Abstract
Measuring climate change impacts on forest ecosystems can be challenging, as many of these changes are imperceptible within the typical time scale of short-term (e.g., 3–4 years) funding of research projects. Boreal trees are notoriously imperturbable, given their tolerance to harsh conditions and their adaptability. However, the buildup of decades of warming should now translate into measurable alterations of boreal ecosystem processes. The boreal forest is host to numerous northern animals; therefore, any change in boreal forest dynamics should affect wildlife. In this chapter, we aim to provide a nonexhaustive synthesis of documented impacts of climate change on selected key processes driving boreal forest ecosystem dynamics. We focus on the themes of plant and wildlife range shifts and stand growth and death, as they are keystone parameters of boreal forest ecosystem health that are symptomatic of climate change impacts on the boreal biota. For each theme, we introduce the general concepts and processes, convey some of the limitations of current assessments, and suggest future pressing challenges.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
1 Theme 1: Plants and Wildlife Range Shifts, Expansions, and Contractions
1.1 General Concepts and Processes at Play
For decades, wildlife range shifts have fascinated biologists, biogeographers, hunters, and conservationists given the forces at play in nature that dictate the rules of wildlife–habitat relationships. Fundamentally dynamic (Laliberte & Ripple, 2004), species ranges respond to a variety of factors, including geological forcing, climate change, and the forces driving the recent and projected biodiversity decline, namely anthropogenic habitat loss, overexploitation, and invasive species (Purvis et al., 2000). Moreover, many of these drivers interact; for example, an ecosystem can show a greater vulnerability or an enhanced resistance to invasive species because of climate change (Walther et al., 2009). Understanding the factors contributing to species range shifts, contractions, or expansions is crucial not only to develop rigorous predictions of future changes in species distributions but also to implement conservation and management strategies that can slow these changes or at least mitigate the added influence of humans on flora and fauna (Laliberte & Ripple, 2004). This is especially relevant now that many authors consider human-related activities as the ultimate driver explaining worldwide range shifts, contractions, and expansions (Channell & Lomolino, 2000; Lawler et al., 2009).
In this section, we focus on range shifts driven by climate change−including the synergistic effects of other drivers of change in local biodiversity−by briefly surveying the relevant methods for studying, modeling, and predicting future shifts, expansions, and contractions in species distributions. We also discuss the limitations of these approaches and document three contrasting case studies to illustrate different scenarios. To facilitate an understanding of the mechanisms at play, we propose using the following definitions of concepts central to the study of range shifts. First, we use the term effect to refer to a change in the environment that results from a disturbance and the term impact to represent the consequences of this change for wildlife populations (Wärnbäck & Hilding-Rydevik, 2009). We also differentiate between climate and weather. According to Watson, (1963), climate refers to the interplay between solar energy, temperature, air movement, rain and snow, atmospheric humidity, and mist and fog. The seasonal pattern of change in these variables and the similar interactions from year to year constitute the climate per se in its geographical meaning. The short-term (daily, weekly) deviations from this pattern are called weather. Finally, we refer to several common concepts in ecology, e.g., physiological constraints, competition, predation, diseases, parasites, as potential mechanisms linking drivers of change, e.g., climate change, to the species of interest via their interplay with other species and components of the ecosystems.
Box 30.1 Observed Climate Change in Boreal Forest Ecosystems
North American boreal forests have experienced an average 2 °C warming since the 1950s, with the greatest warming observed in northern Canada (Zhang et al., 2015). Eurasian boreal forests are also warming rapidly, with 1.35 and 2.00 °C increases in summer and winter temperatures since 1881, respectively, and the warming rate is accelerating (Groisman & Soja, 2009). Warming patterns vary seasonally; winter temperatures have been increasing faster than summer temperatures across boreal forests, with over 4 °C warming in some areas of North America since the late 1940s. Concurrently, annual precipitation has remained constant in Eurasia, whereas precipitation increases of 5–30% have been recorded in North American boreal forests, albeit with wide spatial variations and great uncertainty (Zhang et al., 2015). Extreme temperatures have also shifted toward more extreme-warm days and fewer extreme-cold days. Such warming has lengthened the growing season by approximately two weeks over the last 30 years, concurrent with a reduction in spring snow cover. Such warming, combined with limited changes in precipitation, has led to increased heat-induced drought and a rapid degradation of the permafrost.
1.2 Brief Overview of the Methods (and Limits)
Multiple approaches are used to assess species’ vulnerability to environmental change, thereby paving the way to understand range shifts, contractions, and expansions induced by climate change or other drivers. Pacifici et al. (2015) categorized these approaches into four types: correlative, mechanistic, trait-based, and a combination of different approaches.
Correlative approaches, often referred to as species distribution models (SDMs) or ecological niche models (ENMs), are frequently used to assess the impacts of disturbances on species distribution across a geographical range within the limits of the species’ realized niche (Guisan & Thuiller, 2005). These models are supported by correlations between current species distributions and current environmental covariates; the models are then run with the predicted changes in environmental covariates to extrapolate future species distributions for a variety of taxa (Pacifici et al., 2015) and across spatial scales (Harrison et al., 2006). Although correlative models have the advantages of being spatially explicit, generally user-friendly, and adaptable to various types of data, they are also limited by their inherent correlative nature (Sinclair et al., 2010), debatable underlying assumptions, e.g., the current distribution of a species reflects an equilibrium with its environment, and the inability to capture the complexity of the biological processes driving shifts in species distributions over time.
Mechanistic models require more parameters than correlative approaches to document the behavior or mechanisms developed by organisms to cope with changing environmental conditions (Huey et al., 2012). They rely on empirical relationships linking climate parameters and demographic rates or physiological tolerances and generally focus on a single species (Deutsch et al., 2008). Mechanistic species distribution models are viewed as being more robust than correlative models for predicting species’ responses to climate change (Evans et al., 2015). These models provide insights into the fundamental niche of the climatic space that an organism can occupy rather than the realized niche of a species−the latter more commonly obtained through a correlative approach (Morin & Thuiller, 2009). Nonetheless, mechanistic niche models are limited by the need for large amounts of data, being often species-specific, and not fully accounting for the dispersal ability or biotic interactions of the modeled species.
Trait-based approaches rely on the representative biological characteristics of a species, which translate into the sensitivity and adaptability of a species to future change (Aubin et al., 2016; Moyle et al., 2013; Rowland et al., 2011). Contrary to the two abovementioned approaches, trait-based models are simpler, easier to use, and apply to multiple species. These advantages could account for the popularity of trait-based approaches among practitioners in conservation and management agencies. However, the accuracy of trait-based approaches is limited by the arbitrary selection of vulnerability thresholds for the various traits under analysis, as thresholds are based on expert opinion or observations for which environmental variations are poorly understood. This arbitrary selection can add uncertainty to predictions (Foden et al., 2013), lead to inconsistent results within species (Lankford et al., 2014), and produce incoherent comparisons between taxonomic groups when different traits are selected. Trait-based approaches are often difficult to validate, have low explanatory power, and are of limited utility for conservation and management (Angert et al., 2011).
Given these limitations, there is growing consensus that combining different types of models and data is the most suitable approach (Pacifici et al., 2015), including criteria-based approaches (Thomas et al., 2011), correlative trait–based approaches (Barbet‐Massin et al., 2012), and mechanistic–correlative approaches (Dullinger et al., 2012). The latter include the very effective dynamic-range models (Lurgi et al., 2015), which are supported by spatially explicit demographic individual-based models (McLane et al., 2011).
1.3 Case Study 1.1 The Northern Biodiversity Paradox
Although we are currently observing a worldwide loss of biodiversity, some northern regions are experiencing (or will experience) an intriguing phenomenon: an increase in local species richness. This phenomenon, called the northern biodiversity paradox, involves climate change–induced increases in local biodiversity in the northern latitudes. Given that the ranges of several species are currently limited by low temperatures, e.g., ectotherms (Araújo et al., 2006), this concept holds that climate warming will lead to a northern range shift of many species (Parmesan & Yohe, 2003).
Berteaux et al., (2018) applied climate–niche modeling, using 1961–1990 data, to assess the potential impacts of climate change on the probability of occurrence for 529 species within 1,749 protected areas spread over approximately 600,000 km2 in Québec (Canada). This extensive study area encompassed the northern limit of the distribution of several species of animals (birds and amphibians) and trees and other vascular plants. The regional climate is currently characterized by cold winters and short summers, limiting several species that are poorly adapted to the harsh winter conditions and short growing seasons. Berteaux et al.’s modeling suggested that a major species turnover is very likely within a 50- to 80-year horizon (CE 2071–2100), assuming all studied species can track their suitable climatic conditions. Depending on the specific protected area, their model projects either a relative gain in species diversity (12−530%) or relative loss (7−55%). The greatest gains are predicted for the northernmost parts of Québec’s protected area network (approximately 50°−52°N), and losses will occur mainly in the southern areas. Overall, average species richness is predicted to increase in this northeastern region of North America because of climate warming, illustrating well the northern biodiversity paradox.
