Abstract
Currently, one of the most promising approaches for the treatment of oncological patients is the selective activation of T-cell antitumor immunity using immune cells. The generation of functionally active DCs in vitro constitutes a promising approach in the development of DC-based anticancer vaccines to mobilize patient defense systems, because their activation by tumor-specific antigens to induce cytotoxic responses, and their increased efficiency of antigen presentation to induce cytotoxic T lymphocytes (CTLs) via costimulatory molecules and cytokines can be controlled. The purpose of this study was to investigate the functional characteristics of peripheral blood DC subsets in colorectal cancer (CRR), breast cancer (BC), and non-small cell lung cancer (NSCLC) patients and the development of an antitumor cytotoxic response by mononuclear cells (MNCs) from patients using in vitro generated antigen-primed DCs.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
- Antigen-primed dendritic cells
- Antitumor cytotoxic response
- Colorectal cancer
- Breast cancer
- Non-small cell lung cancer
Introduction
The generation of functionally active DCs in vitro constitutes a promising approach in the development of DC-based anticancer vaccines to mobilize patient defense systems, because their activation by tumor-specific antigens to induce cytotoxic responses, and their increased efficiency of antigen presentation to induce cytotoxic T lymphocytes (CTLs) via costimulatory molecules and cytokines can be controlled [5, p. 459; 1, p. 1]. DC-based anticancer vaccines are delivered using various methods, including the use of free peptides [10, p. 1868], tumor lysates [3, p. 2827], DNA [12, p. 1339; 8, p. 122] or RNA vaccines [9, p. 1], as well as DCs primed with various tumor antigens [6, p. 57]. Because tumors have a heterogeneous structure, the surface of cells comprising the tumor bears an individual set of TAAs. The use of tumor lysate as a source of tumor immunogens has the potential advantage of stimulating a response against a variety of known and unknown TAAs typical of both a particular tumor type and a particular patient. This method enables the induction of a polyclonal immune response, stimulating both helper CD4+ and cytotoxic CD8+ immune responses, thereby reducing the risk of the tumor escaping immune surveillance [7, p. 139]. The use of a tumor lysate reduces the time and effort spent identifying and synthesizing immunodominant peptide epitopes, enabling DCs to process tumor antigens naturally.
Materials and Methods
Study subjects. Peripheral blood and tumor samples were obtained from:
-
44 patients aged 42–83 years (mean = 66.8 years) with colorectal cancer at stages I–IV [I–II–30 patients (68.2%), III–IV–14 patients (31.8%)], including 21 men (47.7%), and 23 women (52.3%). Colorectal adenocarcinoma was confirmed in all patients.
-
16 patients with the histologically verified diagnosis of non-small cell lung cancer (NSCLC) stage IIA, IIB, and IIIA as well as tumor biopsy material obtained during surgery (14 males, 2 females; mean age 60.7±1.5 years).
-
20 patients aged 35–77 years (mean age, 57.9 years) with the histologically and immunohistochemically verified diagnosis breast cancer at stages I–II.
All patients provided their informed consent to participate in this study. Patient diagnostic and histology data were kindly provided by the medical institution (City Clinical Hospital No. 1 and Novosibirsk Regional Clinical Oncology Center, Novosibirsk, Russia), where patient examination and clinical surveillance (treatment) was performed.
DC preparation. Peripheral blood MNCs were isolated using standard Ficoll-Urografin density-gradient [2, p. 97]. Cells with enhanced adherence were isolated from the resulting MNC population using a short incubation period (2 h) in a 75-cm2 culture flask, in 5% CO2 at 37 ℃. Cells were grown in RPMI-1640 complete medium supplemented with 10% fetal bovine serum (FBS), 40 μg/mL gentamicin, 200 U/mL penicillin, 2 mM L-glutamine, 5 × 10−5 M 2-mercaptoethanol, and 10 mM HEPES. The adherent MNC fraction was cultured in 48-well plates at 1 × 106 cells/mL in 0.5 mL complete medium. The adherent MNC fraction was supplemented with 50 ng/mL rhGM-CSF and 100 ng/mL rhIL-4 to produce immature DCs through a 4-day incubation. To obtain tumor antigen-loaded DCs, immature DCs were supplemented with autologous tumor cell lysate at a concentration of 100 μg/mL after 48 h of incubation. To generate DCs transfected with tumor cell RNA, we performed magnetic transfection using Promokine reagents (Germany), according to the manufacturer protocol. The culture was then supplemented with rhTNF-α (25 ng/mL) in fresh culture medium and an equivalent volume to generate mature cells over the following 24 h. The fraction of non-adherent cells was maintained in a 75-cm2 culture flask at 2 × 106 cells/mL in RPMI 1640 complete medium until decantation.
