Abstract
We report a systematic exploration of the interrelation of sound intensity, ear of entry, individual loudness judgments, and brain activity across hemispheres, using auditory functional magnetic resonance imaging (fMRI). The stimuli employed were 4 kHz-bandpass filtered noise stimuli, presented monaurally to each ear at levels from 37 to 97 dB SPL. One diotic condition and a silence condition were included as control conditions. Normal hearing listeners completed a categorical loudness scaling procedure with similar stimuli before auditory fMRI was performed. The relationship between brain activity, as inferred from blood oxygenation level dependent (BOLD) contrasts, and both sound intensity and loudness estimates were analyzed by means of linear mixed effects models for various anatomically defined regions of interest in the ascending auditory pathway and in the cortex. The results indicate distinct functional differences between midbrain and cortical areas as well as between specific regions within auditory cortex, suggesting a systematic hierarchy in terms of lateralization and the representation of sensory stimulation and perception.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
- Neural activation
- Sound pressure level
- Categorical loudness scaling
- Auditory cortex
- Auditory pathway
- Monaural stimuli
1 Introduction
Loudness is mainly the perceptual correlate of sound intensity, which is usually expressed in dB SPL as sound pressure level on a logarithmic scale. However, loudness judgments are also affected by several other variables, including physical sound parameters like e.g. duration, spectral content and temporal modulation as well many more acoustic and non-acoustic factors, including the particular task or context and personal factors like the individual hearing status (Florentine et al. 2011). While the relationship between sound intensity and neural activity in the human central auditory pathway has been extensively studied by means of neuroimaging techniques, only a small number of studies have investigated the interrelation of sound intensity, loudness and the corresponding brain activity (for a review, see Uppenkamp and Röhl 2014). Some auditory functional magnetic resonance imaging (fMRI) studies suggest that neural activation, at least in auditory cortex (AC), might be more a representation of perceived loudness rather than of physical sound pressure level (Hall et al. 2001; Langers et al. 2007; Röhl and Uppenkamp 2012). The current literature still does not provide definite answers to the following questions: (1) At what stage or stages along the auditory pathway is sound intensity transformed into its perceptual correlate (i.e. loudness)? (2) What are the functional differences across regions within AC with respect to loudness-related activation? Promoting a better understanding of the neural basis of loudness might, in the long run, improve diagnostics and treatment of hearing disorders characterized by a distorted loudness perception, e.g. loudness recruitment.
The present study is aimed at extending the current literature by providing a detailed characterization of the neural representation of sound intensity and loudness, as reflected by functional MRI. In a group of normal hearing listeners, we systematically explored the interrelation of ear of entry, sound pressure level, individual loudness and brain activation, as inferred from blood oxygenation level dependent (BOLD) contrasts, in the ascending auditory pathway and within AC.
2 Methods
2.1 Participants and Procedure
Thirteen normal hearing volunteers (aged 34 ±8 years, 4 females) participated in this study. Each participant attended two experimental sessions. In the first session, standard audiometry and an adaptive categorical loudness scaling procedure (Brand and Hohmann 2002) were performed in a sound booth. In the second session, auditory fMRI was performed while subjects were doing a simple listening task in the MRI scanner.
2.2 Stimuli
All stimuli consisted of 1/3 octave band-pass low-noise noise (Pumplin 1985) bursts at 4 kHz center frequency and were delivered via MRI compatible insert earphones (Sensimetrics S14, Sensimetrics Corporation, Malden, MA). In the loudness scaling procedure, single noise bursts with a maximum intensity of 105 dB SPL were used under left monaural, right monaural and diotic stimulus conditions. In the MRI experiment, trains of noise bursts with a total duration of 4.75 s were presented left and right monaurally at 37, 52, 67, 82 and 97 dB SPL and diotically at 82 dB SPL.
