Abstract
The aim of this study is to evaluate and mitigate the impact of a high-speed railway (HSR) line on functional connectivity for the European tree frog (Hyla arborea), an amphibian species highly sensitive to habitat fragmentation. The method consists of modeling its ecological network using graph theory before and after the implementation of the infrastructure and of evaluating changes in connectivity. This diachronic analysis helps visualize the potential impact of the HSR line and to identify areas likely to be most affected by the infrastructure.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Among world’s vertebrates, amphibians have the highest proportion of threatened species, currently estimated at 32% by the IUCN Red List (IUCN 2016). This proportion could increase in the future because at least 42% of all amphibian species are declining in population (Stuart et al. 2008). Habitat loss, degradation and fragmentation are the greatest threats to amphibians. Fragmentation is a spatial process which affects habitat patches by decreasing their size or increasing their isolation (Fahrig 2003), making landscape less permeable to wildlife movements and gene flow (Cushman 2006; Forman and Alexander 1998). Amphibians are particularly affected because of the importance of movements during their life cycle (Joly et al. 2003). Most species occupy an aquatic habitat for breeding and during the larval period, and a terrestrial habitat after breeding and during hibernation. Daily movements and seasonal migrations across the landscape matrix connect these two types of habitat. Furthermore, many species are structured into metapopulations, in which several subpopulations occupy spatially distinct habitat patches separated by a more or less unfavorable matrix. Dispersal events allow individuals to colonize new ponds or to recolonize sites where the species is nearing extinction. A literature review about amphibian dispersal (Smith and Green 2005) showed that the median distance is less than 400 m but 7% of observed species may reach 10 km.
The major causes of landscape fragmentation are farming practices, urban development, and the construction of transportation infrastructures (Forman and Alexander 1998). Apart from direct loss of suitable habitats and road-kills, linear infrastructures cause the loss of landscape connectivity (Forman and Alexander 1998; Geneletti 2004), which is recognized as a key functional factor for the viability of species and their genetic diversity (Fahrig et al. 1995). Major infrastructures such as motorways or high-speed railway lines act as barriers to the movement of animals and isolate organisms in small subpopulations which become more sensitive to the risk of extinction (Forman and Alexander 1998). This is especially the case for populations of amphibians whose daily movements, seasonal migrations and dispersal events mean they regularly cross the landscape matrix (Allentoft and O’Brien 2010; Cushman 2006; Fahrig et al. 1995; Scherer et al. 2012).
Several case studies have contributed to identifying and quantifying the effects of linear infrastructures on species distribution in many regions of the world, using various methods. Authors have related data describing species (e.g. abundance, collisions) to proximity of infrastructures (Brotons and Herrando 2001; Fahrig et al. 1995; Huijser and Bergers 2000; Kaczensky et al. 2003; Li et al. 2010) and to the degree of habitat fragmentation (Fu et al. 2010; Serrano et al. 2002; Vos and Chardon 1998). These studies measure the real impact of the infrastructure using data on species collected after its construction. However, before the construction phase, an impact prediction stage is also necessary to compare alternative infrastructure routes (Fernandes 2000; Geneletti 2004; Vasas et al. 2009) or to guide the mitigation measures from the beginning of the project (Clauzel et al. 2015a, b; Girardet et al. 2016; Mörtberg et al. 2007; Noble et al. 2011).
Reviews by Geneletti (2006) and Gontier et al. (2006) show that the effects of landscape fragmentation are more difficult to predict than the direct loss of habitat. According to these authors, current assessment methods are often restricted to protected areas or to a narrow strip on either side of the infrastructure. However, landscape fragmentation may have consequences on a far broader scale (Forman 2000).
