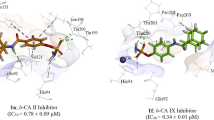
Summary
A review is presented of quantitative structure-activity relationships (QSARs) of different categories of carbonic anhydrase (CA) inhibitors, which are basically benzenesulfonamides, heterocyclic sulfonamides and aliphatic sulfonamides. The review shows that in all categories, the inhibition potency depends largely on the electronic properties of the sulfonamide group, which can be affected by the electronic characteristics of the substituents present on the nucleus (benzene or heterocyclic ring) of the sulfonamide molecules. Substituents themselves can be involved, along with the nucleus, in some dispersion interaction with the enzyme. Based on this review, a schematic model is presented to represent the interaction of sulfonamides with the CA.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Abbreviations
- AM1:
-
Austin model 1 (quantum mechanical method)
- CA:
-
carbonic anhydrase
- QSAR:
-
quantitative structure-activity relationship
- K1 :
-
enzyme inhibition constant
- k:
-
rate constant
- K:
-
binding constant
- υd :
-
14N nuclear quadrupole resonance difference frequency
- kHz:
-
kiloHertz
- IC50 :
-
inhibitor concentration leading to 50% inhibition of the enzyme
- RA:
-
relative activity
- BE:
-
binding energy
- Evdw :
-
van der Waals interaction energy
- Q:
-
atomic charge
- EH :
-
energy of highest occupied molecular orbital
- EL :
-
energy of lowest unoccupied molecular orbital
- μ:
-
dipole moment of the molecule
- μx, μy and μz :
-
components of μ in x-, y- and z-directions, respectively
- Пxx, Пyy and Пzz :
-
polarizabilities of molecule along the axes of lowest, medium and highest inertia, respectively
- П:
-
Пxx, Пyy and Пzz
- COSMO:
-
conductive shielding model
- ΔHS :
-
difference between heats of formation of vacuum and COSMO
- d1 :
-
local dipole index based on Mulliken charges
- fE :
-
frontier electron density electrophilic
- SN :
-
nucleophilic superdelocalizability index
- SE :
-
electrophilic superdelocalizability index
- Pxx, Pyy and Pzz :
-
same as Пxx, Пyy and Пzz, respectively
- Axx, Ayy and Azz :
-
lengths of the molecule in x-, y- and z-directions, respectively
- 1χv :
-
Kier’s first-order valence molecular connectivity index
- Px :
-
electrotopological state (E-State) index of an atom x
- hCA I and hCA II:
-
human CA isozymes of types I and II
- bCA IV:
-
bovine CA isozyme of type IV
- B5 :
-
Sterimol width parameter describing tha maximum width of a substitute
- Vw :
-
van der Waals volume
- Vs :
-
molar volume
- π:
-
hydrophobic constant
- σ:
-
Hammett’ electronic constant
- P:
-
octanol-water partition coefficient
- ClogP:
-
calculated logP.
- n:
-
number of data points
- r:
-
corelation coefficient
- q2 :
-
cross-validated r2 obtained by leave-one-out jackknife procedure
- s:
-
standard deviation
- Fx,y :
-
F-ratio between the variances of calculated and observed activities (x:number of independent variables; y=n−x−1)
References
C.T. Supuran, in: I. Puscas (ed.): Carbonic Anhydrase and Modulation of Physiologic Pathologic Process in the Organism. Helicon, Timisoara, Romania 1994, 29–111.
Y. Pocker and J.E. Meany: J. Am. Chem. Soc.87 1809 (1965).
Y. Pocker and J.T. Stone: J. Am. Chem. Soc.87 5497 (1965).
C. Tu, H.G Thomas, G.C. Wynns and D.N. Silverman: J. Biol. Chem.261 10100 (1986).
E.T. Kaiser and K.W. Lo: J. Am. Chem. Soc.91 4912 (1969).
L.M. Pullan and E.A. Noltman: Biochemistry24 635 (1985).
P. Henkart, G. Guidotti and J.J. Edsall: J. Biol. Chem.243 2447 (1968).
B.G. Malmstrom, P.O. Nyman, B. Strandberg and B. Tilander, in: T.W. Goodwin, J.T. Harris and B.S. Hartley (eds.): Structure and Activity of Enzymes. Academic Press, New York 1964, 121–137.
P.L. Whitney, G. Folsch, P.O. Nyman and B.G. Malmstrom: J. Biol. Chem.242 4206 (1967).
C.T. Supuran, C.W. Conroy and T.H. Maren: Proteins27 272 (1997).
F. Briganti, S. Mangani, A. Scozzafava, G. Vernaglione and C.T. Supuran: J. Biol. Inorg. Chem.4 528 (1999).
K.K. Kannan, M. Ramanadham and T.A Jones, in: R. Tashian and D. Hewett-Emmet (eds.): Biology and Chemistry Of The Carbonic Anhydrase. New York Academy of Science, New York 1984, 49–60.