Berteaux et al.’s results nonetheless suggest that the arrival of several new colonizing species in northern areas could alter the structure and functioning of northern ecosystems, as observed by others (Elmhagen et al., 2015; Foxcroft et al., 2017; Gallant et al., 2020), compromising further several at-risk species in northern environments (Alda et al., 2013). Many studies confirm that wildlife species are expanding their distribution range at their high-latitude or high-elevation (cool) margins, whereas these species’ low-latitude and low-elevation (warm) margins are retracting to higher latitudes and elevations. For example, Gallant et al., (2020) demonstrated that the impressive >1,700 km poleward range shift of the red fox (Vulpes vulpes) into the Arctic over the last century occurred both during cooling and warming climate phases; however, they showed that the highest migration rate of the red fox occurred during warmer winters. More globally, the meta-analysis conducted by Chen et al., (2011) determined that the range limits of 764 plant and animal species have moved, on average, 16.9 km northward per decade owing to climate warming. Wilson et al., (2005) highlighted range contractions along the warmer margins of distribution ranges for 16 butterflies species. The altitude rise of the butterflies’ lower elevation limit (averaging 212 m over 30 years) caused a 33% loss in suitable habitat for these taxa. Serious issues arise when the southern (warmer) edge of a range contracts faster than the northern (cooler) margin can extend (Jackson & Sax, 2010), as shown by Wiens (2016) for many taxa that were part of a survey of 976 animal and plant species. Over the long term, such an asymmetrical shift in range boundaries results in species becoming trapped by the displacement of suitable habitat conditions. This situation can counterbalance or even reverse the current regional increase in species richness observed in northern latitudes (Berteaux et al., 2018).
1.4 Case Study 1.2 Compositional Shifts in Tree Regeneration
Poleward migration in response to warming is an expected response of tree species as suitable climate conditions shift northward in the boreal forest (Périé & de Blois, 2016); however, trees and plant species require multiple generations of dispersal and a successful establishment at previously unoccupied sites before sustained range shifts can be detected. An approach for detecting range shifts is the percentile method, which links changes in species presence to latitude. This method provides evidence of range shifts when combined with broad spatial assessments of plot occupancy (gain, loss, or unchanged; Boisvert-Marsh et al., 2014). Analyzing latitudinal tree shifts across broad geographic areas requires consistent survey methods through time (Woodall et al., 2009), the precise recording of survey locations (Tingley & Beissinger, 2009), and extensive data coverage (Shoo et al., 2006). In 1970, the Québec Ministry of Forests, Wildlife and Parks established an extensive network of inventory plots south of 53°N to characterize forest resources for commercial purposes in the province. At present, four inventories have been completed across more than 6,200 permanent plots (approximately 761,000 km2), of which over 70% are located in the boreal forest. Recent studies in forest ecology have used this exceptional data set, demonstrating that tree regeneration patterns and overall community dynamics are shifting mainly at the transition between the northern temperate forest and the southern boreal forest (Boisvert-Marsh, 2020; Boisvert-Marsh et al., 2014; Duchesne & Ouimet, 2008).
Recruitment patterns of juvenile stages, e.g., saplings, can provide early evidence of shifting regeneration patterns and migration trends, revealing the biotic or abiotic factors that facilitate or hinder range shifts. Using this approach, Boisvert-Marsh (2020) detected southward latitudinal shifts between 1970 and 2015 for black spruce (Picea mariana), white spruce (Picea glauca), and balsam fir (Abies balsamea). These shifts were driven by occupancy gains into mixed temperate balsam fir–yellow birch (Betula alleghaniensis) domains and southern boreal balsam fir–white birch (B. papyrifera) domains. White birch and trembling aspen (Populus tremuloides) showed evidence of a northward latitudinal shift combined with occupancy gains toward the northern edge of the inventory area. Red maple (Acer rubrum) also showed a northward shift, increasing its presence within the southern edge of the boreal forest. Climate is not the only factor, however, that can drive such large-scale migrations in regeneration. Notably, changes to stand dynamics precipitated by disturbances elicit species turnover and can break the inertia that inhibits more southerly species from moving northward. Although there is some evidence that sugar maple (Acer saccharum) could colonize sites with conditions typical of the boreal forest (Kellman, 2004), edaphic and climatic factors interact to mitigate the extent of its northward migration, at least for now (Boisvert-Marsh & de Blois, 2021; Boisvert‐Marsh et al. 2019; Collin et al., 2018). Moderate to major disturbances are accelerating species turnover toward stands dominated by red maple, white birch, and trembling aspen (Brice et al., 2019), particularly in the southern boreal where balsam fir is common. Harvesting is linked to red maple regeneration in this area, and the expansion of red maple is aided further by its ability to recruit into plots with white birch. As expected, white birch and trembling aspen recruitment in the boreal forest has occurred into plots where black spruce or balsam fir were formerly present but that have been removed through harvesting (Brice et al., 2019). As fires, insect outbreaks, and windthrow are expected to become more frequent and intense with climate change−coupled with the northward expansion of harvesting activities−disturbances could create conditions for other temperate species to follow suit. Such changes to plant and tree communities influence the spatial arrangement and availability of wildlife habitat, e.g., resources and shelter, such that range shifts in one species can trigger shifts in others.
1.5 Case Study 1.3. Under Pressure: The Case of Boreal Populations of Woodland Caribou
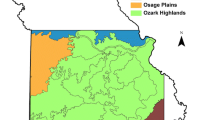
Although several studies have documented recent shifts in species’ ranges, range expansion or contraction, and projections of future range shifts related to climate change, many other species have experienced displacement by a combination of the five main drivers of biodiversity loss (listed above), in particular anthropogenic habitat loss. Across the Northern Hemisphere, we have witnessed a global decline of caribou (in North America) and reindeer (in Eurasia) subspecies, mainly driven by human-induced disturbances (Vors & Boyce, 2009). In North America, the boreal populations of woodland caribou, an ecotype of the Rangifer tarandus caribou subspecies, has been historically associated with the pan-Canadian belt of boreal and temperate forests, extending its range as far south as New England (Fig. 30.1; reviewed in Bergerud & Mercer, 1989). The current distribution range of boreal caribou is considerably reduced relative to its historical range, as this ecotype has been extirpated from much of the Maritimes, the northeastern United States, and south of the St. Lawrence River (Fig. 30.1; COSEWIC, 2014). Similar range contractions have been recorded in southwestern Canada and the northwestern United States where local populations of caribou have disappeared (Grant et al., 2019; Seip & Cichowski, 1996).

Adapted from Environment Canada (2011), permission courtesy of Environment and Climate Change Canada (ECCC)
Historical and current distribution (orange shading) of boreal caribou across the boreal ecozones (green shading) of Canada. Note that because of the lack of information on the historical distribution of boreal caribou in British Columbia, the historical southern extent in that province tracks the boreal ecozone boundary (red line).
Multiple studies have highlighted the major role of industrial activities in causing this range contraction (e.g., Rudolph et al., 2017; Schaefer, 2003; Vors & Boyce, 2009)−related to an increase in the number and efficacy of predators (e.g., Seip, 1992; Whittington et al., 2011) and maladaptive caribou behavior in human-disturbed landscapes (e.g., Lafontaine et al., 2019; Leclerc et al., 2014; Losier et al., 2015). In contrast, few studies have identified climate change as a driver of this caribou range contraction (however, see Yannic et al., (2014) for the last 21,000 years). Untangling the respective roles of past climate change and anthropogenic-related habitat loss as drivers of boreal caribou range contraction is not an easy task, as both have occurred and expanded their influence simultaneously during the last decades. Land use–related impacts, because of their significance within the boundaries of the caribou range, have received most of the research effort. Schaefer (2003), for example, related the 1880–1990 southern range contraction (34,800 km2 per decade) of caribou in Ontario to the northern advance of timber harvesting, reporting a northward range recession of 34 km per decade for the caribou; however, climate change was not linked to this shift. Such conclusions are not unique, as most recent short-term studies have struggled to isolate the effects of a changing climate as a potential explanation of caribou decline. This is not surprising considering that climate change impacts are more complex to study over the long term than human-induced disturbances. Nevertheless, a smaller (but growing) number of researchers have recently focused their efforts on distinguishing the potential impacts of climate change from the effect of anthropogenic habitat loss along the boundaries of current and future caribou distribution ranges.
Using multiple environmental suitability models, Murray et al. (2015) suggested that the distribution range of woodland caribou in the boreal forest will decrease by approximately 29–52% by 2080 under various climate change scenarios. Barber et al. (2018) applied the analytical framework of Whitman et al. (2017) to model changes to the extent of future caribou habitat in Alberta. They point out that the boreal caribou range in Alberta will experience a severe contraction under various climate change scenarios. This decrease is triggered by a marked increase in grassland vegetation by the 2080s that results in the contraction of mixedwood and coniferous forests, which are suitable habitats for caribou (following Schneider et al., 2009). This projected shift in vegetation favors an increase in white-tailed deer (Odocoileus virginianus) and its predators (gray wolf, Canis lupus; see Latham et al., 2011) and also an increased prevalence of deer-related disease, as highlighted previously (Pickles et al., 2013).