2.3 MRI Data Acquisition
Functional and structural images were acquired with a 3-Tesla MRI system (Siemens MAGNETOM Verio). Functional images were obtained using T2*-weighted gradient echo planar imaging (EPI), with a sparse temporal sampling paradigm to reduce the influence of the acoustic noise created by the scanner (Hall et al. 1999). Stimuli were presented in pseudorandomized order during 5 s gaps of scanner silence in between two successive volume acquisitions. Stimuli of each of the eleven conditions plus a silence condition, which served as baseline, were presented 36 times over the course of the experiment. For the purpose of maintaining the participants’ attention towards the acoustic stimuli, they were asked to count the number of occasionally presented deviants, characterized by a transient dip in sound level in one of the noise bursts.
2.4 Psychoacoustic Evaluation
Individual loudness judgments obtained in the scaling procedure were used to fit loudness functions for each participant by means of a recently suggested fitting method (Oetting et al. 2014). Loudness estimates for the stimulus conditions used in the MRI experiment were extracted from the individual loudness functions and were used for further analyses.
2.5 MRI Data Analysis
Standard preprocessing of the imaging data (including spatial smoothing with a 5 mm FWHM Gaussian kernel) and general linear model (GLM) estimation was done using SPM8 (Functional Imaging Laboratory, The Wellcome Department of Imaging Neuroscience, London, UK, http://www.fil.ion.ucl.ac.uk/spm/). A general linear model was set up to model the BOLD signal in every voxel as a function of ear of entry and sound level for each participant. The model included one separate regressor for each of the 11 stimulus conditions, while the silence condition was implicitly modeled as baseline. Region of interest (ROI) analyses were carried out to characterize the relationship between neural activation and sound intensity or loudness for left or right stimuli across subjects. For each participant, twelve auditory ROIs were defined based on anatomical landmarks in the individual structural images: Left and right inferior colliculus (IC), medial geniculate body (MGB), Planum temporale (PT), posterior medial (HGpm), central (HGc) and anterolateral (HGal) parts of the first Heschl’s gyrus. The average signal change from baseline of all voxels within spheres of 5 mm radius centered at individual ROI coordinates was calculated based on the regression coefficients for every stimulus condition and entered into linear mixed effects models (LMMs) with random intercepts and slopes. For each of the twelve ROIs, eight separate LMMs (2 × 2 × 2) were estimated modeling the ROI percent signal change as a linear or a quadratic function of sound intensity (expressed in dB SPL) or individual loudness (in categorical units from 0 to 50), for left or right stimuli. Model parameters were estimated by means of maximum-likelihood. Likelihood-ratio tests were conducted to assess significance of the models: First, linear models were tested against “null models” containing only the constant terms. Then, quadratic models were tested against the corresponding linear models. In both steps, models were considered significant at a level of p < 0 .05, Bonferroni-corrected. To provide measures of the models’ goodness-of-fits in terms of explanatory power, marginal R² statistics (R²m), representing that part of variance in the model explained by the fixed effects, were calculated as suggested by (Johnson 2014).
3 Results
3.1 Categorical Loudness Scaling
Figure 1 shows the results of the categorical loudness scaling procedure for the group of 13 participants. Group averaged fitted loudness curves (Oetting et al. 2014) for left monaural, right monaural and binaural stimuli are shown, along with the interindividual standard deviations of loudness estimates for the stimulus intensities presented in the MRI experiment. All three curves are characterized by a nearly linear growth of categorical loudness with sound intensity between 20 and 80 dB SPL and an increase in the steepness of the slope around 90 dB SPL. There was virtually no difference in perceived loudness between left and right ear of entry. For diotic stimulation, the expected effect of binaural loudness summation is clearly visible.