Preparation of autologous tumor cells. A tumor sample was washed in RPMI-1640 with a doubled concentration of antibiotics. To obtain autologous tumor cells, a tumor fragment from each patient was crushed and left in 0.25% trypsin solution at +4 ℃ overnight. Warm complete RPMI-1640 was used to inactivate the enzyme. The cell suspension was filtered to remove large aggregates, washed twice, and frozen in FBS with 10% DMSO (Panreacsintesis, Spain). To obtain tumor cell lysates, a tumor fragment was mechanically homogenized and the resulting suspension was successively frozen at −70 ℃ and thawed at +37 ℃ through three cycles. Cells were pelleted by centrifugation and sterilized by passing through a 0.45-μm filter. Total protein in the lysates was determined by calculating the 260/280 nm absorbance ratio using a NanoDrop device (Thermo Scientific, USA).
Co-culture of DC and MNC. Co-culture of DC and MNC was carried out in several parallel cultures for subsequent functional tests under uniform conditions. The concentration of non-adherent cultured MNC was 1 × 106 cells/ml, the DC:MNC ratio was 1:10, and rhIL-12 (10 ng/ml; PeproTech, USA) and rhIL-18 (100 ng/ml; MBL, USA) were applied to stimulate Th1-polarization. Mononuclear cells used to assess cytotoxicity against autologous tumor cells were cultured for 4 days in complete RPMI-1640 in the presence or absence of recombinant cytokines. Mononuclear cells used for assessing perforin levels were cultured in complete RPMI-1640 in the presence or absence of recombinant cytokines for 2 days; the cultures were subsequently washed of growth factors, and cultured for a further 48 h.
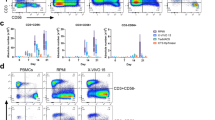
Determination of perforin-positive cell count. The cells to be analyzed were washed once with PBS and fixed with 1% paraformaldehyde in cold PBS for 20 min. They were centrifuged and the pellet was resuspended in 0.2 ml PBS containing 0.2% Tween 20 (Panreacsintesis, Spain), and incubated for 20 min to permeabilize the cell membranes, after which the cells were centrifuged and incubated with fluorochrome-labeled monoclonal antibodies against perforin (perforin-FITC, BD) for 30 min. The cells were washed to remove excess antibody and the number of positive cells was determined by flow cytometry in the lymphocyte region.
Determination of antitumor cytotoxic effect. Cytotoxicity was assessed by analyzing the lactate dehydrogenase (LDH) content in the conditioned medium obtained by co-culture of MNCs stimulated by transfected or control DCs (effector cells) and tumor cells (target cells), using a CytoTox 96® Non-Radioactive Cytotoxicity Assay (G1780, Promega, USA). The ratio effector cell: tumor cell was 10:1. Released LDH in culture supernatants is measured with a 30-min coupled enzymatic assay, which results in the conversion of a tetrazolium salt (INT) into a red formazan product. Visible wavelength absorbance data are collected using a standard 96-well plate reader. The amount of color formed is proportional to the number of lysed cells. To convert the concentration of LDH in the supernatant into percentage cytotoxicity, we applied the formula:
To determine the value OD (maximum target lysis), we lysed the tumor cells by Lysis Solution.
Statistical analysis. Statistical data were processed using the Statistica 6.0 program. The Friedman test and Newman–Keuls multiple comparison test were used to detect statistically significant differences. The Shapiro–Wilk test was applied to determine sample normality. The data are presented as the mean and standard error for normal distribution, and the median and interquartile range were used for the abnormal distribution.
Results
Cytotoxic T cells play a central role in the antitumor immune response because they directly lyse tumor cells and produce immunomodulatory cytokines, such as IL-2, TNF-α, GM-CSF, and IFN-γ, which indirectly affect malignant cells. The protective antitumor response involves the killing of tumor cells. Thus, we evaluated MNC cytotoxicity, which is activated via exposure of transfected DCs to autologous tumor cells, by quantifying the cytoplasmic LDH levels of lysed tumor cells. One of the mechanisms through which tumor cell death is triggered is the granule-dependent pathway, which is primarily mediated by perforin, granzyme, and granulysin. Perforin causes pore formation in the target cell membrane and results in cell death [13, p. 35; 14 p. 56]. To examine the role of the perforin-dependent mechanism in the lysis of tumor cells, we evaluated the effect of transfected DCs on perforin expression in co-cultures of MNCs from cancer patients.