A. Liljas, K.K. Kannan, P.C. Bergsten, I. Waara, K. Friedborg, B. Stranberg, U. Carlbom, L. Jarup, S. Lougren and M. Petef: Nat. New Biol.235 131 (1972).
K. Hakansson, M. Carlsson, A. Svensson and A. Liljas: J. Mol. Biol.227 1192 (1992).
A.E. Eriksson and A. Liljas: Proteins Struct. Funct. Genet.16 29 (1993).
T. Stams, S.K. Nair, T. Okuyama, A. Waheed, W.S. Sly and D.W. Christianson: Proc. Natl. Acad. Sci. USA 93, 13589 (1996).
P.A. Boriack-Sjodin, R.W. Heck, P.J. Laipis, D.N. Silverman and D.W. Christianson: Proc. Natl. Acad. Sci. USA92 10949 (1995).
A.E. Eriksson and A. Lilijas, in: S.J. Dodgson, R.E. Tashian, G. Gros and N.D. Carter (eds.): The Carbonic Anhydrases. Plenum, New York 1991, 33–48.
A. Liljas, K. Hakansson, B.H. Jonsson and Y. Xue: Eur. J. Biochem.219 1 (1994).
D.N. Silverman and S. Lindskog: Acc. Chem. Res.21 30 (1988).
T.T. Baird, A. Waheed, T. Okuyama, W.S. Sly and C.A. Fierce: Biochemistry 36, 2669 (1997).
I. Simonsson, B.H. Jonsson and S. Lindskog: Biochem. Biophys. Res. Commun.108 1406 (1982).
E Briganti, R. Pieratelli, A. Scozzafava and C.T. Supuran: Eur. J. Med. Chem.31 1001 (1996).
I. Bertini and C. Luchinat: Acc. Chem. Res.16 272 (1983).
I. Bertini, C. Luchinat and A. Scozzafava: Struct. Bonding48 45 (1982).
I. Bertini, C. Luchinat, S. Mangani and R. Pieratelli: Comments Inorg. Chem.17 1 (1995).
F. Briganti, S. Mangani, P. Orioli, A. Scozzafava, G. Vernaglione and C.T. Supuran: Biochemistry36 10384 (1997).
A.E. Eriksson, RM. Kylsten, T.A. Jones and A. Liljas: Proteins Struct. Funct. Genet. 4, 283 (1988).
S. Mangani and K. Hakansson: Eur. J. Biochem.210 867 (1992).
V. Kumar and K.K. Kannan: J. Mol. Biol.241 226 (1994).
S. Mangani and A. Liljas: J. Mol. Biol.232 9 (1993).
S.K. Nair, P.A. Ludwig and D.W. Christianson: J. Am. Chem. Soc.116 3659 (1994).
G.M. Smith, R.S. Alexander, D.W. Christianson, B.M. McKeever, G.S. Ponticello, J.P. Springer, W.C. Randall, J.J. Baldwin and C.N. Habecker: Protein Sci. 3, 118 (1994).
S.K. Nair, and D.W. Christianson: Eur. J. Biochem.213 507 (1993).
D.W. Christianson and C.A. Fierce: Acc. Chem. Res.29 331 (1996).
S. Lindskog and A. Liljas: Roum. Chem. Quart. Rev. 2243 (1994).
J.W. Kassebaum and D.N. Silverman: J. Am. Chem. Soc.111 2691 (1989).
C.K. Tu and D.N. Silverman: J. Am. Chem. Soc.108 6065 (1986)
D. Hewett-Emmett and R.E. Tashian: Mol. Phyl. Evol. 5, 50 (1996).
K.S. Smith, C. Jakubzick, T.S. Whittam and J. Ferry: Proc. Natl. Acad. Sci. USA 96, 15184 (1999).
T.H. Maren, in: F. Botré, G. Gros and B.T. Storey (eds.): Carbonic Anhydrase-From Biochemistry and Genetics to Physiology and Clinical Medicine. VCH, Weinheim 1991, 186–207.
W.R. Chegwidden, S.J. Dodgson and I.M. Spencer, in: W.R. Chegwidden, Y. Edwards and N. Carter (eds.): The Carbonic Anhydrases — New Horizons. Birkhäuser, Basel/Boston/ Berlin 2000, 343–363.
E. Cabiscol and R.L. Leuine: J. Biol.Chem.270 14742 (1995).
A.K. Parkkila, A.L. Scarim, S. Parkkila, A. Waheed, J.A. Corbett and W.S. Sly: J. Biol. Chem.273 24620 (1998).