3.2 Region-of-Interest Analysis
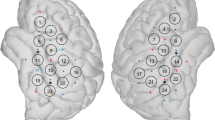
The results of the ROI analysis are illustrated in Fig. 2. R²m statistics, representing that part of variance explained by the fixed effects, display a largely symmetrical pattern across hemispheres. The highest values were found in the posterior medial parts of Heschl’s gyri, whereas the subcortical ROIs and anterolateral HG were characterized by comparatively low R²m. Throughout all investigated regions, explanatory powers of sound levels or loudness estimates were higher for contralateral as compared to ipsilateral stimuli, albeit with varying degrees of lateralization across ROIs. In all regions, linear loudness models yielded at least slightly better goodness-of-fits as compared to linear sound intensity models, although still being outmatched by quadratic fits with sound intensity in the majority of cortical ROIs. The example plots of the different model fits in the left and right posterior medial HG (Fig. 2b) reveal that the significant quadratic component is attributable to a steeper increase of BOLD responses at the highest sound levels (in this regard, the examples are representative for all ROIs). Across ROIs, only 4 (out of the total 24) quadratic loudness models reached significance, indicating that the relationship between the responses and categorical loudness is sufficiently described by a linear model.
Results of the region-of-interest (ROI) analyses. a Marginal R² (R²m) statistics, representing that part of variance explained by fixed effects, of linear and quadratic model fits with sound intensity as well as linear fits with loudness for left (L) and right (R) monaural stimuli corresponding to each auditory ROI. Non-significant models are marked with a white “X”. b Percent signal change from baseline in the left and right HGpm plotted as a function of ear of entry and sound intensity or categorical loudness. Overlaid are the fitted curves as predicted by the fixed effects of the linear (straight lines) and quadratic (dotted curves) models with their corresponding R²m statistics. Quadratic fits with loudness are not shown, since only 4 of the corresponding models reached significance across ROIs
4 Discussion
We used auditory fMRI to measure responses to unilateral narrowband noise stimuli in the human auditory system of normal hearing subjects. Based on these measures, we investigated the interrelation of ear of entry, sound pressure level, individual loudness and the corresponding brain activity by means of linear mixed effects models for a large number of distinct regions of interest in the ascending auditory pathway and in the cortex. This approach allowed us to characterize the neural representation of sound intensity and loudness in a detailed way.
4.1 Response Characteristics in Relation to Sound Intensity
Throughout all investigated stages of the auditory system, except for ipsilateral stimuli in IC bilaterally and in left MGB, neural activation as reflected by the fMRI BOLD response was significantly and positively related to physical sound intensity. Specifically, at cortical level, changes in sound pressure level were reflected by nonlinear (quadratic) increases of activation magnitude, with steeper slopes at the highest sound levels. While this is in agreement with some previous findings (Hart et al. 2002, 2003), other studies reported a more or less linear increase of BOLD signal strength with sound intensity (Hall et al. 2001; Langers et al. 2007; Röhl and Uppenkamp 2012) and still others found indications of response saturation at the highest levels (Mohr et al. 1999). To what extent these differences can be explained in terms of the use of different types of stimuli, dynamic ranges, fMRI paradigms or other factors (e.g., perceived loudness) is an interesting topic by itself, but this is not the scope of the present study. As opposed to AC, activation in the investigated subcortical structures increased predominantly linearly with increasing sound pressure level, which is in line with previous findings (Röhl and Uppenkamp 2012).
Our findings further suggest considerable differences along the auditory pathway and across auditory regions in cortex with respect to the strength of the relationship between neural activity and sound intensity. The relationship was most pronounced in the posterior medial section of HG (which includes primary AC), a little less so in PT and comparatively weak in the anterolateral sections of HG and in subcortical regions. These findings largely conform to earlier studies reporting the most robust level dependence in primary AC, but less consistent results with regard to PT and a generally weak sensitivity to level changes in more lateral and anterior regions of HG (Hart et al. 2002; Brechmann et al. 2002; Ernst et al. 2008).