These indirect effects are all expected to compromise the long-term persistence of caribou in the boreal forest landscapes of this Canadian province (Barber et al., 2018). These examples predict coarse changes in caribou distribution through correlative or mechanistic models; however, we still lack sufficient knowledge of the fine-scale mechanisms linking the behavior and demography of caribou, predators, and their alternate prey under variable weather (e.g., Bastille-Rousseau et al., 2018; Leclerc et al., 2019). These relationships should be linked with changing climate, especially in regard to a synergy with intensive land use within the caribou range, e.g., timber harvesting, mining, and oil and gas extraction (Festa-Bianchet et al., 2011), to assist in orienting conservation efforts and ensure the persistence of this iconic species.
1.6 Future Challenges
Understanding species range shifts, expansions, and contractions has broad implications for scientists but also for politicians, industries, NGOs, and many other stakeholders. These implications range from predictions of the functioning of ecosystems, sources of food provisioning, conservation efforts for protecting currently endangered species, and the management of potentially depredating species. Researchers have provided decision-makers with various modeling tools that can support predictions of future range displacement; despite all these efforts, however, uncertainty remains and requires greater attention. Although climate change could facilitate species’ range expansion into regions where they are currently unable to survive and reproduce (Walther et al., 2009), other facets of change could limit a species’ ability to track suitable climate and habitat. Schloss et al. (2012) estimate that across the Americas, approximately 9.2% of mammals will likely be unable to keep pace with projected climate change. Among the 87% of mammalian species that will suffer range contraction, 20% will likely be limited by their dispersal capacities. The authors conclude that mammalian vulnerability to climate change may be more extensive than previously anticipated; therefore, dispersal capacities must be included in range-shift models to improve our projections of species distribution and vulnerabilities as well as our conservation efforts, thus joining conclusions obtained by other research teams (e.g., Barbet‐Massin et al., 2012; Sinclair et al., 2010).
Similarly, integrating the level of plasticity and local adaptations to our understanding of current distributions can improve future predictions (Peterson et al., 2019; Valladares et al., 2014). As our world is profoundly impacted by the human footprint—characterized by impressive levels of loss and fragmentation of natural habitats (Fahrig, 2003)—a better understanding of the interactions between these key pressures on biodiversity and climate change is urgently needed (Hof et al., 2011; Howard et al., 2020; Opdam & Wascher, 2004).
2 Theme 2: The Life and Death of Warmer Boreal Forests
2.1 General Concepts and Processes at Play
Shifts in demographic indices, e.g., growth and mortality rates, are major indicators of changes in stand health (Berdanier & Clark, 2016), whereas the relationship between these indices and environmental drivers yields information on the capacity of species to cope with climate change (Buechling et al., 2017; Foster et al., 2016). In this sense, changes in demographic performance indices may be easier to detect than actual range shifts, which require the local extirpation of all individuals or a marked migratory movement (Vanderwel & Purves, 2014).
All trees found in the boreal forest ecosystem must confront the challenge of surviving long, cold winters and then reacting quickly to the ticking clock during the brief warm summer when species must complete their life cycle. Therefore, a reasonable expectation is that boreal plants should thrive under a warmer climate. Warming has been shown to release some constraints, such as low soil fertility and the short growing season, imposed by harsh climates (D’Orangeville et al., 2014; Myneni et al., 1997; Peñuelas et al., 2009). However, these benefits may be outweighed by the increased metabolic cost to the plant under warmer, drier conditions; for example, an earlier spring increases summer drought stress (see Buermann et al., 2018) and frost damage (Marquis et al., 2020). Species may also struggle to maintain their competitive fitness under a warmer climate (Clark et al., 2014) and intensified disturbance regime (Gauthier et al., 2015).
Under controlled conditions, some physiological processes of boreal tree species can tolerate warming as long as the tree has access to sufficient resources to sustain the co-occurring increase in metabolic cost. For example, a mechanistic model calibrated with physiological data demonstrated that the growth of black spruce is optimal at temperatures found at its southern range limit; however, at these southern latitudes, the tree requires much greater amounts of resources (Bonan & Sirois, 1992). Nonetheless, other life cycle processes of boreal tree species, such as bud break or seed production, may be poorly adapted to warming. As competition for water, nutrients, light, and space resources is already the main driver of closed-crown forest mortality and growth (Franklin et al., 1987; Oliver & Larson, 1996), climate-related shifts in resource availability, e.g., water, have the potential to radically transform a species’ competitive fitness, with the specific consequences dependent on stand composition, structure, and density (Clark et al., 2016). Notably, warming-induced drought stress can halt photosynthesis and deplete carbohydrate reserves in trees, thus reducing carbon allocation to growth or defensive compounds (Anderegg et al., 2015; Waring, 1987). In turn, these weakened defense mechanisms, coupled with warming-induced shifts in the range limits of certain pests and pathogens, can heighten a tree species’ susceptibility to secondary stressors and damage from insect outbreaks (Anderegg et al., 2015; Kurz et al., 2008; Navarro et al., 2018). Drought-induced tree mortality often takes years or decades to occur, and this slow drought-imposed trajectory to tree death has been referred to as the death spiral (Franklin et al., 1987; Manion, 1991).
2.2 Brief Overview of Methods (and Limits)
Three data streams are commonly used to monitor how boreal forest ecosystems adjust to ongoing climate change: tree-ring records, remote sensing information, and permanent sample-plot data (Marchand et al., 2018). By matching annual tree-ring width to its year of formation, we can turn back time and reconstruct growth trends by relating ring width to climate (Girardin et al., 2016). Nonetheless, the usual lack of concurrent information on past changes in stand structure−the fading record problem−can introduce considerable bias when linking long-term, i.e., several decades, growth changes to climate when such growth changes can also be due to variations in stand density and stand development (Swetnam et al., 1999). Although remote sensing can capture information over quite large areas, several trade-offs for this broad coverage remain, including a coarse spatial and temporal resolution and the challenge of relating productivity indices, such as the Normalized Difference Vegetation Index, to specific ecosystem processes (National Academies of Sciences, Engineering, and Medicine, 2019). Permanent sample plots (PSP) provide a coarser time resolution than tree-ring records; nonetheless, they offer an exhaustive record of all changes in growth and mortality within a given plot over time. Another approach is to use snapshot data, i.e., a single measurement in time, to measure impacts among a diversity of forest stands following climatic anomalies (Michaelian et al., 2011).
Impacts from extreme climatic events are easier to detect than long-term, gradual climate change–driven shifts in stand demographics. Stand demographics vary naturally with stand development following stand-replacing disturbances such as fire or forest management (Lutz & Halpern, 2006). These processes may further be affected by secondary stressors, e.g., pests/pathogens, which are often difficult to detect. Researchers are faced with the challenging task of assessing the interactions between climate and stand processes or controlling for all these driving factors, sometimes leading to the exclusion of more than 95% of initial study plots; these issues raise the question of the representativeness of the obtained conclusions (Ma et al., 2012; Peng et al., 2011).
2.3 Case Study 2.1 Mortality in the Boreal Forest of North America
Here, we review the recent mortality trends and pulses for boreal forest trees of North America and the role of climate change in explaining these patterns. The 2001−2002 drought that affected boreal aspen stands in western Canada offers a striking glimpse of the possible impact of future climate anomalies on northern forest health. Precipitation was halved that year, and extensive mortality−up to 80% in some stands−quickly followed in this water-limited boreal ecosystem (Hogg et al., 2008; Michaelian et al., 2011). Climate warming has also been related to the unprecedented severity of recent insect outbreaks. The most recent mountain pine beetle (Dendroctonus ponderosae) outbreak in western Canada was ten times larger than all previous recorded outbreaks (Kurz et al., 2008), related to a combination of warming, which enabled the survival of the insect outside its typical range, and recent drought, which weakened the host trees (Taylor et al., 2006). By 2013, 53% of all merchantable pine in British Columbia had been killed by the insect (Walton, 2013). This deadly combination of warmer temperatures and drought similarly favored the expansion of the spruce beetle (D. rufipennis) into colder areas of Alaska’s boreal forest (Berg et al., 2006). The punctual nature of insect outbreaks makes it difficult to identify warming-associated trends over time; however, the robust correlations between temperature and outbreak events provide a compelling case for a climate influence on both spruce (Berg et al., 2006) and mountain pine beetle (Logan & Powell, 2001). Similar conclusions can be reached regarding forest fires. Temperature is one of the best predictors of long-term trends of area burned (Flannigan et al., 2005); thus, unsurprisingly, annual burned forest areas have increased markedly to 2.5 million ha·yr−1 since the 1970s, closely tracking regional human‐induced warming (Gillett et al., 2004). There is, however, marked regional variability in some regions of Canada, particularly in eastern Canada, which shows a decrease in annual area burned (Hanes et al., 2019).