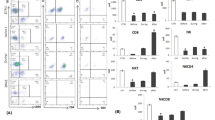
We demonstrated in patients with breast cancer that the use of lysate-activated dendritic cells or dendritic cells transfected total RNA stimulates the cytotoxic response of the MNC culture that is expressed in an increased cytotoxic activity of MNCs against autologous cells (Figs. 1a and 2). The number of perforin-positive cells increased in the MNC culture stimulated lysate-activated dendritic cells (Fig. 1b).
Cytotoxicity against autologous tumor cells (a) and percentage of perforin-positive cells (b) co-cultured of MNCs and tumor lysate-loaded DCs (n = 20) in patients with breast cancer. Data are presented as median (Me) and interquartile range. Arrows indicate statistically significant differences. (P < 0.05). MNC—the MNC control culture; MNC + DC (0)—co-culture of MNCs and DCs unloaded with tumor lysate antigens; MNC + DC (Lysate)—co-culture of MNCs and DCs loaded with tumor lysate antigens
Cytotoxicity against autologous tumor cells co-cultured of MNCs and tumor DCs, transfected total RNA (n = 16) in patients with breast cancer. Data are presented as median (Me) and interquartile range. Arrows indicate statistically significant differences. (P < 0.05). MNC—the MNC control culture; MNC + DC (0)—co-culture of MNCs and DCs unloaded with tumor antigens; MNC + DC (RNA)—co-culture of MNCs and DCs transfected with total tumor RNA
In patients with NSCLC, DCs primed with an antigen’s lysate and DCs transfected with RNA were shown to increase the cytotoxic activity of MNCs against autologous NSCLC cells, compared with the control group (MNC and MNC c-culture with non-primed DCs) (Fig. 3). In the total lymphocyte population after co-culturing lysate-primed DCs and RNA-transfected DCs increase the relative level of perforin-bearing cells in the lymphocyte population compared with control groups (Fig. 4).
The effect of antigen-primed DCs on the cytotoxic activity of MNCs against autologous NSCLC cells in vitro, n = 14. Data are presented as median (Me) and interquartile range. Arrows indicate statistically significant differences. (P < 0.05). MNC—the MNC control culture; MNC + DC (0)—co-culture of MNCs and DCs unloaded with tumor antigens; MNC + DC (Lysate)—co-culture of MNCs and DCs loaded with tumor lysate antigens; MNC + DC (RNA)—co-culture of MNCs and DCs transfected with total tumor RNA
The effect of antigen-primed DCs on the relative amount of perforin + lymphocytes in the co-culture of MNCs from NSCLC patients, n = 16. Data are presented as median (Me) and interquartile range. Arrows indicate statistically significant differences. (P < 0.05). MNC—the MNC control culture; MNC + DC (0)—co-culture of MNCs and DCs unloaded with tumor antigens; MNC + DC (Lysate)—co-culture of MNCs and DCs loaded with tumor lysate antigens; MNC + DC (RNA)—co-culture of MNCs and DCs transfected with total tumor RNA
In colorectal cancer patients, results indicate that cytotoxicity increased following use of immunogenic lysate-primed DCs, compared with all control groups. The cytotoxicity for the original MNC culture was 11.6%, whereas treatment with lysate-primed DCs increased this to 23.2% (Fig. 5).
Cytotoxic activity of colorectal cancer patients MNCs co-cultured with autologous DCs loaded with tumor lysates against autologous tumor cells (n = 17). Data are presented as median (Me) and interquartile range. Arrows indicate statistically significant differences. (P < 0.05). MNC—the MNC control culture; MNC + DC (0)—co-culture of MNCs and DCs unloaded with tumor antigens; MNC + DC (Lysate)—co-culture of MNCs and DCs loaded with tumor lysate antigens
Because tumor cell lysates contain a variety of relevant antigens, a wide range of TAAs is presented to T cells, inducing a pronounced immune response. By using complete tumor lysates in experimental models and clinical trials, high efficacy and low toxicity have been observed in various cancers, including colorectal [11, p. 6445; 4, p. 475]. However, the lysate may contain proteins suppressed the immune response [3, p. 2827] and residual tumor cells in patient organism can change their antigenic composition under the course of treatment that may also complicate the use of the lysate. We demonstrate that the use of dendritic cells to prime the lysate’s antigens enables efficient activation of the effector functions of immune cells at various cancers. Our findings indicated an elevated cytotoxicity via the perforin pathway, which was corroborated by an increase in the ratio of perforin-positive cells within the total T-cell populations in the group that received RNA-transfected and lysate-primed DCs. Thus, we can speak about the activation of the cytotoxic potential of effector cells, determined by the number of perforin-positive cells.
