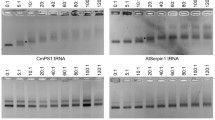
Abstract
The serpins are a family of structurally conserved protease inhibitors found mainly in the animal and plant kingdoms. This chapter will focus on the evolution of plant serpins brought to light by mining of the growing number of whole plant genomes. Our analysis shows remarkable diversification of plant serpins as compared to those in animals. Indeed, a degree of conservation between species is noted only at the level of comparison within plant orders, whereas in animals conservation of structure and function is noted at the phylum level. Within plants one serpin, containing the amino acids Leu-Arg in the P2–P1 position of the reactive center loop, is exceptional in that it is conserved in all genomes examined. The recently discovered function in programmed cell death is described for this serpin. We will further review the knowledge recently acquired on other plant serpins and their possible functions in cellular processes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ahn JW, Atwell BJ, Roberts TH (2009) Serpin genes AtSRP2 and AtSRP3 are required for normal growth sensitivity to a DNA alkylating agent in Arabidopsis. BMC Plant Biol. doi:10.1186/1471-2229-9-52
Alvarez-Alfageme F, Maharramov J, Carrillo L, Vandenabeele S, Vercammen D et al (2011) Potential use of a serpin from Arabidopsis for pest control. Plos One. doi:10.1371/journal.pone.0020278
Bateman A, Bennett HP (2009) The granulin gene family: from cancer to dementia. Bioessays 31:1245–1254
Beers EP, Jones AM, Dickerman AW (2004) The S8 serine, C1A cysteine and Al aspartic protease families in Arabidopsis. Phytochemistry 65:43–58
Beinrohr L, Harmat V, Dobo J, Lorincz Z, Gal P et al (2007) C1 inhibitor serpin domain structure reveals the likely mechanism of heparin potentiation and conformational disease. J Biol Chem 282:21100–21109
Cao J, Schneeberger K, Ossowski S, Gunther T, Bender S et al (2011) Whole-genome sequencing of multiple Arabidopsis thaliana populations. Nat Genet 43:956–963
Chen HJ, Huang DJ, Hou WC, Liu JS, Lin YH (2006) Molecular cloning and characterization of a granulin-containing cysteine protease SPCP3 from sweet potato (Ipomoea batatas) senescent leaves. J Plant Physiol 163:863–876
Coupe SA, Sinclair BK, Watson LM, Heyes JA, Eason JR (2003) Identification of dehydration-responsive cysteine proteases during post-harvest senescence of broccoli florets. J Exp Bot 54:1045–1056
Dahl SW, Rasmussen SK, Hejgaard J (1996) Heterologous expression of three plant serpins with distinct inhibitory specificities. J Biol Chem 271:25083–25088
Francis SE, Ersoy RA, Ahn JW, Atwell BJ, Roberts TH (2012) Serpins in rice: protein sequence analysis, phylogeny and gene expression during development. BMC Genomics. doi:10.1186/1471-2164-13-449
Gettins PGW (2002) Serpin structure, mechanism, and function. Chem Rev 102:4751–4803
Gu C, Shabab M, Strasser R, Wolters PJ, Shindo T et al (2012) Post-translational regulation and trafficking of the granulin-containing protease RD21 of Arabidopsis thaliana. Plos One. doi:10.1371/journal.pone.0032422
Hara-Nishimura I, Hatsugai N (2011) The role of vacuole in plant cell death. Cell Death Differ 18:1298–1304
Harrak H, Azelmat S, Baker EN, Tabaeizadeh Z (2001) Isolation and characterization of a gene encoding a drought-induced cysteine protease in tomato (Lycopersicon esculentum). Genome 44:368–374
Hatsugai N, Kuroyanagi M, Yamada K, Meshi T, Tsuda S et al (2004) A plant vacuolar protease, VPE, mediates virus-induced hypersensitive cell death. Science 305:855–858
Heit C, Jackson BC, McAndrews M, Wright MW, Thompson DC et al (2013) Update of the human and mouse SERPIN gene superfamily. Hum Genomics 7:22. doi:10.1186/1479-7364-7-22
Herron MD, Hackett JD, Aylward FO, Michod RE (2009) Triassic origin and early radiation of multicellular volvocine algae. Proc Natl Acad Sci USA 106:3254–3258
Hirst CE, Buzza MS, Bird CH, Warren HS, Cameron PU et al (2003) The intracellular granzyme B inhibitor, proteinase inhibitor 9, is up-regulated during accessory cell maturation and effector cell degranulation, and its overexpression enhances CTL potency. J Immunol 170:805–815
Huntington JA, Fan B, Karlsson KE, Deinum J, Lawrence DA et al (1997) Serpin conformational change in ovalbumin. Enhanced reactive center loop insertion through hinge region mutations. Biochemistry 36:5432–5440
Irving JA, Pike RN, Lesk AM, Whisstock JC (2000) Phylogeny of the serpin superfamily. Implications of amino acid conservation for structure and function. Genome Res 10:1845–1864
