Abstract
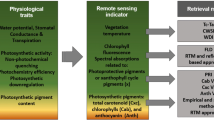
Healthy green vegetation absorbs red and blue light wavelengths preferentially for use in photosynthesis. Green light (wavelength 545–565 nm), however, is mostly reflected leading to the green appearance of living biomass. This characteristic coincides with interactions outside the visible portion of the electromagnetic spectrum. Near-infrared (NIR, wavelength 841–876 nm) light also interacts with healthy vegetation in a slightly different way. The presence of chlorophyll in green vegetation does not utilize green light due to properties of the molecules themselves and the harnessing of energy by the plant. NIR light is reflected mainly due to the physical structure of healthy leaf tissue (see Fig. 3.1 for a representation of these phenomena). These characteristics are not static in time, as plants continue to develop and transition through their life cycle; they eventually loose leaf structure and chlorophyll concentrations for many reasons including seasonal changes, disease, age, water scarcity, etc. These realities change the relationship between vegetation and light. This is particularly apparent in agricultural systems where plants undergo a predictable transition from planting to maturity and eventually senescence at the end of the growing season. This senescence is characterized mainly by a loss of chlorophyll, a collapse of leaf structure, and an investment by the plant of resources into the production of fruiting bodies and grain. These principles are the basis for much of remote sensing within agricultural ecosystems.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
3.1 Photo-Reflective Properties of Plants
Healthy green vegetation absorbs red and blue light wavelengths preferentially for use in photosynthesis. Green light (wavelength 545–565 nm), however, is mostly reflected leading to the green appearance of living biomass. This characteristic coincides with interactions outside the visible portion of the electromagnetic spectrum. Near-infrared (NIR, wavelength 841–876 nm) light also interacts with healthy vegetation in a slightly different way. The presence of chlorophyll in green vegetation does not utilize green light due to properties of the molecules themselves and the harnessing of energy by the plant. NIR light is reflected mainly due to the physical structure of healthy leaf tissue (see Fig. 3.1 for a representation of these phenomena). These characteristics are not static in time, as plants continue to develop and transition through their life cycle; they eventually loose leaf structure and chlorophyll concentrations for many reasons including seasonal changes, disease, age, water scarcity, etc. These realities change the relationship between vegetation and light. This is particularly apparent in agricultural systems where plants undergo a predictable transition from planting to maturity and eventually senescence at the end of the growing season. This senescence is characterized mainly by a loss of chlorophyll, a collapse of leaf structure, and an investment by the plant of resources into the production of fruiting bodies and grain. These principles are the basis for much of remote sensing within agricultural ecosystems.
3.1.1 Imaging Fields and Landscapes with Satellites
Satellite-based remote sensing relies on the principles of plant light reflectance previously described. Scientists can freely access data provided by the US National Aeronautics and Space Administration as well as the European Space Administration. These agencies maintain many different satellites that are capable of detecting and producing a large variety of energy and light. The majority of remote sensing relies on the detection of reflected sunlight from the Earth’s surface and has applications in many disciplines. The primary advantage of remote sensing as a technique is based on the large spatial scale inherent to a space-borne sensor. This is particularly true for environmental scientists who seek to understand large-scale processes and patterns. Typical procedures for remote sensing studies involve image acquisition from the particular agency responsible for the chosen satellite, followed by subsequent image analysis of study sites (usually done via computer coding or special software such as ArcGIS), and lastly results are analyzed and conclusions drawn.
3.1.2 Vegetation Indices
One of the primary remote sensing metrics used by environmental scientists to study nature is what is known as a vegetation index. A vegetation index is a mathematical formula that relates two or more quantities of reflected light to determine characteristics of the land surface. The first index to be developed, and arguably the most commonly used, is known as the Normalized Difference Vegetation Index (NDVI), given here, where NIR stands for near infrared and VIS for visible light, respectively:
This equation has been used for decades [1] to serve as a measure of the health of observed vegetation. This remains true today, although many improvements and alterations have been made to this basic formula over the decades.
3.2 Satellite Image Analysis