4.2 Interrelation With the Ear of Entry
Activity at all investigated stages of the auditory system appeared to be more closely related to changes in sound level for stimuli presented at the contralateral ear. This is in good agreement with the anatomy and with previous investigations suggesting that auditory information crosses to the contralateral hemisphere in the lower brain stem followed by ipsilateral signal propagation towards AC (e.g., Langers et al. 2005). However, in the present study, the degree of lateralization differed considerably between regions, being particularly pronounced in the IC of the brain stem and rather weak in the posteromedial and anterolateral sections of HG. In fact, in stark contrast to the IC, changes in sound intensity were highly predictive of signal changes in HGpm irrespective of the ear of entry, pointing towards strong interhemispheric connections in the upper stages of the auditory system.
4.3 Transformation of Sound Intensity into Perceived Loudness
As expected from the strong relationship between sound intensity and loudness, a very similar pattern across auditory regions was found for the relationship between BOLD responses and individual loudness. Nonetheless, it was possible to identify distinct differences with regard to the representation of sensory stimulation and perception in the human auditory system. Specifically, while changes in sound pressure level were reflected by a quadratic growth of activation magnitude, especially in cortical areas, the relation between activation and categorical loudness can be described as predominantly linear in all investigated auditory regions. This finding, in conjunction with the similarities between the growth of categorical loudness (Fig. 1) and BOLD signal changes (Fig. 2b) with sound intensity, lends support to previous auditory fMRI studies suggesting that neural activation in AC might be more a direct linear reflection of perceived loudness rather than of physical sound pressure level (Langers et al. 2007; Röhl and Uppenkamp 2012). By contrast, in the previous study by Röhl and Uppenkamp (2012) no significant relationship between the BOLD signal strength and categorical loudness was found at the level of the IC or MGB. One interpretation in that study was that at the level of the IC, “neural activation might be more a reflection of physical sound intensity”. The current results are different, given that activity in subcortical regions was—albeit only slightly—more closely related to individual loudness as compared to sound pressure. However, the interpretation of BOLD fMRI results from these regions appears difficult, given the poor signal-to-noise ratio caused by the small size of the structures and their susceptibility to cardiac-related, pulsatile motion effects. Taking measures to improve the signal quality in subcortical regions, such as cardiac gated image acquisition (Guimaraes et al. 1998), is therefore strongly advised for future investigations.
The largely linear relationship between categorical loudness and sound intensity over the range of presented levels, as well as the fact that our participants were from a rather homogenous group of normal hearing listeners with similar loudness perception, might have limited the ability to differentiate between the contributions of both measures to neural activation in the human auditory system. A promising next step would be the inclusion of hearing impaired participants characterized by an altered relationship between sound intensity and perceived loudness (e.g., loudness recruitment). Moreover, this approach could provide additional valuable information about the nature of hearing disorders associated with distorted loudness perceptions. This in the end might open a potential for the diagnostic use of auditory fMRI. Although it is yet to prove how well the present findings pertain to the hearing impaired listeners, our results so far point towards the activation of the posterior medial section of Heschl’s gyrus as the most reliable indicator of individually perceived loudness.
4.4 Conclusion
Our findings support the notion that neural activation in auditory cortex as well as in certain stages of the ascending auditory pathway is more a direct linear reflection of perceived loudness rather than of physical sound intensity. There are, however, systematic differences between the investigated auditory regions in terms of the strength of this relationship and the degree of lateralization. We therefore suggest that functional differentiation, both between cortical and subcortical regions as well as between regions of auditory cortex, is an important issue to consider in the pursuit of a complete and comprehensive understanding of the physiological correlates of loudness perception.