Jongsma MA, Bolter C (1997) The adaption of insects to plant protease inhibitors. J Insect Physiol 43:885–895
Jordan RE (1983) Antithrombin in vertebrate species – conservation of the heparin-dependent anticoagulant mechanism. Arch Biochem Biophys 227:587–595
Kaiserman D, Bird PI (2005) Analysis of vertebrate genomes suggests a new model for clade B serpin evolution. BMC Genomics. doi:10.1186/1471-2164-6-167
Kaiserman D, Knaggs S, Scarff KL, Gillard A, Mirza G et al (2002) Comparison of human chromosome 6p25 with mouse chromosome 13 reveals a greatly expanded ov-serpin gene repertoire in the mouse. Genomics 79:349–362
Koizumi M, Yamaguchishinozaki K, Tsuji H, Shinozaki K (1993) Structure and expression of 2 genes that encode distinct drought-inducible cysteine proteinases in Arabidopsis thaliana. Gene 129:175–182
Lampl N, Budai-Hadrian O, Davydov O, Joss TV, Harrop SJ et al (2010) Arabidopsis AtSerpin1: crystal structure and in vivo interaction with its target protease responsive to desiccation-21 (RD21). J Biol Chem 285:13550–13560
Lampl N, Alkan N, Davydov O, Fluhr R (2013) Set-point control of RD21 protease activity by AtSerpin1 controls cell death in Arabidopsis. Plant J 74:498–510
Letunic I, Bork P (2011) Interactive Tree Of Life v2: online annotation and display of phylogenetic trees made easy. Nucleic Acids Res. doi:10.1093/nar/gkr201
Luke CJ, Pak SC, Askew YS, Naviglia TL, Askew DJ et al (2007) An intracellular serpin regulates necrosis by inhibiting the induction and sequelae of lysosomal injury. Cell 130:1108–1119
Mangan MS, Kaiserman D, Bird PI (2008) The role of serpins in vertebrate immunity. Tissue Antigens 72:1–10
Ostergaard H, Rasmussen SK, Roberts TH, Hejgaard J (2000) Inhibitory serpins from wheat grain with reactive centers resembling glutamine-rich repeats of prolamin storage proteins – Cloning and characterization of five major molecular forms. J Biol Chem 275:33272–33279
Ramel F, Birtic S, Ginies C, Soubigou-Taconnat L, Triantaphylides C et al (2012) Carotenoid oxidation products are stress signals that mediate gene responses to singlet oxygen in plants. Proc Natl Acad Sci USA 109:5535–5540
Rastogi A, Yadav DK, Szymanska R, Kruk J, Sedlarova M et al (2014) Singlet oxygen scavenging activity of tocopherol and plastochromanol in Arabidopsis thaliana: relevance to photooxidative stress. Plant Cell Environ 37:392–401
Roberts TH, Hejgaard J (2008) Serpins in plants and green algae. Funct Integr Genomics 8:1–27
Silverman GA, Whisstock JC, Askew DJ, Pak SC, Luke CJ et al (2004) Human clade B serpins (ov-serpins) belong to a cohort of evolutionarily dispersed intracellular proteinase inhibitor clades that protect cells from promiscuous proteolysis. Cell Mol Life Sci 61:301–325
Vercammen D, van de Cotte B, De Jaeger G, Eeckhout D, Casteels P et al (2004) Type II metacaspases Atmc4 and Atmc9 of Arabidopsis thaliana cleave substrates after arginine and lysine. J Biol Chem 279:45329–45336
Vercammen D, Belenghi B, van de Cotte B, Beunens T, Gavigan JA et al (2006) Serpin1 of Arabidopsis thaliana is a suicide inhibitor for Metacaspase 9. J Mol Biol 364:625–636
Wang DYC, Kumar S, Hedges SB (1999) Divergence time estimates for the early history of animal phyla and the origin of plants, animals and fungi. Proc R Soc B Biol Sci 266:163–171
Wolfe KH, Gouy ML, Yang YW, Sharp PM, Li WH (1989) Date of the monocot dicot divergence estimated from chloroplast DNA-sequence data. Proc Natl Acad Sci USA 86:6201–6205
Wolpert TJ, Dunkle LD, Ciuffetti LM (2002) Host-selective toxins and avirulence determinants: What’s in a name? Annu Rev Phytopathol 40:251–285
Yoo BC, Aoki K, Xiang Y, Campbell LR, Hull RJ et al (2000) Characterization of Cucurbita maxima phloem serpin-1 (CmPS-1) – a developmentally regulated elastase inhibitor. J Biol Chem 275:35122–35128
Zhou Q, Snipas S, Orth K, Muzio M, Dixit VM et al (1997) Target protease specificity of the viral serpin CrmA. Analysis of five caspases. J Biol Chem 272:7797–7800
Acknowledgments
We wish to acknowledge the support of the Lerner Family Plant Science Research Fund and the I-CORE Program of the Planning and Budgeting Committee and the Israel Science Foundation (757/12) to R.F.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Cohen, M., Roberts, T.H., Fluhr, R. (2015). Serpins in Plants. In: Geiger, M., Wahlmüller, F., Furtmüller, M. (eds) The Serpin Family. Springer, Cham. https://doi.org/10.1007/978-3-319-22711-5_2
Download citation
DOI: https://doi.org/10.1007/978-3-319-22711-5_2
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-22710-8
Online ISBN: 978-3-319-22711-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)