J. Pastorek, S. Pastorekova, I. Callebaut, J.P. Mornon, V. Zelnik, R. Opaysky, M. Zatovicova, S. Liao, D. Portetelle, E.J. Stanbridge et al.: Oncogene9 2877(1994).
S. Pastorekova, S. Parkkila, A.K. Parkkila, R. Opaysky, V. Zelnik, J. Saarnio and J. Pastorek: Gastroenterology112 398 (1997).
D.A. Lovejoy, D. Hewett-Emmett, C.A. Porter, D. Cepoi, A. Shiffield, W.W. Vale and R.E. Tashian: Genomics 54, 484 (1998).
N.C.H. Bergenhem, M. Hallberg and S. Wisen: Biochem. Biophys. Acta1384 294 (1998).
O. Tureci, U. Sahin, E. Vollmar, S. Siemer, E. Gottert, G. Seitz, A.K. Parkkila, G.N. Shah, J.H. Grubb, M. Pfreundschuh et al.: Proc. Natl. Acad. Sci. USA 95, 7608 (1998).
P. Karhumaa, S. Parkkila, O. Tureci, A. Waheed, J.H. Grubb, G. Shah, A. Parkkila, K. Kaunisto, J. Tapanainen, W.S. Sly and H. Rajaniemi: Mol. Hum. Reprod. 6, 68 (2000).
K. Mori, Y. Ogawa, K. Ebihara, N. Tamura, K. Tashiro, T. Kuwahara, M. Mukoyama, A. Sugawara, S. Ozaki, I. Tanaka and K. Nakao: J. Biol. Chem.274 15701 (1999).
T.T. Baird, A. Waheed, T. Okuyama, W.S. Sly and C.A. Fierce: Biochemistry 36, 2669 (1997).
S.V. Ivanov, I. Kuzmin, M.H. Wie, S. Pack, L. Geil, B.E. Johnson, E.J. Stanbridge and M.I. Lerman: Proc. Natl. Acad. Sci. USA 95, 12596 (1998).
T.H. Maren: Physiol. Rev.47 595 (1967).
C.T. Supuran: Roum. Chem. Quart. Rev.1 77 (1993).
S. Lindskog and P.J. Wistrand, in: M. Sandler (ed.): Design of Enzyme Inhibitors. Oxford Univ. Press 1987, 698–723.
I. Puscas: Ann. N. Y. Acad. Sci.429 587 (1984).
E.R. Swenson, in: F. Botré, G. Gros and B.T. Storey (eds.): Carbonic Anhydrase-From Biochemistry and Genetics to Physiology and Clinical Medicine. VCH, Weinheim 1991, 393–403.
N. Kakeya, N. Yata and A. Kamada: Chem. Pharm. Bull. (Tokyo)17 1010 (1969).
N. Kakeya, N. Yata and A. Kamada: Chem. Pharm. Bull. (Tokyo)17 2558 (1969).
E.J. Lien, M. Hussain and G.L. Tong: J. Pharm. Sci. 59, 865 (1970).
K. Kumar, M.C. Bindal, P. Singh and S.P. Gupta: Int. J. Quantum Chem.20 123 (1981).
S.P. Gupta and Y.S. Prabhakar: J. Scient. Indus. Res.44 189 (1985).
C. Hansch, J. McClarin and R. Langridge: Mol. Pharmacol.27 493 (1985).
R.W. King and A.S.V. Burgen: Proc. R. Soc. London, B.193 107 (1976).
S. Lindskog, L.E. Henderson, K.K. Kannan, A. Liljas, P.O. Nyman and B. Strandberg, in: P.D. Boyer (ed.): The Enzymes, 3rd ed., Academic Press, New York 1971, vol. 5, 587–601.
B. Testa and W.P. Purcell: Eur. J. Med. Chem.13 509 (1978).
S.N. Subbarao and P.J. Bray: J. Med. Chem.22 111 (1979).
Y. Shinagawa and Y. Shinagawa: Int. J. Quantum Chem. Quantum Biol. Symp. No.1 169 (1974).
H.A. Krebs: Biochem. J.43 525 (1984).
K.H. Beyer, in: M. Fucks and J.H. Moyer (eds.): Edema. Saunders, Philadelphia 1960, 270–285.
A. Vedani and E.F. Meyer, Jr: J. Pharm. Sci. 73, 352, (1984).
M.C. Menziani, P.G. De Benedetti, F. Gago and W.G. Richards: J. Med. Chem.32 951 (1989).
K. Kishida and R. Manabe: Med. J. Osaka Univ.30 95 (1980).
A. Carotti, C. Raguseo, F. Compangna, R. Langridge and T.E. Klein: Quant. Struct.-Act. Relat.8 1 (1989).
A. Verloop, W. Hoogenstraaten and J. Tipker, in: E.J. Ariëns (ed.): Drug Design, vol. VII. Academic Press, New York 1976, 165–207.