Whereas linkages between climate change and disturbance-induced mortality are unanimously supported by the peer-reviewed literature, the exact role of climate change on mortality within undisturbed boreal stands remains more tenuous. The monitoring of permanent sample plots has revealed a threefold increase in mortality rates across western boreal North America since the 1950s (e.g., Hember et al., 2017; Luo & Chen, 2013, 2015; Peng et al., 2011; Thorpe & Daniels, 2012; Zhang et al., 2015). In comparison, eastern Canada shows no strong evidence of increasing mortality rates, from a weak 0.2% increase in annual mortality (Peng et al., 2011) to no change at all (Ma et al., 2012). Is climate change linked to this recent increase in mortality? In the cool and wet foothills of west-central Alberta, Thorpe and Daniels, (2012) could not detect any relationship between climate and increasing mortality rates. Rather, stand development processes, mainly tree size and basal area, appear to drive these mortality increases. In drier boreal forests, the water deficit displays only a weak covariation with long-term mortality trends (Luo & Chen, 2015; Peng et al., 2011; Zhang et al., 2015). Given the low temperatures observed in boreal forests, if a water deficit is not predominant, warming effects could be affecting multiple stand processes, such as growth or competition, thereby affecting our ability to establish clear causal relationships. For instance, analysis of large-scale tree-ring collections and exceptionally old trees has established that higher growth rates reduce tree longevity (Black et al., 2008; Di Filippo et al., 2015). Such concomitant increases in growth and mortality rates in western Canada have been observed by some researchers (Chen & Luo, 2015; Luo & Chen, 2015; Searle & Chen, 2018) but not all (Ma et al., 2012; Zhang et al., 2015). If true, this would support the hypothesis of a temperature-driven acceleration of stand developmental processes, potentially related to improved water-use efficiency with increasing CO2 (Giguère-Croteau et al., 2019), a longer growing season (D’Orangeville et al., 2016, 2018), or increased microbial activity in soils releasing more nutrients (D’Orangeville et al., 2014). Similarly, joint increases in competition and mortality over time have also been reported, despite limited evidence for their interaction with climate (Luo & Chen, 2013; Zhang et al., 2015). Given the control exerted by competition over tree growth response to climate (Clark et al., 2011; Ford et al., 2017), our poor understanding of the mechanisms behind this increase in competition over time is astonishing (Price et al., 2015). Uncovering these mechanisms will require the acquisition of species-specific demographic response curves to determine the interactive effects of warming and drying.
2.4 Case Study 2.2 Recent Growth Trends in Asian Boreal Forests
Whereas Scots pine (Pinus sylvestris) is associated with the southern boreal forests of Asia, Dahurian larch (Larix gmelinii) dominates the northern forests, where its growth is restricted by the presence of permafrost. The high productivity reported for larch trees growing on upland sites is contrasted by the extremely low productivity of this species on permafrost plains and wetlands (Gauthier et al., 2015). Regional climate warming, via the thawing of permafrost to greater depths, is triggering complex shifts in tree growth. Larch has been experiencing large increases in growth on the plains (Fig. 30.2; Zhang et al., 2019a) and decreased growth in wetlands (Juřička et al., 2020). The positive growth response observed on permafrost plains could be transitory, however, as climate warming is likely to convert some areas of the permafrost plains into waterlogged wetlands. These results stress the important role of microtopography and permafrost type for predicting future larch tree growth in the region.
In the southern boreal forests of Asia, Scots pine also displays similarly contrasted growth responses to warming. Although previous studies observed a negative effect of temperature on tree growth across most Scots-pine populations (e.g., Reich & Oleksyn, 2008), increased growth has been reported for the northern part of boreal Asian Scots-pine forests (Zhang et al., 2019b). In this region, the recent rapid warming is advancing the growth onset sufficiently to overlap temporally with the snowmelt period; this overlap allows Scots pine to benefit from the warmer climate and have new access to an additional water resource. Hence, these Scots-pine forests may be in a unique position of withstanding or even benefiting, at least temporarily, from the current rise in temperatures.
Birch (Betula pendula), Siberian fir (Abies sibirica), and Norway spruce (Picea abies) are important pioneer and accompanying species in southern Asian boreal forests. Warming-induced extensions of the growing season have heightened the growth of birch forests of western Siberia. In contrast, the same species has experienced a decline in the drier regions of the Trans-Baikal forest–steppe ecotone because of warming-induced water stress (Kharuk et al., 2014). Using a network of 34 tree-ring chronologies for Siberian larch (Larix sibirica) and Siberian pine (Pinus sibirica) in the Altai mountains of central Asia, Kang et al. (2021) suggested that tree radial growth in the region may decline with future projected climate change. Similarly, drought stress appears to drive a negative growth response for fir (Kharuk et al., 2017) and Norway spruce (Kharuk et al., 2015) in other arid parts of the Asian boreal forest; this pattern contrasts with the trend of increased growth found in the colder northern regions (Schaphoff et al., 2016). The current decline in arid Asian boreal forest species is likely to turn the Eurasian carbon sink into a source by 2100 (Kicklighter et al., 2014).
2.5 Future Challenges
The effective management of boreal forest ecosystems to minimize tree growth decline and mortality losses under rapidly warming global temperatures and increased biotic disturbances−including pests and pathogens−is a key challenge for the future. Our ability to develop effective management strategies is limited by a lack of formal understanding of the potentially strong interactive effects of climate change, insects, and disease (Allen et al., 2015; Anderegg et al., 2015). If we are to maintain the health, vigor, and ecosystem services provided by the boreal forest, we must improve our understanding of forest demographic responses to novel climate and disturbance regimes. Improving this understanding will require a combination of large-scale studies of sufficient spatial and temporal scale to allow for a meaningful inference of the key drivers of forest demographics and improved forest models able to approximate the hypothesized complex interactions (Anderegg et al., 2015). Traditional forestry models, including growth and yield models that have been parameterized using historical experimental data, may not be capable of representing such complex interactions or may not be applicable under novel non-analog future conditions. These model limitations lead to significant uncertainty and variability in forecasts of future boreal forest dynamics (Purves & Pacala, 2008). One potentially fruitful avenue for improving our understanding of boreal forest response to future climate and disturbance is to apply recent methodological advances in ecological forecasting (Dietze et al., 2018). These approaches allow large-scale historical and experimental data to be synthesized or fused with existing forest models to improve the model-based representation of complex forest responses to changing climate and disturbance regimes. Thus, predictions of future forest conditions are more informed and accurate. Development of these types of model frameworks and their application to new, broad-scale boreal forest data sets and experimental data on species-specific demographic parameters (reproduction, growth, seed production) will be key to sustainably managing boreal forest ecosystems well into the future.
3 Conclusions
Climate change is not the only driver of human-related change in boreal ecosystems. Nearly two-thirds of boreal forests are under some form of management, e.g., timber harvesting, plantations, fire suppression, or insect control (Gauthier et al., 2015). Despite the negative impacts of historical management regimes on critical aspects of boreal ecosystems, e.g., species diversity and structure conservation, management is perhaps our best ally to help forests adapt to ongoing changes. Silvicultural interventions such as assisted migration could help implement better warm-adapted genotypes of indigenous tree species to maintain continuous closed-crown forest habitats under climate change. With nearly 600 million trees planted each year in Canada following harvesting, the infrastructure to grow and transport these trees is already in place. Yet, our current knowledge gap in terms of species’ abilities to cope with ongoing changes and the feasibility of assisted migration are urgent matters that remain to be addressed with adequate resources. From a wildlife management perspective, various timber-harvesting strategies that reflect the variability of forest attributes resulting from natural disturbances (Gauthier et al., 2009) should be used to generate a range of landscapes and stand structures that reflect the variability of forest attributes resulting from natural disturbances and that are likely to be ecologically sustainable for wildlife (Drapeau et al., 2016), especially for threatened and endangered species (e.g., Nadeau Fortin et al., 2016). Such an increase in forest structural complexity could also provide additional resilience against global change (Messier et al., 2013).
References
Alda, F., González, M. A., Olea, P. P., et al. (2013). Genetic diversity, structure and conservation of the endangered Cantabrian Capercaillie in a unique peripheral habitat. European Journal of Wildlife Research, 59, 719–728. https://doi.org/10.1007/s10344-013-0727-6.
Allen, C. D., Breshears, D. D., & McDowell, N. G. (2015). On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere, 6(8), 129. https://doi.org/10.1890/ES15-00203.1.
Anderegg, W. R. L., Hicke, J. A., Fisher, R. A., et al. (2015). Tree mortality from drought, insects, and their interactions in a changing climate. New Phytologist, 208, 674–683. https://doi.org/10.1111/nph.13477.