To assess the long-distance effects of linear infrastructures on species distributions, models must include connectivity metrics that take into account both structural (arrangement of habitat patches) and functional (behavior of the organisms) aspects. With this aim in mind, the development of methods based on graph theory in landscape modelling is promising (Dale and Fortin 2010; Urban et al. 2009). For our purposes, a graph is a set of habitat patches of a given species (called “nodes”) potentially connected by functional relationships (“links”). It provides a simplified representation of ecological networks and is considered an interesting trade-off between information content and data requirements (Calabrese and Fagan 2004). Several metrics can be computed at global level to assess connectivity for the entire graph or at local level for individual nodes or links (Galpern et al. 2011). Graph-based studies have been used to identify the key elements for connectivity (Bodin and Saura 2010; Saura and Pascual-Hortal 2007), to evaluate the importance of connectivity for species distribution (Foltête et al. 2012a; Galpern and Manseau 2013; Lookingbill et al. 2010), to assess the impact assessment of a given development on connectivity (Clauzel et al. 2013; Fu et al. 2010; Girardet et al. 2013; Gurrutxaga et al. 2011; Liu et al. 2014) or to propose mitigation measures for improving connectivity (Clauzel et al. 2015a, b; Girardet et al. 2016; Mimet et al. 2016).
In this case study, we propose to assess and to mitigate the potential impact of a HSR line on connectivity for the European tree frog (Hyla arborea) in eastern France. Populations of this species are declining in western Europe due to several causes: climate change, increased UV-radiation (Alford and Richards 1999), predation or competition, pollution and eutrophication of ponds (Borgula 1993), road-kill (Elzanowski et al. 2008), and above all the destruction and fragmentation of its habitat (Andersen et al. 2004; Cushman 2006; Vos and Stumpel 1996). In the region under study, the development of the HSR line and consecutive changes in connectivity may therefore impact the viability of tree frog populations.
The analysis consists of modeling the ecological network of the European tree frog with and without the HSR line in the landscape map. The presence of the HSR line probably entails a loss of connectivity among suitable habitats thus restricting the potential movements of the species. Comparison of the connectivity values between the two graphs provides an assessment of the changes in connectivity induced by the HSR line. The analysis is supplemented by a search for the best locations for amphibian crossing structures in order to improve connectivity. This study is based on a predictive modelling approach that estimates a potential impact but does not measure a real impact. This approach is therefore put in place before the construction of an infrastructure and allows areas potentially affected by isolation to be mapped.
Materials and Methods
Data Preparation
Study Area
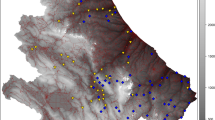
Our study was carried out in a zone of 4600 km2 in the Burgundy-Franche-Comté region of eastern France (Fig. 13.1). In this zone altitude ranges from 184 to 768 m, the landscape mosaic is dominated by forests (42% of total area), intensive agriculture (27%) and grasslands (20%). This area is strategic for environmental conservation because it contains many threatened species of birds (such as the little bustard, Tetrax tetrax), mammals (lesser horseshoe bat, Rhinolophus hipposideros), reptiles (European pond turtle, Emys orbicularis) and amphibians (European tree frog, Hyla arborea) (Paul 2011).
In December 2011, a high-speed railway line came into service in the region after 4 years of construction. It is part of a larger project that improves connections for eastern France with both Paris and the south of France. This infrastructure is 140 km long and 30 m wide on average and crosses the study area from west to east following the Ognon valley. The line includes a total 1300 m of viaducts and 2000 m of tunnels. In this study, this infrastructure is considered as impassable either because it forms a physical barrier or because traffic noise can disrupt the animals’ behaviors (Eigenbrod et al. 2009; Lengagne 2008) even when the infrastructure is on a viaduct. This simplified representation of reality is used to better predict the potential impact of the infrastructure on landscape connectivity and by repercussion on tree frog populations.
Study Species
The tree frog is widely distributed in Europe from Spain to western Russia, but its populations have declined in north-western Europe over the last 50 years (Corbett 1989). The species is classified as endangered and is on the IUCN Regional Red List of Threatened Species for Burgundy-Franche-Comté where it is mainly present in the Ognon valley (Fig. 13.1) (Pinston et al. 2000).
The tree frog has a two-phase life cycle with aquatic and terrestrial stages. The breeding pond consists of shallow and sunny ponds, marshlands, gravel-pits or river pools. Pond size does not appear decisive and ranges from 1 to 4000 m2 (Grosse and Nöllert 1993). In fact, the presence of the tree frog does not seem to be related to pond size but rather to the amount of terrestrial habitat surrounding the pond (Vos and Stumpel 1996). Although the aquatic habitat is essential for reproduction, the species spends most of its time in terrestrial habitats. In agricultural environments, the species often prefers edge habitats like ponds located on river banks, ditches, fields and forest edges (Pellet et al. 2004).