P.G. De Benedetti, M.C. Menziani and C. Frassineti: Quant. Struct.-Act. Relat.4 23 (1985).
C.T. Supuran and B.W. Clare: Eur. J. Med. Chem.30 687 (1995).
B.W. Clare and C.T. Supuran: Eur. J. Med. Chem.32 311 (1997).
C.T. Supuran and B.W. Clare: Eur. J. Med. Chem. 33, 489 (1998).
C.T. Supuran and B.W. Clare: Eur. J. Med. Chem. 34, 41 (1999).
B.W. Clare and C.T. Supuran: Eur. J. Med. Chem. 34, 463 (1999).
B.W. Clare and C.T. Supuran: Eur. J. Med. Chem. 35, 859 (2000).
R.O. Roblin and J.W. Clap: J. Am. Chem. Soc.72 4890 (1950).
W.H. Miller, A.M. Dessert and R.W. Roblin: J. Am. Chem. Soc.72 4893 (1950).
G.S. Ponticello, M.B. Freedman, C.N. Habecker, P.A. Lyle, H. Schwam, S.L. Varga, M.E. Christy, W.C. Randall and J.J. Baldwin: J. Med. Chem.30 591 (1987).
C.A. Hunt, P.J. Mallorga, S.R. Michelson, H. Schwam, J.M. Sondey, R.L. Smith, M.F. Sugrue and K.L. Shephard: J. Med. Chem. 37, 240 (1994).
C.T. Supuran, A.T. Balaban, M.D. Gheorghiu, A. Schiketanz, A. Dinculescu and I. Puscas: Rev. Roum. Chim.35 399 (1990).
C.T. Supuran, G. Manole, A. Schiketanz, M.D. Gheorghiu and I. Puscas: Rev. Roum. Chim.36 251 (1991).
C.T. Supuran, A. Popescu and M.D. Banciu: Rev. Roum. Chim. 37, 289 (1992).
C.T. Supuran, G. Manole, M.A. Dinculescu, A. Schiketanz, M.D. Gheorghiu, I. Puscas and A.T. Balaban: J. Pharm. Sci.81 716 (1992).
C.T. Supuran, M.A. Ilies and A. Scozzafava: Eur. J. Med. Chem.33 739 (1998).
P.G. De Benedetti, M.C. Menziani, M. Cocchi and G. Frassineti: Quant. Struct-Act. Relat.6 51 (1987).
M.C. Menziani and P.G. De Benedetti, in: F. Botré, G. Gros and B.T. Storey (eds.): Carbonic Anhydrase-From Biochemistry and Genetics to Physiology and Clinical Medicine. VCH, Weinheim 1991, 126–129.
M.C. Menziani and P.G. De Benedetti: Struct. Chem.3 215 (1992).
M.C. Menziani and P.G. De Benedetti, in: C. Silip and A. Vittoria (eds.): QSAR-Rational Approaches to the Design of Bioactive Compounds. Elsevier, Amsterdam 1991, 331–334.
K. Kishida: Chem. Pharm. Bull. (Tokyo)26 1049, (1978).
J. Vidgren, A. Liljas and N.P.C. Walker: Int. J. Bio. Med.12 342 (1990).
J. Vidgren, A. Svensson and A. Liljas: Int. J. Bio. Med.15 97 (1993).
A. Liljas, K. Hakansson, B.H. Jonsson and Y. Xue: Eur. J. Biochem.219 1 (1994).
T.H. Maren and C.W. Conroy: J. Biol. Chem.268 26233 (1993).
A. Scozzafava and C.T. Supuran: J. Med. Chem.43 3677 (2000).
S.P. Gupta, V. Maheswaran, V. Pande and Dalip Kumar: J. Enzyme Inhibn. Med. Chem. 18, 7 (2003).
L.B. Kier and L.H. Hall: Molecular Connectivity in Chemistry and Drug Research. Academic Press, New York 1976.
L.B. Kier and L.H. Hall: J. Pharm. Sci.72 1170 (1983).
L.B. Kier and L.H. Hall: Molecular Structure Descriptors-Electrotopological State Indices. Academic Press, San Diego 1999.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2003 Springer Basel AG
About this chapter
Cite this chapter
Gupta, S.P. (2003). Quantitative structure-activity relationships of carbonic anhydrase inhibitors. In: Jucker, E. (eds) Progress in Drug Research. Progress in Drug Research, vol 60. Birkhäuser, Basel. https://doi.org/10.1007/978-3-0348-8012-1_6
Download citation
DOI: https://doi.org/10.1007/978-3-0348-8012-1_6
Publisher Name: Birkhäuser, Basel
Print ISBN: 978-3-0348-9402-9
Online ISBN: 978-3-0348-8012-1
eBook Packages: Springer Book Archive