Angert, A. L., Crozier, L. G., Rissler, L. J., et al. (2011). Do species’ traits predict recent shifts at expanding range edges? Ecology Letters, 14, 677–689. https://doi.org/10.1111/j.1461-0248.2011.01620.x.
Araújo, M. B., Thuiller, W., & Pearson, R. G. (2006). Climate warming and the decline of amphibians and reptiles in Europe. Journal of Biogeography, 33, 1712–1728. https://doi.org/10.1111/j.1365-2699.2006.01482.x.
Aubin, I., Munson, A. D., Cardou, F., et al. (2016). Traits to stay, traits to move: A review of functional traits to assess sensitivity and adaptive capacity of temperate and boreal trees to climate change. Environmental Reviews, 24, 164–186. https://doi.org/10.1139/er-2015-0072.
Barber, Q. E., Parisien, M. A., Whitman, E., et al. (2018). Potential impacts of climate change on the habitat of boreal woodland caribou. Ecosphere, 9, e02472. https://doi.org/10.1002/ecs2.2472.
Barbet-Massin, M., Thuiller, W., & Jiguet, F. (2012). The fate of European breeding birds under climate, land-use and dispersal scenarios. Global Change Biology, 18, 881–890. https://doi.org/10.1111/j.1365-2486.2011.02552.x.
Bastille-Rousseau, G., Schaefer, J. A., Peers, M. J. L., et al. (2018). Climate change can alter predator-prey dynamics and population viability of prey. Oecologia, 186, 141–150. https://doi.org/10.1007/s00442-017-4017-y.
Berdanier, A. B., & Clark, J. S. (2016). Multiyear drought-induced morbidity preceding tree death in southeastern U.S. forests. Ecological Applications, 26, 17–23. https://doi.org/10.1890/15-0274.
Berg, E. E., David Henry, J., Fastie, C. L., et al. (2006). Spruce beetle outbreaks on the Kenai Peninsula, Alaska, and Kluane National Park and Reserve, Yukon territory: Relationship to summer temperatures and regional differences in disturbance regimes. Forest Ecology and Management, 227, 219–232. https://doi.org/10.1016/j.foreco.2006.02.038.
Bergerud, A., & Mercer, W. (1989). Caribou introductions in eastern North America. Wildlife Society Bulletin, 17, 111–120.
Berteaux, D., Ricard, M., St-Laurent, M. H., et al. (2018). Northern protected areas will become important refuges for biodiversity tracking suitable climates. Scientific Reports, 8, 4623. https://doi.org/10.1038/s41598-018-23050-w.
Black, B. A., Colbert, J. J., & Pederson, N. (2008). Relationships between radial growth rates and lifespan within North American tree species. Ecoscience, 15, 349–357. https://doi.org/10.2980/15-3-3149.
Boisvert-Marsh, L. (2020). Patterns and processes affecting northward migration of tree species in a changing climate. Ph.D. thesis, McGill University.
Boisvert-Marsh, L., & de Blois, S. (2021). Unravelling potential northward migration pathways for tree species under climate change. Journal of Biogeography, 48, 1088–1100. https://doi.org/10.1111/jbi.14060.
Boisvert-Marsh, L., Périé, C., & de Blois, S. (2014). Shifting with climate? evidence for recent changes in tree species distribution at high latitudes. Ecosphere, 5, art83. https://doi.org/10.1890/ES14-00111.1.
Boisvert-Marsh, L., Périé, C., & de Blois, S. (2019). Divergent responses to climate change and disturbance drive recruitment patterns underlying latitudinal shifts of tree species. Journal of Ecology, 107, 1956–1969. https://doi.org/10.1111/1365-2745.13149.
Bonan, G. B., & Sirois, L. (1992). Air temperature, tree growth, and the northern and southern range limits to Picea mariana. Journal of Vegetation Science, 3, 495–506. https://doi.org/10.2307/3235806.
Brice, M. H., Cazelles, K., Legendre, P., et al. (2019). Disturbances amplify tree community responses to climate change in the temperate–boreal ecotone. Global Ecology and Biogeography, 28, 1668–1681. https://doi.org/10.1111/geb.12971.
Buechling, A., Martin, P. H., & Canham, C. D. (2017). Climate and competition effects on tree growth in Rocky Mountain forests. Journal of Ecology, 105, 1636–1647. https://doi.org/10.1111/1365-2745.12782.
Buermann, W., Forkel, M., O’Sullivan, M., et al. (2018). Widespread seasonal compensation effects of spring warming on northern plant productivity. Nature, 562, 110–114. https://doi.org/10.1038/s41586-018-0555-7.
Channell, R., & Lomolino, M. V. (2000). Trajectories to extinction: Spatial dynamics of the contraction of geographical ranges. Journal of Biogeography, 27, 169–179. https://doi.org/10.1046/j.1365-2699.2000.00382.x.
Chen, H. Y. H., & Luo, Y. (2015). Net aboveground biomass declines of four major forest types with forest ageing and climate change in western Canada’s boreal forests. Global Change Biology, 21, 3675–3684. https://doi.org/10.1111/gcb.12994.
Chen, I. C., Hill, J. K., Ohlemüller, R., et al. (2011). Rapid range shifts of species associated with high levels of climate warming. Science, 333, 1024–1026. https://doi.org/10.1126/science.1206432.
Clark, J. S., Bell, D. M., Hersh, M. H., et al. (2011). Climate change vulnerability of forest biodiversity: Climate and competition tracking of demographic rates. Global Change Biology, 17, 1834–1849. https://doi.org/10.1111/j.1365-2486.2010.02380.x.
Clark, J. S., Bell, D. M., Kwit, M. C., et al. (2014). Competition-interaction landscapes for the joint response of forests to climate change. Global Change Biology, 20, 1979–1991. https://doi.org/10.1111/gcb.12425.
Clark, J. S., Iverson, L., Woodall, C. W., et al. (2016). The impacts of increasing drought on forest dynamics, structure, and biodiversity in the United States. Global Change Biology, 22, 2329–2352. https://doi.org/10.1111/gcb.13160.
Collin, A., Messier, C., Kembel, S. W., et al. (2018). Can sugar maple establish into the boreal forest? Insights from seedlings under various canopies in southern Quebec. Ecosphere, 9, e02022. https://doi.org/10.1002/ecs2.2022.
COSEWIC. (2014). COSEWIC assessment and status report on the Caribou Rangifer tarandus, Newfoundland population, Atlantic-Gaspésie population and Boreal population, in Canada. Ottawa: Committee on the Status of Endangered Wildlife in Canada.
D’Orangeville, L., Houle, D., Côté, B., et al. (2014). Soil response to a 3-year increase in temperature and nitrogen deposition measured in a mature boreal forest using ion-exchange membranes. Environmental Monitoring and Assessment, 186, 8191–8202. https://doi.org/10.1007/s10661-014-3997-x.
D’Orangeville, L., Duchesne, L., Houle, D., et al. (2016). Northeastern North America as a potential refugium for boreal forests in a warming climate. Science, 352, 1452–1455. https://doi.org/10.1126/science.aaf4951.
D’Orangeville, L., Houle, D., Duchesne, L., et al. (2018). Beneficial effects of climate warming on boreal tree growth may be transitory. Nature Communications, 9, 3213. https://doi.org/10.1038/s41467-018-05705-4.
Deutsch, C. A., Tewksbury, J. J., Huey, R. B., et al. (2008). Impacts of climate warming on terrestrial ectotherms across latitude. Proceedings of the National Academy of Sciences of the United States of America, 105, 6668–6672. https://doi.org/10.1073/pnas.0709472105.
Di Filippo, A., Pederson, N., Baliva, M., et al. (2015). The longevity of broadleaf deciduous trees in Northern Hemisphere temperate forests: Insights from tree-ring series. Frontiers in Ecology and Evolution, 3, 46. https://doi.org/10.3389/fevo.2015.00046.
Dietze, M. C., Fox, A., Beck-Johnson, L. M., et al. (2018). Iterative near-term ecological forecasting: Needs, opportunities, and challenges. Proceedings of the National Academy of Sciences of the United States of America, 115, 1424–1432. https://doi.org/10.1073/pnas.1710231115.
Drapeau, P., Villard, M. A., Leduc, A., et al. (2016). Natural disturbance regimes as templates for the response of bird species assemblages to contemporary forest management. Diversity and Distributions, 22, 385–399. https://doi.org/10.1111/ddi.12407.
Duchesne, L., & Ouimet, R. (2008). Population dynamics of tree species in southern Quebec, Canada: 1970–2005. Forest Ecology and Management, 255, 3001–3012. https://doi.org/10.1016/j.foreco.2008.02.008.
Dullinger, S., Gattringer, A., Thuiller, W., et al. (2012). Extinction debt of high-mountain plants under twenty-first-century climate change. Climate Change, 2, 619–622. https://doi.org/10.1038/nclimate1514.
Elmhagen, B., Kindberg, J., Hellström, P., et al. (2015). A boreal invasion in response to climate change? Range shifts and community effects in the borderland between forest and tundra. Ambio, 44, S39–S50. https://doi.org/10.1007/s13280-014-0606-8.