The seasonal migrations between this terrestrial habitat and the breeding pond range from 250 to 1000 m (Stumpel 1993). These dispersal events are very important in the tree frog’s life cycle. Juveniles disperse from the breeding pond into the surrounding landscape to access other ponds. Despite the fidelity of the tree frog to its breeding ground, some adults may disperse and change ponds (Fog 1993). Observed dispersal distances are generally less than 2000 m but may reach up to 4000 m (Vos and Stumpel 1996).
Landscape Data
The study required the creation of two landscape maps, the first describing the initial state before construction of the HSR line, the second including the linear infrastructure. Except for the HSR line, the land cover was identical on both maps so only the impact of the infrastructure was estimated. Different data sources were combined using ArcGis 10. The 1 m-accuracy vectorial landscape databases provided by French cartographic services (DREAL Franche-Comté, BD Topo IGN) were used to represent wetlands, hedgerows, forests, buildings, roads, railways and rivers. In agricultural areas, the French Record of Agricultural Plots were used to separate grassland and bare ground. A morphological spatial pattern analysis was also applied to the forest category to identify forest edges. All these data elements were combined into a single raster layer with a resolution of 10 m. In this raster layer, the HSR line was represented by a 3-pixel wide line to reflect its actual size. It also avoided potential discontinuities induced by the conversion of linear elements in a raster format (Adriaensen et al. 2003). Altogether, thirteen landscape categories were obtained (Table 13.1).
Landscape Graph Construction
The nodes of the graphs corresponding to the habitat patches were defined as the aquatic habitat units (breeding ponds) adjacent to an area of potential terrestrial habitat. The quality of the habitat patches, i.e. capacity, was defined as the amount of terrestrial habitat and suitable elements around ponds. The links between nodes were defined in cost distance because the ability of the tree frog to disperse depends greatly on the surrounding matrix. The ecological literature and expert opinions were thus used to classify each landscape category according to its resistance to movement (Table 13.1). Radio tracking experiments (Pellet et al. 2004; Vos and Stumpel 1996) have concluded that wooded grasslands and linear elements like hedgerows or forest edges facilitate movement and often provided the species’ terrestrial habitat. Rivers and wetlands are less favorable because their permeability depends on the density of vegetation. Conversely, grasslands, roads and railways tend to constrain movement. Finally, the cores of forest patches, bare ground, buildings and motorways are considered highly impassable and are mostly avoided by the tree frog.
Clauzel et al. (2013) performed several tests by varying the cost values and the number of classes to find the model that best explained the occurrence of the tree frog in the Franche-Comté region. The results showed that highly contrasting values between favorable and unfavorable landscape categories were the most relevant. Consequently, in the present study (Table 13.1), aquatic and terrestrial habitats were assigned a cost of 1. Suitable elements such as wetlands or wooded grasslands were assigned a low cost (10), unfavorable landscape elements a cost of 100, and highly unfavorable elements a high cost (1000). In the second landscape map including the HSR line, a cost of 10,000 was assigned to the infrastructure so as to remove any links crossing it, i.e. considering it as an absolute barrier. From the two landscape maps, without and with the HSR line, two graphs were built and thresholded at a distance of 2500 m corresponding approximately to the dispersal distance for the tree frog. This distance was selected in line with the results by Clauzel et al. (2013), where several maximum distances were tested. The model using the distance of 2500 m proved the most relevant. Consequently, only links shorter than this distance were kept.
Landscape Graph Analysis
For each graph, connectivity metrics were calculated, and then compared over time by computing their rates of variation. The magnitude of the difference between metric values provided an assessment of the impact of the HSR line. Two levels of analysis were investigated. The regional-scale analysis provided an assessment of changes on the overall connectivity throughout the study area. The local-scale analysis provided a finer assessment by identifying the most severely affected patches and corridors, i.e., those that experienced the largest changes in local connectivity or that removed by the infrastructure.