As mentioned in Sect. 2.3, the CRNS detects all forms of hydrogen within its footprint (Fig. 2.2). This includes the hydrogen contained within green growing biomass. As such, biomass water equivalent must be quantified within the footprint of any CRNS deployed in the field for proper calibration to be achieved. Studies have shown that vegetation indices derived from satellite remote sensing images can be used to reliably estimate agricultural biomass [2,3,4]. This has recently been expanded upon in the context of the CRNS calibration function. Avery et al. [2] demonstrated that the use of satellite-based remote sensing can be used to determine biomass within agricultural systems through the use of vegetation indices. It is from this study that the following procedures are derived. Satellite imaging eliminates the need for time-consuming and difficult in situ sampling campaigns. Moreover, remote sensing provides the most feasible solution for support of mobile CRNS devices to monitor soil moisture over larger areas without the need for extensive multi-field in situ sampling campaigns.
As detailed in Nguy-Robertson and Gitelson [3] and Nguy-Robertson et al. [4], the best known relationship to date between a vegetation index and actual biomass for maize and soybean as determined by in situ experiments is the Green Wide Dynamic Range Vegetation Index (GrWDRVI). Its formula is a modified version of the classic NDVI (Eq. 3.1) developed in an effort to improve the statistical relationship between satellite data and surface biomass (determined via destructive sampling) [5]. The equation is given here, note that NIR (wavelength 841–876 nm) stands for near-infrared light and Green (wavelength 545–565 nm) for green light:
The following is a step by step guide for determining standing wet biomass (to be used for determining BWE) via satellite image analysis for use in the CRNS calibration function (see Eq. 2.1). This index calculates wet biomass but not dry biomass. As such, the use of remote sensing in this publication to calculate the BWE is dependent on knowledge of crop growth stage and/or existing crop models that can give an estimate of the ratio of water mass to dry mass within the plant. It is important to note that these procedures use images produced by NASA’s Terra satellite (http://earthexplorer.usgs.gov/), specifically the 500 m resolution Moderate Resolution Imaging Spectroradiometer (MODIS).
- Step One::
-
Pick a study area.
- Step Two::
-
Navigate online to http://earthexplorer.usgs.gov/, this is the website that will provide the downloadable images from many different satellites including Terra. This website and data are free to access, but one must create an account initially.
- Step Three::
-
The website will present with a map of the Earth. This map can be navigated and examined down to a field scale to find any particular study site or sites a researcher may be interested in. Select the study site(s) by clicking on the map to place a point and then clicking another point to connect them with a line. Continue placing points until the area between the points has been created upon which your area has been delineated.
- Step Four::
-
Once your study area has been selected, press the blue “data sets” button on the bottom left. This will bring up a list of available datasets for the specific study area that was defined in step three. Navigate to the tab titled: “NASA LPDAAC Collections” and expand the drop-down list. Click the drop-down list next to the option titled: “MODIS Land Surface Reflectance.” Select the check box next to the first option: “MOD09A1” this is the global land surface reflectance taken every 8 days at a 500 m spatial scale.
- Step Five::
-
Once the data has been chosen, thumbnail images will appear on the left with the top image being the most recent. These images correspond to the days that the satellite passed over the selected study area on an 8-day rotation. To choose which days to download, simply click the “download options” button and select “HDF Format.”
- Step Six::
-
Once the data have been downloaded onto the computer, place the HDF files into a folder with an appropriate title. This will serve as a source for the computer to look for files to process.
Note: The following steps require the use of three pieces of software:
-
1.
ArcGIS, or similar image processing software (not an open-source software).
-
2.
IDLE (Integrated Development and Learning Environment): this is an open-source user interface software designed to be used with the computer coding language Python which serves as the basis for MODIS image processing in this publication. It is important to mention that other computer coding languages, imaging software, or user interfaces can be used if so desired but will likely require changes to the code or other adaptations by a qualified scientist.
-
3.
Python: this is a computer coding language that can be freely accessed via the internet (open source).
- Step Seven::
-
Once the HDF raw files are in the correct folder, the following Python code (denoted in the outlined boxes) should be entered into IDLE (windowed user interface where code is to be written); this code is very sensitive to indentations (note: this code was developed by Dr. Anthony Nguy-Robertson while at the University of Nebraska-Lincoln), and file names are merely an example and must be replaced with the names chosen in any study following this guide.
The purpose of this step is to extract the NIR and Green light into separate files from the overall reflectance data for further processing.
This code serves to identify the file location and file types to be used in the image analysis process:

This code serves to extract the individual NIR and Green light wavelengths for further processing via the GrWDRVI equation and ultimately into standing wet biomass (SWB):

This code serves to alert the user when the processing described above is successful or unsuccessful:

Note: Before the code is shown, it must be stated that a text file (.txt) must be present in the folder containing the newly processed NIR and Green reflectance files created from the previous code. This .txt file must be placed alongside these files as a reference for the next section of code to properly eliminate incidental cloud cover.
The .txt file needs to contain these numbers exactly as shown:
-
0 7 : 1
-
7 8 : 0
-
8 71 : 1
-
71 72 : 0
-
72 135 : 1
-
135 136 : 0
-
136 100000 : 1
- Step Eight::
-
Once the code outlined in step seven has been written into IDLE, and has run correctly (Python code in IDLE can be activated by clicking the drop-down box on the top left of the window and selecting “run module”), then the next piece of code can be written. This step should have its code written into a new IDLE file (not the same as the previous step seven code). The following code is intended to remove files that correspond to days with incidental cloud cover; this is for the purpose of data quality control.
This code serves to locate the recently processed files from step seven and signify where they will be placed upon the next step:

This code serves to remove files that have been downloaded corresponding to days with heavy cloud cover (this can skew the results by changing light reflectance to the satellite):

This code serves to alert the user if the process was successful via a message and a sound:

- Step Nine::
-
Once the code removing incidental cloud cover has been run, the next piece of code can be written. This code should be written in a new IDLE file separate from the previous two. The purpose of this next section of code is to process the separated NIR and Green light reflectance files that have now been corrected for days of heavy cloud cover, into the vegetation index detailed in Sect. 3.2 (Eq. 3.2).
This code serves to locate the recently processed files from step eight and signify where they will be placed upon the next step:

This code serves to calculate the GrWDRVI from the now separated NIR and Green light wavelength files created in step seven:

The index used in this publication (GrWDRVI) was developed through many comparisons of actual biomass determined via destructive sampling and biomass values determined through satellite image analysis [3, 4]. This research yielded coefficients from linear statistical relationships between the two methods that allowed for a mathematical transformation of the GrWDRVI values (between 0 and 1) into biomass (kg/m2). These biomass values are equivalent to “standing wet biomass” (SWB) and as such are used to determine biomass water equivalent for use in the CRNS calibration function (see Eq. 2.1) (Fig. 3.2).
- Step Ten::
-
Once the GrWDRVI values have been calculated, the resulting files should have been created as a TIFF image file (.tif). This file extension can be placed into image processing software such as ArcGIS in which individual GrWDRVI values will be visually represented as interlocking four-sided polygons that correspond to the spatial resolution of the satellite imagery. Here is a representation of what they look like:
- Step Eleven::
-
Locate the areas of interest in which the aforementioned polygons overlap. This is easiest to do by overlaying a basic satellite image of any particular study area. Once the appropriate areas have been located, use the “Identify” button (ArcGIS) or similar function if using any other image processing software, to identify the GrWDRVI value of each polygon that overlaps the area of interest. These numbers now can be averaged to determine the mean index value for each study site.
- Step Twelve::
-
The reflective behavior of plant material changes over the course of a growing season (see Sect. 2.1). This means that the linear relationship between biomass and the GrWDRVI changes from the beginning and peak of the growing season (denoted as “Green-Up” in this publication), to the end of the growing season (denoted as “Senescence” in this publication) [2,3,4]. Calculation of biomass must be done with separate equations to reflect the differences in each relationship. Additionally, GrWDRVI values below 0.25 are not to be used due to the fact that the biomass is too small during these growth stages and the satellite data cannot accurately predict plant biomass. Note: the equations are given here as derived from 11 years of observation from Nebraska, USA [2]:
Note: Each equation contains coefficients developed for one particular crop type, in this case maize. These coefficients change for other crops, and new datasets comparing GrWDRVI values with in situ biomass estimates must be made for each and every crop type that is included in a study. Studies conducted by Nguy-Robertson and Gitelson [3] and Nguy-Robertson et al. [4] determined that maize exhibits different statistics when it exists in a rain-fed or irrigated setting. As such, Eqs. 3.4a and 3.4b correspond to rain-fed values and irrigated values, respectively. It is important to note that work conducted by Avery et al. (2016), Nguy-Robertson et al. (2012), and Nguy-Robertson and Gitelson (2015), [2,3,4] has produced the coefficients represented in the above equations within an agricultural environment in eastern Nebraska in the United States. These values can be used for studies in similar environments but will likely need to be tailored for use in different regions.
Step Eleven: Once standing wet biomass (SWB) values have been calculated (kg/m2), they can be transformed into standing dry biomass (SDB) by either assuming fully grown maize is approximately 70–80% H2O by weight or by referring to previous estimates of biomass water weight percent determined from in situ destructive sampling performed via procedures detailed in Chap. 2. Note that maize typically dries out to 25–30% by harvest. Now, BWE can be determined via Eq. 3.1 for use in the CRNS calibration functions.
3.3 Conclusions
This section summarizes the use of remote sensing for determining agricultural crop biomass. Additionally, it details step by step instructions on how to use remote sensing data to determine biomass and subsequently biomass water equivalent for use in the CRNS calibration process. The need for accurate biomass data is important for effective use of the CRNS technology within agricultural systems. However, eliminating or minimizing the need for time-consuming in situ destructive biomass sampling campaigns is also valuable for large-scale use of the CRNS method or its mobile versions making the incorporation of remote sensing a worthwhile endeavor.
References
Rouse JW, Scheel RH, Deering DW (1974) Monitoring vegetation systems in the great plains with ERTS. In: Proceedings, 3rd Earth Resource Technology Satellite (ERTS) symposium, vol 1, p 48
Avery WA, Finkenbiner C, Franz TE, Wang T, Nguy-Robertson AL, Suyker A, Arkebauer T, Munoz-Arriola F (2016) Incorporation of globally available datasets into the roving cosmic ray neutron probe method for estimating field-scale soil water content. Hydrol Earth Syst Sci 20:3859
Nguy-Robertson AL, Gitelson AA (2015) Algorithms for estimating green leaf area index in C3 and C4 crops for MODIS, Landsat TM/ETMC, MERIS, Sentinel MSI/OLCI, and Venus sensors. Remote Sens Lett 6:1336
Nguy-Robertson AL, Gitelson A, Peng Y, Viña A, Arkebauer T, Rundquist D (2012) Green leaf area index estimation in maize and soybean: combining vegetation indices to achieve maximal sensitivity. Agron J 104:360
Gietelson AA (2004) Wide dynamic range vegetation index for remote quantification of biophysical characteristics of vegetation. J Plant Physiol 161:165
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This chapter is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
Copyright information
© 2018 International Atomic Energy Agency (IAEA)
About this chapter
Cite this chapter
Avery, W. (2018). Remote Sensing via Satellite Imagery Analysis. In: Cosmic Ray Neutron Sensing: Estimation of Agricultural Crop Biomass Water Equivalent. Springer, Cham. https://doi.org/10.1007/978-3-319-69539-6_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-69539-6_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-69538-9
Online ISBN: 978-3-319-69539-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)