Environment Canada. (2011). Scientific assessment to inform the identification of critical habitat for woodland caribou (Rangifer tarandus caribou), boreal population, in Canada: 2011 update (p. 102). Environment Canada.
Evans, T. G., Diamond, S. E., & Kelly, M. W. (2015). Mechanistic species distribution modelling as a link between physiology and conservation. Conservation Physiology, 3, cov056 https://doi.org/10.1093/conphys/cov056.
Fahrig, L. (2003). Effects of habitat fragmentation on biodiversity. Annual Review of Ecology, Evolution, and Systematics, 34, 487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419.
Festa-Bianchet, M., Ray, J. C., Boutin, S., et al. (2011). Conservation of caribou (Rangifer tarandus) in Canada: An uncertain future. Canadian Journal of Zoology, 89, 419–434. https://doi.org/10.1139/z11-025.
Flannigan, M. D., Logan, K. A., Amiro, B. D., et al. (2005). Future area burned in Canada. Climate Change, 72, 1–16. https://doi.org/10.1007/s10584-005-5935-y.
Foden, W. B., Butchart, S. H. M., Stuart, S. N., et al. (2013). Identifying the world’s most climate change vulnerable species: A systematic trait-based assessment of all birds, amphibians and corals. PLoS ONE, 8, e65427. https://doi.org/10.1371/journal.pone.0065427.
Ford, K. R., Breckheimer, I. K., Franklin, J. F., et al. (2017). Competition alters tree growth responses to climate at individual and stand scales. Canadian Journal of Forest Research, 47, 53–62. https://doi.org/10.1139/cjfr-2016-0188.
Foster, J. R., Finley, A. O., D’Amato, A. W., et al. (2016). Predicting tree biomass growth in the temperate-boreal ecotone: Is tree size, age, competition, or climate response most important? Global Change Biology, 22, 2138–2151. https://doi.org/10.1111/gcb.13208.
Foxcroft, L. C., Pyšek, P., Richardson, D. M., et al. (2017). Plant invasion science in protected areas: Progress and priorities. Biological Invasions, 19, 1353–1378. https://doi.org/10.1007/s10530-016-1367-z.
Franklin, J. F., Shugart, H. H., & Harmon, M. E. (1987). Tree death as an ecological process. BioScience, 37, 550–556. https://doi.org/10.2307/1310665.
Gallant, D., Lecomte, N., & Berteaux, D. (2020). Disentangling the relative influences of global drivers of change in biodiversity: A study of the twentieth-century red fox expansion into the Canadian Arctic. Journal of Animal Ecology, 89, 565–576. https://doi.org/10.1111/1365-2656.13090.
Gauthier, S., Vaillancourt, M. A., Leduc, A., et al. (Eds.). (2009). Ecosystem management in the boreal forest (p. 572). Presses de l’Université du Quebec.
Gauthier, S., Bernier, P., Kuuluvainen, T., et al. (2015). Boreal forest health and global change. Science, 349, 819–822. https://doi.org/10.1126/science.aaa9092.
Giguère-Croteau, C., Boucher, É., Bergeron, Y., et al. (2019). North America’s oldest boreal trees are more efficient water users due to increased [CO2], but do not grow faster. Proceedings of the National Academy of Sciences of the United States of America, 116, 2749–2754. https://doi.org/10.1073/pnas.1816686116.
Gillett, N. P., Weaver, A. J., Zwiers, F. W., et al. (2004). Detecting the effect of climate change on Canadian forest fires. Geophysical Research Letters, 31, L18211. https://doi.org/10.1029/2004GL020876.
Girardin, M. P., Bouriaud, O., Hogg, E. H., et al. (2016). No growth stimulation of Canada’s boreal forest under half-century of combined warming and CO2 fertilization. Proceedings of the National Academy of Sciences of the United States of America, 113(52), E8406–E8414. https://doi.org/10.1073/pnas.1610156113.
Grant, L., Johnson, C., & Thiessen, C. (2019). Evaluating the efficacy of translocation: Maintaining habitat key to long-term success for an imperiled population of an at-risk species. Biodiversity and Conservation, 28, 2727–2743. https://doi.org/10.1007/s10531-019-01789-6.
Groisman, P., & Soja, A. J. (2009). Ongoing climatic change in Northern Eurasia: Justification for expedient research. Environmental Research Letters, 4(4), 045002. https://doi.org/10.1088/1748-9326/4/4/045002.
Guisan, A., & Thuiller, W. (2005). Predicting species distribution: Offering more than simple habitat models. Ecology Letters, 8, 993–1009. https://doi.org/10.1111/j.1461-0248.2005.00792.x.
Hanes, C. C., Wang, X., Jain, P., et al. (2019). Fire-regime changes in Canada over the last half century. Canadian Journal of Forest Research, 49(3), 256–269. https://doi.org/10.1139/cjfr-2018-0293.
Harrison, P. A., Berry, P. M., Butt, N., et al. (2006). Modelling climate change impacts on species’ distributions at the European scale: Implications for conservation policy. Environmental Science & Policy, 9, 116–128. https://doi.org/10.1016/j.envsci.2005.11.003.
Hember, R. A., Kurz, W. A., & Coops, N. C. (2017). Relationships between individual-tree mortality and water-balance variables indicate positive trends in water stress-induced tree mortality across North America. Global Change Biology, 23, 1691–1710. https://doi.org/10.1111/gcb.13428.
Hof, C., Levinsky, I., Araújo, M. B., et al. (2011). Rethinking species’ ability to cope with rapid climate change. Global Change Biology, 17, 2987–2990. https://doi.org/10.1111/j.1365-2486.2011.02418.x.
Hogg, E. H., Brandt, J. P., & Michaelian, M. (2008). Impacts of a regional drought on the productivity, dieback, and biomass of western Canadian aspen forests. Canadian Journal of Forest Research, 38, 1373–1384. https://doi.org/10.1139/X08-001.
Howard, C., Stephens, P. A., Pearce-Higgins, J. W., et al. (2020). Disentangling the relative roles of climate and land cover change in driving the long-term population trends of European migratory birds. Diversity and Distributions, 26, 1442–1455. https://doi.org/10.1111/ddi.13144.
Huey, R. B., Kearney, M. R., Krockenberger, A., et al. (2012). Predicting organismal vulnerability to climate warming: Roles of behaviour, physiology and adaptation. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 367, 1665–1679. https://doi.org/10.1098/rstb.2012.0005.
Jackson, S. T., & Sax, D. F. (2010). Balancing biodiversity in a changing environment: Extinction debt, immigration credit and species turnover. Trends in Ecology & Evolution, 25, 153–160. https://doi.org/10.1016/j.tree.2009.10.001.
Juřička, D., Novotná, J., Houška, J., et al. (2020). Large-scale permafrost degradation as a primary factor in Larix sibirica forest dieback in the Khentii massif, northern Mongolia. Journal of Forest Research, 31, 197–208. https://doi.org/10.1007/s11676-018-0866-4.
Kang, J., Jiang, S., Tardif, J. C., et al. (2021). Radial growth responses of two dominant conifers to climate in the Altai Mountains Central Asia. Agricultural and Forest Meteorology, 298–299, 108297. https://doi.org/10.1016/j.agrformet.2020.108297.
Kellman, M. (2004). Sugar maple (Acer saccharum Marsh.) establishment in boreal forest: Results of a transplantation experiment. Journal of Biogeography, 31, 1515–1522. https://doi.org/10.1111/j.1365-2699.2004.01128.x.
Kharuk, V. I., Kuzmichev, V. V., Im, S. T., et al. (2014). Birch stands growth increase in Western Siberia. Scandinavian Journal of Forest Research, 29, 421–426. https://doi.org/10.1080/02827581.2014.912345.
Kharuk, V. I., Im, S. T., Dvinskaya, M. L., et al. (2015). Climate-induced mortality of spruce stands in Belarus. Environmental Research Letters, 10, 125006. https://doi.org/10.1088/1748-9326/10/12/125006.
Kharuk, V. I., Im, S. T., Petrov, I. A., et al. (2017). Fir decline and mortality in the southern Siberian Mountains. Regional Environmental Change, 17, 803–812. https://doi.org/10.1007/s10113-016-1073-5.
Kicklighter, D. W., Cai, Y., Zhuang, Q., et al. (2014). Potential influence of climate-induced vegetation shifts on future land use and associated land carbon fluxes in Northern Eurasia. Environmental Research Letters, 9, 035004. https://doi.org/10.1088/1748-9326/9/3/035004.
Kurz, W. A., Dymond, C. C., Stinson, G., et al. (2008). Mountain pine beetle and forest carbon feedback to climate change. Nature, 452, 987–990. https://doi.org/10.1038/nature06777.
Lafontaine, A., Drapeau, P., Fortin, D., et al. (2019). Exposure to historical burn rates shapes the response of boreal caribou to timber harvesting. Ecosphere, 10, e02739. https://doi.org/10.1002/ecs2.2739.