The identification of the best locations for potential wildlife crossings was based on a cumulative method developed by Foltête et al. (2014) and Girardet et al. (2016). The method consisted of testing each graph link crossed by the HSR line and to validate the one maximizing the global connectivity of the tree frog network. In the first step, all links cut by the HSR line were removed and the global connectivity was calculated. Then, an iterative process added each link and computed again connectivity. After testing all links individually, the one that produced the biggest increase in the connectivity was validated. The process was repeated until the desired number of new crossings was reached by integrating changes in the graph topology induced by the addition of previous crossings.
For all analysis, connectivity was quantifying by the Probability of Connectivity (PC) developed by Saura and Pascual-Hortal (2007). The PC index is a global metric given by the expression:
where a i and a j are the capacities of the patches i and j, \(p_{ij}^{*}\) is the maximum probability of all potential paths between patches i and j, and A is the total area under study. The maximum \(p_{ij}^{*}\) is obtained from p ij which is determined by an exponential function such that:
where d ij is the least-cost distance between these patches and α (0 < α < 1) expresses the intensity of the decrease of the dispersal probabilities resulting from this exponential function (Foltête et al. 2012a).
From the global metric PC, a patch-based metric was derived, the PCflux (Foltête et al. 2014), which is the contribution of each patch to the global PC index. For a given patch j, PCflux(j) is given by:
where a i and a j are the capacities of the patches i and j, \(p_{ij}^{*}\) is the maximum probability of all potential paths between patches i and j, and A is the total area under study.
Graphab 2.0 software (Foltête et al. 2012b) was used to construct landscape graphs and perform all analysis (Software available at http://thema.univ-fcomte.fr/productions/graphab/en-home.html).
Results
The graph modeling the ecological network of the tree frog contains 1464 nodes ranging in size from 0.01 to 86 ha (mean 0.6 ha) and 2624 links (Fig. 13.1). The network is highly fragmented with 264 components, i.e. the subparts of the graph within which the species may move from one patch to another at the dispersal scale. The largest components are located in the Ognon valley, where landscape is dominated by wetlands and grasslands. This area has the largest number of sites of this species in the region.
The diachronic analysis of the global PC value without and with the HSR line shows that the infrastructure has a low effect (−1.36%) on connectivity at the regional scale. This is explained by the already high level of fragmentation. However, as the HSR line runs through the Ognon valley, a much stronger local impact can be expected. Indeed, the implementation of the HSR line removes 61 links and let to a decline in local connectivity in 339 habitat patches (23%) mainly located in the Ognon valley (Fig. 13.2). The decrease in the PC flux values on these impacted patches is, on average, −71%, with a maximum of −99%. Some patches located more than 12 km north also experience a decline in their connectivity (about −77%).
The search algorithm tests the 61 links removed by the HSR line to select the 10 best locations for new amphibian crossing structures, i.e. those maximizing the global connectivity. The curve in Fig. 13.3 shows that the PC value increases greatly with the first two new crossing structures (+60%) and tends to gradually stabilize from the seventh one. The construction of only 4 amphibian passes is theoretically sufficient to restore 85% of the initial connectivity. These 4 first crossing structures reconnect several components to the main one along the Ognon valley. The combination of these four amphibian passes provides the greatest increase in connectivity because it concerns the largest components of the network. The other crossing structures reconnect smallest components or reinforce connections inside the main component.
Discussion
This study proposes an integrative approach to assess the potential impact of a high-speed railway line on connectivity for the European tree frog in the eastern France. The analysis can improve knowledge in the fields of environmental impact forecasting and species conservation.
The diachronic analysis of the local connectivity values shows that the impact of the HSR line ranges from a few meters to several kilometers. The impact is often located near the infrastructure but, in some sections, it may occur up to 12 km from the line. This variability is related to the landscape configuration and the initial state of the connectivity of the habitat patches. Indeed, all impacted habitat patches are into components fragmented by the HSR line. The extent of disturbance therefore depends on the size of these components, with a large component increasing the distance of the impact as in the north-east of the study area. This highlights the value of a regional-scale analysis for taking into account the long-distance effect of an infrastructure on connectivity. From this perspective, graph-based methods are interesting because they include both structural and functional aspects of connectivity. Our results are consistent with several previous studies that highlighted the importance of integrating the barrier effect in addition to the direct loss of habitat in environmental impact assessment (Clauzel et al. 2013; Forman and Alexander 1998; Fu et al. 2010; Girardet et al. 2013; Liu et al. 2014).