References
Brand T, Hohmann V (2002) An adaptive procedure for categorical loudness scaling. J Acoust Soc Am 112:1597–1604
Brechmann A, Baumgart F, Scheich H (2002) Sound level-dependent representation of frequency modulations in human auditory cortex: a low-noise fMRI study. J Neurophysiol 87(1):423–433
Ernst SMA, Verhey JL, Uppenkamp S (2008) Spatial dissociation of changes of level and signal-to-noise ratio in auditory cortex for tones in noise. Neuroimage 43(2):321–328
Florentine M, Popper AN, Fay RR (Eds., 2011) Loudness. Springer handbook of auditory research, vol 37. Springer, New York
Guimaraes AR, Melcher JR, Baker JR, Ledden P, Rosen BR, Kiang NYS, Fullerton BC, Weisskopf RM (1998) Imaging subcortical auditory activity in humans. Hum Brain Mapp 6:33–41
Hall DA, Haggard MP, Summerfield AQ, Akeroyd MA, Palmer AR (2001) Functional magnetic resonance imaging of sound-level encoding in the absence of background scanner noise. J Acoust Soc Am 109:1559–1570
Hall DA, Summerfield AQ, Goncalves MS, Foster JR, Palmer AR, Bowtell RW (1999) “Sparse” temporal sampling in auditory fMRI. Hum Brain Mapp 7:213–223
Hart HC, Hall DA, Palmer AR (2003) The sound-level-dependent growth in the extent of fMRI activation in Heschl’s gyrus is different for low- and high-frequency tones. Hear Res 179(1–2):104–112
Hart HC, Palmer AR, Hall DA (2002) Heschl’s gyrus is more sensitive to tone level than non-primary auditory cortex. Hear Res 171(1–2):177–190
Johnson PCD (2014) Extension of Nakagawa & Schielzeth’s R²GLMM to random slope models. Meth Ecol Evol 5(9):944–946
Langers DRM, van Dijk P, Backes WH (2005) Lateralization, connectivity and plasticity in the human central auditory system. Neuroimage 28(2):490–499
Langers DRM, van Dijk P, Schoenmaker ES, Backes WH (2007) fMRI activation in relation to sound intensity and loudness. Neuroimage 35(2):709–718
Mohr CM, King WM, Freeman AJ, Briggs RW, Leonard CM (1999) Influence of speech stimuli intensity on the activation of auditory cortex investigated with functional magnetic resonance imaging. J Acoust Soc Am 105:2738–2745
Oetting D, Brand T, Ewert SD (2014) Optimized loudness-function estimation for categorical loudness scaling data. Hear Res 316:16–27
Pumplin J (1985) Low-noise noise. J Acoust Soc Am 78:100–104
Röhl M, Uppenkamp S (2012) Neural coding of sound intensity and loudness in the human auditory system. J Assoc Res Otolaryngol 13(3):369–379
Uppenkamp S, Röhl M (2014) Human auditory imaging of sound intensity and loudness. Hear Res 307:65–73
Acknowledgments
Oliver Behler is funded by the PhD programme ‘Signals and Cognition’ (Niedersächsisches Ministerium für Wissenschaft und Kultur).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
<SimplePara><Emphasis Type="Bold">Open Access</Emphasis> This chapter is distributed under the terms of the Creative Commons Attribution-Noncommercial 2.5 License (http://creativecommons.org/licenses/by-nc/2.5/) which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.</SimplePara> <SimplePara>The images or other third party material in this chapter are included in the work's Creative Commons license, unless indicated otherwise in the credit line; if such material is not included in the work's Creative Commons license and the respective action is not permitted by statutory regulation, users will need to obtain permission from the license holder to duplicate, adapt or reproduce the material.</SimplePara>
Copyright information
© 2016 The Author(s)
About this paper
Cite this paper
Behler, O., Uppenkamp, S. (2016). Auditory fMRI of Sound Intensity and Loudness for Unilateral Stimulation. In: van Dijk, P., Başkent, D., Gaudrain, E., de Kleine, E., Wagner, A., Lanting, C. (eds) Physiology, Psychoacoustics and Cognition in Normal and Impaired Hearing. Advances in Experimental Medicine and Biology, vol 894. Springer, Cham. https://doi.org/10.1007/978-3-319-25474-6_18
Download citation
DOI: https://doi.org/10.1007/978-3-319-25474-6_18
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-25472-2
Online ISBN: 978-3-319-25474-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)