Laliberte, A. S., & Ripple, W. J. (2004). Range contractions of North American carnivores and ungulates. BioScience, 54, 123–138. https://doi.org/10.1641/0006-3568(2004)054[0123:RCONAC]2.0.CO;2.
Lankford, A. J., Svancara, L. K., Lawler, J. J., et al. (2014). Comparison of climate change vulnerability assessments for wildlife. Wildlife Society Bulletin, 38, 386–394. https://doi.org/10.1002/wsb.399.
Latham, A. D. M., Latham, M. C., McCutchen, N. A., et al. (2011). Invading white-tailed deer change wolf–caribou dynamics in northeastern Alberta. Journal of Wildlife Management, 75, 204–212. https://doi.org/10.1002/jwmg.28.
Lawler, J. J., Shafer, S. L., White, D., et al. (2009). Projected climate-induced faunal change in the Western Hemisphere. Ecology, 90, 588–597. https://doi.org/10.1890/08-0823.1.
Leclerc, M., Dussault, C., & St-Laurent, M. H. (2014). Behavioural strategies towards human disturbances explain individual performance in woodland caribou. Oecologia, 176, 297–306. https://doi.org/10.1007/s00442-014-3012-9.
Leclerc, M., Tarroux, A., Fauchald, P., et al. (2019). Effects of human-induced disturbances and weather on herbivore movement. Journal of Mammalogy, 100, 1490–1500. https://doi.org/10.1093/jmammal/gyz101.
Logan, J. A., & Powell, J. A. (2001). Ghost forests, global warming, and the mountain pine beetle (Coleoptera: Scolytidae). American Entomologist, 47, 160–173. https://doi.org/10.1093/ae/47.3.160.
Losier, C. L., Couturier, S., St-Laurent, M. H., et al. (2015). Adjustments in habitat selection to changing availability induce fitness costs for a threatened ungulate. Journal of Applied Ecology, 52, 496–504. https://doi.org/10.1111/1365-2664.12400.
Luo, Y., & Chen, H. Y. H. (2013). Observations from old forests underestimate climate change effects on tree mortality. Nature Communications, 4, 1655. https://doi.org/10.1038/ncomms2681.
Luo, Y., & Chen, H. Y. H. (2015). Climate change-associated tree mortality increases without decreasing water availability. Ecology Letters, 18, 1207–1215. https://doi.org/10.1111/ele.12500.
Lurgi, M., Brook, B. W., Saltré, F., et al. (2015). Modelling range dynamics under global change: Which framework and why? Methods in Ecology and Evolution, 6, 247–256. https://doi.org/10.1111/2041-210X.12315.
Lutz, J. A., & Halpern, C. B. (2006). Tree mortality during early forest development: A long-term study of rates, causes, and consequences. Ecological Monographs, 76, 257–275. https://doi.org/10.1890/0012-9615(2006)076[0257:TMDEFD]2.0.CO;2.
Ma, Z., Peng, C., Zhu, Q., et al. (2012). Regional drought-induced reduction in the biomass carbon sink of Canada’s boreal forests. Proceedings of the National Academy of Sciences of the United States of America, 109, 2423–2427. https://doi.org/10.1073/pnas.1111576109.
Manion, P. D. (1991). Tree disease concepts. Prentice-Hall.
Marchand, W., Girardin, M. P., Gauthier, S., et al. (2018). Untangling methodological and scale considerations in growth and productivity trend estimates of Canada’s forests. Environmental Research Letters, 13(9), 093001. https://doi.org/10.1088/1748-9326/aad82a.
Marquis, B., Bergeron, Y., Simard, M., et al. (2020). Growing-season frost is a better predictor of tree growth than mean annual temperature in boreal mixedwood forest plantations. Global Change Biology, 26(11), 6537–6554. https://doi.org/10.1111/gcb.15327.
McLane, A. J., Semeniuk, C., McDermid, G. J., et al. (2011). The role of agent-based models in wildlife ecology and management. Ecological Modelling, 222, 1544–1556. https://doi.org/10.1016/j.ecolmodel.2011.01.020.
Messier, C., Puettmann, K., & Coates, K. D. (2013). Managing forests as complex adaptive systems: Building resilience to the challenge of global change (p. 368). Routledge.
Michaelian, M., Hogg, E. H., Hall, R. J., et al. (2011). Massive mortality of aspen following severe drought along the southern edge of the Canadian boreal forest. Global Change Biology, 17, 2084–2094. https://doi.org/10.1111/j.1365-2486.2010.02357.x.
Morin, X., & Thuiller, W. (2009). Comparing niche- and process-based models to reduce prediction uncertainty in species range shifts under climate change. Ecology, 90, 1301–1313. https://doi.org/10.1890/08-0134.1.
Moyle, P. B., Kiernan, J. D., Crain, P. K., et al. (2013). Climate change vulnerability of native and alien freshwater fishes of California: A systematic assessment approach. PLoS ONE, 8, e63883. https://doi.org/10.1371/journal.pone.0063883.
Murray, D. L., Majchrzak, Y. N., Peers, M. J. L., et al. (2015). Potential pitfalls of private initiatives in conservation planning: A case study from Canada’s boreal forest. Biological Conservation, 192, 174–180. https://doi.org/10.1016/j.biocon.2015.09.017.
Myneni, R. B., Keeling, C. D., Tucker, C. J., et al. (1997). Increased plant growth in the northern high latitudes from 1981 to 1991. Nature, 386, 698–702. https://doi.org/10.1038/386698a0.
Nadeau Fortin, M. A., Sirois, L., & St-Laurent, M. H. (2016). Extensive forest management contributes to maintain suitable habitat characteristics for the endangered Atlantic-Gaspésie caribou. Canadian Journal of Forest Research, 46, 933–942. https://doi.org/10.1139/cjfr-2016-0038.
National Academies of Sciences, Engineering, and Medicine. (2019). Understanding northern latitude vegetation greening and browning: Proceedings of a workshop. Washington: The National Academies Press.
Navarro, L., Morin, H., Bergeron, Y., et al. (2018). Changes in spatiotemporal patterns of 20th century spruce budworm outbreaks in eastern Canadian boreal forests. Frontiers in Plant Science, 9, 1905. https://doi.org/10.3389/fpls.2018.01905.
Oliver, C. D., & Larson, B. C. (1996). Forest stand dynamics (Update). Yale School of the Environment Other Publications.
Opdam, P., & Wascher, D. (2004). Climate change meets habitat fragmentation: Linking landscape and biogeographical scale levels in research and conservation. Biological Conservation, 117, 285–297. https://doi.org/10.1016/j.biocon.2003.12.008.
Pacifici, M., Foden, W. B., Visconti, P., et al. (2015). Assessing species vulnerability to climate change. Nature Climate Change, 5, 215–224. https://doi.org/10.1038/nclimate2448.
Parmesan, C., & Yohe, G. (2003). A globally coherent fingerprint of climate change impacts across natural systems. Nature, 421, 37–42. https://doi.org/10.1038/nature01286.
Peng, C., Ma, Z., Lei, X., et al. (2011). A drought-induced pervasive increase in tree mortality across Canada’s boreal forests. Nature Climate Change, 1, 467–471. https://doi.org/10.1038/nclimate1293.
Peñuelas, J., Rutishauser, T., & Filella, I. (2009). Phenology feedbacks on climate change. Science, 324, 887–888. https://doi.org/10.1126/science.1173004.
Périé, C., & de Blois, S. (2016). Dominant forest tree species are potentially vulnerable to climate change over large portions of their range even at high latitudes. PeerJ, 4, e2218. https://doi.org/10.7717/peerj.2218.
Peterson, M. L., Doak, D. F., & Morris, W. F. (2019). Incorporating local adaptation into forecasts of species’ distribution and abundance under climate change. Global Change Biology, 25, 775–793. https://doi.org/10.1111/gcb.14562.
Pickles, R. S. A., Thornton, D., Feldman, R., et al. (2013). Predicting shifts in parasite distribution with climate change: A multitrophic level approach. Global Change Biology, 19, 2645–2654. https://doi.org/10.1111/gcb.12255.
Price, D. T., Cooke, B. J., Metsaranta, J. M., et al. (2015). If forest dynamics in Canada’s west are driven mainly by competition, why did they change? Half-century evidence says: Climate change. Proceedings of the National Academy of Sciences of the United States of America, 112, E4340–E4340. https://doi.org/10.1073/pnas.1508245112.
Purves, D., & Pacala, S. (2008). Predictive models of forest dynamics. Science, 320, 1452–1453. https://doi.org/10.1126/science.1155359.
Purvis, A., Jones, K. E., & Mace, G. M. (2000). Extinction. Bioessays, 22, 1123–1133. https://doi.org/10.1002/1521-1878(200012)22:12%3c1123::AID-BIES10%3e3.0.CO;2-C.