This graph-based approach provides an approximation of the potential impact but not a hard and fast measurement of the true impact. In order to validate our approach, these findings could be confirmed by field observations to test whether the real impact of the infrastructure is similar to that predicted by the model. In June 2011, a specific field survey was conducted in the Ognon valley to observe the tree frog presence after the construction of the HSR line. A total of 227 sites was visited with 42 presences and 185 absences. The results from this survey were compared with the connectivity changes predicted by the model. All presence points were located where there was no impact according to the model. The absence points were located in more or less affected areas, with a potential connectivity change between 0 and −90%. These survey results should be considered carefully because the Spring of 2011 was very unfavorable for the tree frog due to early drying up of water bodies. These climatic conditions could therefore explain the many points of absence of the species. Furthermore, the time delay between the construction of the HSR line and the surveys was not sufficient to assess the real impact of the infrastructure. Other field surveys should be conducted in the coming years to assess precisely the conservation status of the tree frog populations and of their habitats in the region. These surveys will also identify the causes of extinction of breeding populations, as several environmental factors may lie behind the extinction process, and be compounded to the long-distance effects of the infrastructure.
The method used to identify the best locations for new amphibian crossings goes beyond the prioritization of candidate sites developed by García-Feced et al. (2011). It is cumulative and so includes changes made to graph topology by adding previous links before searching for the next one. Graph modelling is used to include broad-scale connectivity as a criterion to be maximized, which is a key factor for the ecological sustainability of landscapes and for the viability of metapopulations (Opdam et al. 2006). In this study, the tested locations corresponded to the links, i.e. corridors potentially used by the tree frog, cut by the HSR line. Relying on the initial network of the species increases the likelihood of functional crossings because these links already connected habitat patches before the implementation of the infrastructure. In addition to cartographic results, a curve of the increased connectivity provided by new crossings is generated. This graphical tool indicates the number of potential crossings to be created in order to reach a given level of connectivity or to detect levels above which crossing creation fails to increase connectivity sufficiently. The method used here can also be applied to other species with different ecological requirements, or to other perspectives such as habitat restoration as in the study of Clauzel et al. (2015a) for the tree frog conservation.
Conclusions
The methodological approach used appears to be a handy tool for planners in forecasting the impact of linear infrastructures at different spatial scales, including the regional level, which is recognized as a gap in current methods (Fernandes 2000; Geneletti 2006; Mörtberg et al. 2007). The map of connectivity changes can help optimize the location of new protected areas or mitigation measures by identifying the areas most affected by the infrastructure. The results also provide information about the maximum distance of the impact, which is often difficult to assess. In the case of the HSR line in the Burgundy-Franche-Comté region, the environmental assessment studies focused only on a strip of 800 m on either side of the HSR line, which allowed the creation of new ponds to replace those destroyed by the construction of the infrastructure.
References
Adriaensen, F., Chardon, J. P., De Blust, G., Swinnen, E., Villalba, S., Gulinck, H., et al. (2003). The application of “least-cost” modelling as a functional landscape model. Landscape and Urban Planning, 64, 233–247.
Alford, R. A., & Richards, S. J. (1999). Global amphibian declines: A problem in applied ecology. Annual Review of Ecology and Systematics, 30, 133–165.
Allentoft, M. E., & O’Brien, J. (2010). Global amphibian declines, loss of genetic diversity and fitness: A review. Diversity, 2, 47–71.
Andersen, L. W., Fog, K., & Damgaard, C. (2004). Habitat fragmentation causes bottlenecks and inbreeding in the European tree frog (Hyla arborea). Proceedings of the Royal Society B: Biological Sciences, 271, 1293–1302.
Bodin, Ö., & Saura, S. (2010). Ranking individual habitat patches as connectivity providers: Integrating network analysis and patch removal experiments. Ecological Modelling, 221, 2393–2405.
Borgula, A. (1993). Causes of the decline in Hyla arborea. In A. H. P. Stumpel & U. Tester (Eds.), Ecology and conservation of the European tree frog. Proceedings of the 1st International Workshop on Hyla arborea (pp. 71–80). Wageningen: Institute for Forestry and Nature Research.