Conclusions
Currently, there is an intensive development of approaches to induce antitumor immunity using DC-based vaccination technologies. In our opinion, investigates should be primarily concentrated on improving to deliver antigens to DCs to stimulate antitumor immune responses. In addition, the in vitro stimulation of an antitumor immune response, which involves the selection and use of various effective stimulators and inhibitors of immunosuppressive molecules to shift the response towards a Th1 response, is also important. In our research, we demonstrated that lysate-primed DC and DC transfected total RNA enhanced MNC antitumor activity, increasing tumor cell death and the percentage of perforin-positive lymphocytes at various cancers. In this regard, we assume that the use of antigens-primed DC in vitro can activate antigen-specific T cells for modulation antitumor response.
References
Benencia, F., Sprague, L., McGinty, J., Pate, M., Muccioli, M.: Dendritic cells the tumor microenvironment and the challenges for an effective antitumor vaccination. J. Biomed. Biotechnol. 2012, 1–15 (2012)
Boyum, A.: Separation of leukocytes from blood and bone marrow. Scand. J. Clin. Lab. Invest. 21, 97 (1968)
Dong, B., Dai, G., Xu, L., Zhang, Y., et al.: Tumor cell lysate induces the immunosuppression and apoptosis of mouse immunocytes. Mol. Med. Rep. 10(6), 2827–2834 (2014)
Figdor, C.G., de Vries, I.J., Lesterhuis, W.J., Melief, C.J.: Dendritic cell immunotherapy: Mapping the way. Nature Med. 10(5), 475–480 (2004)
Fricke, I., Gabrilovich, D.I.: Dendritic cells and tumor microenvironment: A dangerous liaison. Immunol. Invest. 35, 459–483 (2006)
Janikashvili, N., Larmonier, N., Katsanis, E.: Personalized dendritic cell-based tumor immunotherapy. Immunotherapy 2(1), 57–61 (2010)
Liu, L.N., Shivakumar, R., Allen, C., Fratantoni, J.C.: Delivery of whole tumor lysate into dendritic cells for cancer vaccination. In: Li, S. (eds.) Electroporation protocols, Methods Mol. Biol. vol. 423, pp. 139–153. (2008)
Marchini, C., Kalogris, C., Garulli, C.: Tailoring DNA vaccines: designing strategies against HER2-positive cancers. Front Oncol. 3, 122 (2013)
McNamara, M.A., Nair, S.K., Holl, E.K.: RNA-based vaccines in cancer immunotherapy. J. Immunol. Res. 2015, (2015)
Peres, Lde P., da Luz, F.A., Pultz Bdos, A., Brígido, P.C., de Araújo, R.A., Goulart, L.R., Silva, M.J.: Peptide vaccines in breast cancer: the immunological basis for clinical response. Biotechnol. Adv. 33(8), pp. 1868–77. (2015)
Schnurr, M., Galambos, P., Scholz, C., et al.: Tumor cell lysate-pulsed human dendritic cells induce a T-cell response against pancreatic carcinoma cells: An in vitro model for the assessment of tumor vaccines. Cancer Res. 61, pp. 6445–50. (2001)
Ugel, S., Facciponte, J.G., De, Sanctis F., Facciabene, A.: Targeting tumor vasculature: Expanding the potential of DNA cancer vaccines. Cancer Immunol. Immunother. 64(10), 1339–1348 (2015)
Voskoboinik, I., Dunstone, M.A., Baran, K., Whisstock, J.C., Trapani, J.A.: Perforin: Structure, function, and role in human immunopathology. Immunol Rev. 235(1), pp. 35–54. (2010)
Zhou, F.: Perforin: More than just a pore-forming protein. Int. Rev. Immunol. 29(1), pp. 56–76 (2010)
Acknowledgements
Research is carried out with the financial support of the state represented by the Ministry of Education and Science of the Russian Federation. Agreement no. 14.607.21.0043. Unique identifier: RFMEFI60714X0043.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
This chapter is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
Copyright information
© 2018 The Author(s)
About this paper
Cite this paper
Cherkasov, A.P. et al. (2018). The Influence of DCs Loaded with Tumor Antigens on the Cytotoxic Response of MNC Culture Patients with Oncology. In: Anisimov, K., et al. Proceedings of the Scientific-Practical Conference "Research and Development - 2016". Springer, Cham. https://doi.org/10.1007/978-3-319-62870-7_56
Download citation
DOI: https://doi.org/10.1007/978-3-319-62870-7_56
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-62869-1
Online ISBN: 978-3-319-62870-7
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)