Applications of the Split Bregman Method: Segmentation and Surface Reconstruction. J. of Scientific Computing 45(1), 272–293 (2010)
Lin, G., Adiga, U., Olson, K., Guzowski, J.F., Barnes, C.A., Roysam, B.: A hybrid 3D watershed algorithm incorporating gradient cues and object models for automatic segmentation of nuclei in confocal image stacks. Cytometry A 56(1), 23–36 (2003)
Mosaliganti, K., Gelas, A., Gouaillard, A., Noche, R., Obholzer, N., Megason, S.: Detection of Spatially Correlated Objects in 3D Images Using Appearance Models and Coupled Active Contours. In: Yang, G.-Z., Hawkes, D., Rueckert, D., Noble, A., Taylor, C. (eds.) MICCAI 2009. LNCS, vol. 5762, pp. 641–648. Springer, Heidelberg (2009)
Nath, S.K., Palaniappan, K., Bunyak, F.: Cell segmentation using coupled level sets and graph-vertex coloring. In: Larsen, R., Nielsen, M., Sporring, J. (eds.) MICCAI 2006. LNCS, vol. 4190, pp. 101–108. Springer, Heidelberg (2006)
Padfield, D., Rittscher, J., Thomas, N., Roysam, B.: Spatio-temporal cell cycle phase analysis using level sets and fast marching methods. Med. Image Anal. 13(1), 143–155 (2009)
Rousson, M., Deriche, R.: A Variational Framework for Active and Adaptative Segmentation of Vector Valued Images. In: Proc. Workshop Motion Video Comput., pp. 56–62. IEEE Computer Soc., Los Alamitos (2002)
Ortiz de Solorzano, C., Malladi, R., Lelievre, S., Lockett, S.J.: Segmentation of nuclei and cells using membrane related protein markers. J. Micr. 201(3), 404–415 (2001)
Wählby, C., Lindblad, J., Vondrus, M., Bengtsson, E., Björkesten, L.: Algorithms for cytoplasm segmentation of fluorescence labelled cells. Analytical Cellular Pathology 24(2), 101–111 (2002)
Yan, P., Zhou, X., Shah, M., Wong, S.T.C.: Automatic segmentation of high-throughput RNAi fluorescent cellular images. IEEE Trans. Inf. Techn. in Biomed 12(1), 109–117 (2008)
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Bergeest, JP., Rohr, K. (2011). Fast Globally Optimal Segmentation of Cells in Fluorescence Microscopy Images. In: Fichtinger, G., Martel, A., Peters, T. (eds) Medical Image Computing and Computer-Assisted Intervention – MICCAI 2011. MICCAI 2011. Lecture Notes in Computer Science, vol 6891. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-23623-5_81
Download citation
DOI: https://doi.org/10.1007/978-3-642-23623-5_81
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-23622-8
Online ISBN: 978-3-642-23623-5
eBook Packages: Computer ScienceComputer Science (R0)