Brotons, L., & Herrando, S. (2001). Reduced bird occurrence in pine forest fragments associated with road proximity in a Mediterranean agricultural area. Landscape and Urban Planning, 57, 77–89.
Calabrese, J. M., & Fagan, W. F. (2004). A comparison-shopper’s guide to connectivity metrics. Frontiers in Ecology and the Environment, 2, 529–536.
Clauzel, C., Bannwarth, C., & Foltête, J.-C. (2015a). Integrating regional-scale connectivity in habitat restoration: An application for amphibian conservation in eastern France. Journal for Nature Conservation, 23, 98–107.
Clauzel, C., Girardet, X., & Foltête, J.-C. (2013). Impact assessment of a high-speed railway line on species distribution: Application to the European tree frog (Hyla arborea) in Franche-Comté. Journal of Environmental Management, 127, 125–134.
Clauzel, C., Xiqing, D., Gongsheng, W., Giraudoux, P., & Li, L. (2015b). Assessing the impact of road developments on connectivity across multiple scales: Application to Yunnan snub-nosed monkey conservation. Biological Conservation, 192, 207–217.
Corbett, K. (1989). Conservation of European reptiles and amphibians. Bromley, Kent: Helm.
Cushman, S. A. (2006). Effects of habitat loss and fragmentation on amphibians: A review and prospectus. Biological Conservation, 128, 231–240.
Dale, M. R. T., & Fortin, M.-J. (2010). From graphs to spatial graphs. Annual Review of Ecology Evolution and Systematics, 41, 21–38.
Eigenbrod, F., Hecnar, S. J., & Fahrig, L. (2009). Quantifying the road effect zone: Threshold effects of a motorway on anuran populations in Ontario, Canada. Ecology and Society, 14. http://www.ecologyandsociety.org/vol14/iss1/art24/
Elzanowski, A., Ciesiołkiewicz, J., Kaczor, M., Radwańska, J., & Urban, R. (2008). Amphibian road mortality in Europe: A meta-analysis with new data from Poland. European Journal of Wildlife Research, 55, 33–43.
Fahrig, L. (2003). Effects of habitat fragmentation on biodiversity. Annual Review of Ecology Evolution and Systematics, 34, 487–515.
Fahrig, L., Pedlar, J. H., Pope, S. E., Taylor, P. D., & Wegner, J. F. (1995). Effect of road traffic on amphibian density. Biological Conservation, 73, 177–182.
Fernandes, J. P. (2000). Landscape ecology and conservation management—Evaluation of alternatives in a highway EIA process. Environmental Impact Assessment Review, 20, 665–680.
Fog, K. (1993). Migration in the tree frog Hyla arborea. In A. H. P Stumpel & U. Tester (Eds.), Ecology and conservation of the European tree frog. Proceedings of the 1st International Workshop on Hyla arborea (pp. 55–64). Wageningen: Institute for Forestry and Nature Research.
Foltête, J.-C., Clauzel, C., & Vuidel, G. (2012a). A software tool dedicated to the modelling of landscape networks. Environmental Modelling and Software, 38, 16–32.
Foltête, J.-C., Clauzel, C., Vuidel, G., & Tournant, P. (2012b). Integrating graph-based connectivity metrics into species distribution models. Landscape Ecology, 27, 1–13.
Foltête, J.-C., Girardet, X., & Clauzel, C. (2014). A methodological framework for the use of landscape graphs in land-use planning. Landscape and Urban Planning, 124, 240–250.
Forman, R. T. T. (2000). Estimate of the area affected ecologically by the road system in the United States. Conservation Biology, 14, 31–35.
Forman, R. T. T., & Alexander, L. E. (1998). Roads and their major ecological effects. Annual Review of Ecology and Systematics, 29, 207–231.
Fu, W., Liu, S., Degloria, S. D., Dong, S., & Beazley, R. (2010). Characterizing the “fragmentation–barrier” effect of road networks on landscape connectivity: A case study in Xishuangbanna, Southwest China. Landscape and Urban Planning, 95, 122–129.
Galpern, P., & Manseau, M. (2013). Modelling the influence of landscape connectivity on animal distribution: A functional grain approach. Ecography, 36, 1004–1016.