Reich, P. B., & Oleksyn, J. (2008). Climate warming will reduce growth and survival of Scots pine except in the far north. Ecology Letters, 11, 588–597. https://doi.org/10.1111/j.1461-0248.2008.01172.x.
Rowland, E. L., Davison, J. E., & Graumlich, L. J. (2011). Approaches to evaluating climate change impacts on species: A guide to initiating the adaptation planning process. Environmental Management, 47, 322–337. https://doi.org/10.1007/s00267-010-9608-x.
Rudolph, T. D., Drapeau, P., Imbeau, L., et al. (2017). Demographic responses of boreal caribou to cumulative disturbances highlight elasticity of range-specific tolerance thresholds. Biodiversity and Conservation, 26, 1179–1198. https://doi.org/10.1007/s10531-017-1292-1.
Schaefer, J. A. (2003). Long-term range recession and the persistence of caribou in the taiga. Conservation Biology, 17, 1435–1439. https://doi.org/10.1046/j.1523-1739.2003.02288.x.
Schaphoff, S., Reyer, C. P. O., Schepaschenko, D., et al. (2016). Tamm review: Observed and projected climate change impacts on Russia’s forests and its carbon balance. Forest Ecology and Management, 361, 432–444. https://doi.org/10.1016/j.foreco.2015.11.043.
Schloss, C. A., Nuñez, T. A., & Lawler, J. J. (2012). Dispersal will limit ability of mammals to track climate change in the Western Hemisphere. Proceedings of the National Academy of Sciences of the United States of America, 109, 8606–8611. https://doi.org/10.1073/pnas.1116791109.
Schneider, R. R., Hamann, A., Farr, D., et al. (2009). Potential effects of climate change on ecosystem distribution in Alberta. Canadian Journal of Forest Research, 39, 1001–1010. https://doi.org/10.1139/X09-033.
Searle, E. B., & Chen, H. Y. H. (2018). Temporal declines in tree longevity associated with faster lifetime growth rates in boreal forests. Environmental Research Letters, 13, 125003. https://doi.org/10.1088/1748-9326/aaea9e.
Seip, D. R. (1992). Factors limiting woodland caribou populations and their interrelationships with wolves and moose in southeastern British Columbia. Canadian Journal of Zoology, 70, 1494–1503. https://doi.org/10.1139/z92-206.
Seip, D. R., & Cichowski, D. B. (1996). Population ecology of caribou in British Columbia. Rangifer, 16, 73–80. https://doi.org/10.7557/2.16.4.1223.
Shoo, L. P., Williams, S. E., & Hero, J. M. (2006). Detecting climate change induced range shifts: Where and how should we be looking? Austral Ecology, 31, 22–29. https://doi.org/10.1111/j.1442-9993.2006.01539.x.
Sinclair, S. J., White, M. D., & Newell, G. R. (2010). How useful are species distribution models for managing biodiversity under future climates? Ecology & Society, 15(1), 8. https://doi.org/10.5751/ES-03089-150108.
Swetnam, T. W., Allen, C. D., & Betancourt, J. L. (1999). Applied historical ecology: Using the past to manage for the future. Ecological Applications, 9, 1189–1206. https://doi.org/10.1890/1051-0761(1999)009[1189:AHEUTP]2.0.CO;2.
Taylor, S. W., Carroll, A. L., Alfaro, R. I., et al. (2006). The mountain pine beetle: A synthesis of biology, management and impacts in lodgepole pine. Victoria: Canadian Forest Service, Natural Resources Canada.
Thomas, C. D., Hill, J. K., Anderson, B. J., et al. (2011). A framework for assessing threats and benefits to species responding to climate change. Methods in Ecology and Evolution, 2, 125–142. https://doi.org/10.1111/j.2041-210X.2010.00065.x.
Thorpe, H. C., & Daniels, L. D. (2012). Long-term trends in tree mortality rates in the Alberta foothills are driven by stand development. Canadian Journal of Forest Research, 42(9), 1687–1696. https://doi.org/10.1139/x2012-104.
Tingley, M. W., & Beissinger, S. R. (2009). Detecting range shifts from historical species occurrences: New perspectives on old data. Trends in Ecology & Evolution, 24, 625–633. https://doi.org/10.1016/j.tree.2009.05.009.
Valladares, F., Matesanz, S., Guilhaumon, F., et al. (2014). The effects of phenotypic plasticity and local adaptation on forecasts of species range shifts under climate change. Ecology Letters, 17, 1351–1364. https://doi.org/10.1111/ele.12348.
Vanderwel, M. C., & Purves, D. W. (2014). How do disturbances and environmental heterogeneity affect the pace of forest distribution shifts under climate change? Ecography, 37, 10–20. https://doi.org/10.1111/j.1600-0587.2013.00345.x.
Vors, L. S., & Boyce, M. S. (2009). Global declines of caribou and reindeer. Global Change Biology, 15, 2626–2633. https://doi.org/10.1111/j.1365-2486.2009.01974.x.
Walther, G. R., Roques, A., Hulme, P. E., et al. (2009). Alien species in a warmer world: Risks and opportunities. Trends in Ecology & Evolution, 24, 686–693. https://doi.org/10.1016/j.tree.2009.06.008.
Walton, A. (2013). Provincial-level projection of the current mountain pine beetle outbreak. Victoria: British Columbia Forest Service.
Waring, R. H. (1987). Characteristics of trees predisposed to die. BioScience, 37, 569–574. https://doi.org/10.2307/1310667.
Wärnbäck, A., & Hilding-Rydevik, T. (2009). Cumulative effects in Swedish EIA practice−Difficulties and obstacles. Environmental Impact Assessment Review, 29, 107–115. https://doi.org/10.1016/j.eiar.2008.05.001.
Watson, D. J. (1963). Climate, weather, and plant yield. In L. T. Evans (Ed.), Environmental control of plant growth (pp. 337–350). Academic Press.
Whitman, E., Parisien, M. A., Price, D. T., et al. (2017). A framework for modeling habitat quality in disturbance-prone areas demonstrated with woodland caribou and wildfire. Ecosphere, 8, e01787. https://doi.org/10.1002/ecs2.1787.
Whittington, J., Hebblewhite, M., DeCesare, N. J., et al. (2011). Caribou encounters with wolves increase near roads and trails: A time-to-event approach. Journal of Applied Ecology, 48, 1535–1542. https://doi.org/10.1111/j.1365-2664.2011.02043.x.
Wiens, J. J. (2016). Climate-related local extinctions are already widespread among plant and animal species. PLoS Biology, 14, e2001104. https://doi.org/10.1371/journal.pbio.2001104.
Wilson, R. J., Gutiérrez, D., Gutiérrez, J., et al. (2005). Changes to the elevational limits and extent of species ranges associated with climate change. Ecology Letters, 8, 1138–1146. https://doi.org/10.1111/j.1461-0248.2005.00824.x.
Woodall, C. W., Oswalt, C. M., Westfall, J. A., et al. (2009). Tree migration detection through comparisons of historic and current forest inventories. In W. McWilliams, G. Moisen, R. Czaplewski (Eds.), Forest Inventory and Analysis (FIA) Symposium 2008 (p. 9). Fort Collins: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station.
Yannic, G., Pellissier, L., Ortego, J., et al. (2014). Genetic diversity in caribou linked to past and future climate change. Nature Climate Change, 4, 132–137. https://doi.org/10.1038/nclimate2074.
Zhang, J., Huang, S., & He, F. (2015). Half-century evidence from western Canada shows forest dynamics are primarily driven by competition followed by climate. Proceedings of the National Academy of Sciences of the United States of America, 112, 4009–4014. https://doi.org/10.1073/pnas.1420844112.
Zhang, X., Bai, X., Hou, M., et al. (2019a). Warmer winter ground temperatures trigger rapid growth of dahurian larch in the permafrost forests of northeast China. Journal of Geophysical Research. Biogeosciences, 124, 1088–1097. https://doi.org/10.1029/2018JG004882.
Zhang, X., Manzanedo, R. D., D’Orangeville, L., et al. (2019b). Snowmelt and early to mid-growing season water availability augment tree growth during rapid warming in southern Asian boreal forests. Global Change Biology, 25, 3462–3471. https://doi.org/10.1111/gcb.14749.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2023 The Author(s)
About this chapter
Cite this chapter
D’Orangeville, L., St-Laurent, MH., Boisvert-Marsh, L., Zhang, X., Bastille-Rousseau, G., Itter, M. (2023). Current Symptoms of Climate Change in Boreal Forest Trees and Wildlife. In: Girona, M.M., Morin, H., Gauthier, S., Bergeron, Y. (eds) Boreal Forests in the Face of Climate Change. Advances in Global Change Research, vol 74. Springer, Cham. https://doi.org/10.1007/978-3-031-15988-6_30
Download citation
DOI: https://doi.org/10.1007/978-3-031-15988-6_30
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-15987-9
Online ISBN: 978-3-031-15988-6
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)