Galpern, P., Manseau, M., & Fall, A. (2011). Patch-based graphs of landscape connectivity: A guide to construction, analysis and application for conservation. Biological Conservation, 144, 44–55.
García-Feced, C., Saura, S., & Elena-Rosselló, R. (2011). Improving landscape connectivity in forest districts: A two-stage process for prioritizing agricultural patches for reforestation. Forest Ecology and Management, 261, 154–161.
Geneletti, D. (2004). Using spatial indicators and value functions to assess ecosystem fragmentation caused by linear infrastructures. International Journal of Applied Earth Observation and Geoinformation, 5, 1–15.
Geneletti, D. (2006). Some common shortcomings in the treatment of impacts of linear infrastructures on natural habitat. Environmental Impact Assessment Review, 26, 257–267.
Girardet, X., Foltête, J. C., & Clauzel, C. (2013). Designing a graph-based approach in the landscape ecological assessment of linear infrastructures. Environmental Impact Assessment Review, 42, 10–17.
Girardet, X., Foltête, J.-C., Clauzel, C., & Vuidel, G. (2016). Restauration de la connectivité écologique: proposition méthodologique pour une localisation optimisée des passages à faune. VertigO - la revue électronique en sciences de l’environnement. http://vertigo.revues.org/17337
Gontier, M., Balfors, B., & Mörtberg, U. (2006). Biodiversity in environmental assessment—Current practice and tools for prediction. Environmental Impact Assessment Review, 26, 268–286.
Grosse, W. R., & Nöllert, A. K. (1993). Migration in the tree frog Hyla arborea. In A. H. P. Stumpel & U. Tester (Eds.), Ecology and conservation of the European tree frog. Proceedings of the 1st international workshop on Hyla arborea (pp. 37–45). Wageningen: Institute for Forestry and Nature Research.
Gurrutxaga, M., Rubio, L., & Saura, S. (2011). Key connectors in protected forest area networks and the impact of highways: A transnational case study from the Cantabrian range to the Western Alps (SW Europe). Landscape and Urban Planning, 101, 310–320.
Huijser, M. P., & Bergers, P. J. (2000). The effect of roads and traffic on hedgehog (Erinaceus europaeus) populations. Biological Conservation, 95, 111–116.
IUCN. (2016). Amphibians on the IUCN Red List. http://www.iucnredlist.org/initiatives/amphibians
Joly, P., Morand, C., & Cohas, A. (2003). Habitat fragmentation and amphibian conservation: Building a tool for assessing landscape matrix connectivity. Comptes Rendus Biologies, 326, 132–139.
Kaczensky, P., Knauer, F., Krze, B., Jonozovic, M., Adamic, M., & Gossow, H. (2003). The impact of high speed, high volume traffic axes on brown bears in Slovenia. Biological Conservation, 111, 191–204.
Lengagne, T. (2008). Traffic noise affects communication behaviour in a breeding anuran, Hyla arborea. Biological Conservation, 141, 2023–2031.
Li, Z., Ge, C., Li, J., Li, Y., Xu, A., Zhou, K., et al. (2010). Ground-dwelling birds near the Qinghai-Tibet highway and railway. Transportation Research Part D: Transport and Environment, 15, 525–528.
Liu, S., Dong, Y., Deng, L., Liu, Q., Zhao, H., & Dong, S. (2014). Forest fragmentation and landscape connectivity change associated with road network extension and city expansion: A case study in the Lancang River Valley. Ecological Indicators, 36, 160–168.
Lookingbill, T. R., Gardner, R. H., Ferrari, J. R., & Keller, C. E. (2010). Combining a dispersal model with network theory to assess habitat connectivity. Ecological Applications, 20, 427–441.
Mimet, A., Clauzel, C., & Foltête, J.-C. (2016). Locating wildlife crossings for multispecies connectivity across linear infrastructures. Landscape Ecology, 32, 1955–1973.
Mörtberg, U. M., Balfors, B., & Knol, W. C. (2007). Landscape ecological assessment: A tool for integrating biodiversity issues in strategic environmental assessment and planning. Journal of Environmental Management, 82, 457–470.
Noble, B., Hill, M., & Nielsen, J. (2011). Environmental assessment framework for identifying and mitigating the effects of linear development to wetlands. Landscape and Urban Planning, 99, 133–140.
Opdam, P., Steingröver, E., & Rooij, S. (2006). Ecological networks: A spatial concept for multi-actor planning of sustainable landscapes. Landscape and Urban Planning, 75, 322–332.
Paul, J.-P. (2011). Liste rouge des vertébrés terrestres de Franche-Comté. Groupe Naturaliste de Franche-Comté et Conseil Régional de Franche-Comté (Eds.), Besançon.
Pellet, J., Hoehn, S., & Perrin, N. (2004). Multiscale determinants of tree frog (Hyla arborea L.) calling ponds in western Switzerland. Biodiversity and Conservation, 13, 2227–2235.
Pinston H., Craney E., Pépin D., Montadert M., & Duquet M. (2000). Amphibiens et Reptiles de Franche-Comté. Atlas commenté de répartition. Groupe Naturaliste de Franche-Comté et Conseil Régional de Franche-Comté (Eds.), Besançon.
Saura, S., & Pascual-Hortal, L. (2007). A new habitat availability index to integrate connectivity in landscape conservation planning: Comparison with existing indices and application to a case study. Landscape and Urban Planning, 83, 91–103.
Scherer, R. D., Muths, E., & Noon, B. R. (2012). The importance of local and landscape-scale processes to the occupancy of wetlands by pond-breeding amphibians. Population Ecology, 54, 487–498.
Serrano, M., Sanz, L., Puig, J., & Pons, J. (2002). Landscape fragmentation caused by the transport network in Navarra (Spain). Landscape and Urban Planning, 58, 113–123.
Smith, A., & Green, D. (2005). Dispersal and the metapopulation paradigm in amphibian ecology and conservation: Are all amphibian populations metapopulations? Ecography, 28, 110–128.
Stuart, S. N., Hoffmann, M., Chanson, J. S., Cox, N. A., Berridge, R. J., Ramani, P., et al. (2008). Threatened amphibians of the world. Lynx Edicions, Barcelona, Spain; IUCN, Gland, Switzerland; and Conservation International, Arlington, VA.
Stumpel, A. H. P. (1993). The terrestrial habitat of Hyla arborea. In A. H. P. Stumpel & U. Tester (Eds.), Ecology and conservation of the European tree frog. Proceedings of the 1st international workshop on Hyla arborea (pp. 47–54). Wageningen: Institute for Forestry and Nature Research.
Urban, D. L., Minor, E. S., Treml, E. A., & Schick, R. S. (2009). Graph models of habitat mosaics. Ecology Letters, 12, 260–273.
Vasas, V., Magura, T., Jordán, F., & Tóthmérész, B. (2009). Graph theory in action: Evaluating planned highway tracks based on connectivity measures. Landscape Ecology, 24, 581–586.
Vos, C. C., & Chardon, J. P. (1998). Effects of habitat fragmentation and road density on the distribution pattern of the moor frog Rana arvalis. Journal of Applied Ecology, 35, 44–56.
Vos, C. C., & Stumpel, A. H. P. (1996). Comparison of habitat-isolation parameters in relation to fragmented distribution patterns in the tree frog (Hyla arborea). Landscape Ecology, 11, 203–214.
Acknowledgements
The research has been funded by the French Ministry of Ecology, Energy, Sustainable Development and the Sea (ITTECOP Program). The graph analysis was conducted as part of the Graphab project managed by the USR 3124 MSHE 744 Ledoux. Computations were performed on the supercomputer facilities of the MSHE Ledoux.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2017 The Author(s)
About this chapter
Cite this chapter
Clauzel, C. (2017). Evaluating and Mitigating the Impact of a High-Speed Railway on Connectivity: A Case Study with an Amphibian Species in France. In: Borda-de-Água, L., Barrientos, R., Beja, P., Pereira, H. (eds) Railway Ecology. Springer, Cham. https://doi.org/10.1007/978-3-319-57496-7_13
Download citation
DOI: https://doi.org/10.1007/978-3-319-57496-7_13
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-57495-0
Online ISBN: 978-3-319-57496-7
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)