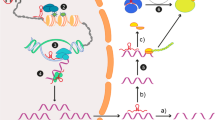
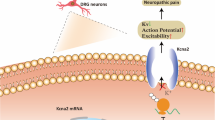
Abstract
In this review we discussed normal physiology of pain; receptors, pathways and transmitters, to compare the normal with abnormal finding in chronic neuropathic pain. We demonstrated the possible new mechanisms explaining mechanisms of chronic pain with probable new coming pharmacological solutions and recent trials to introduce new drugs capable for reducing pain intensity to improve the quality of life. Glial cells in CNS showed an important role and component in central sensitization pathway. Long non-coding RNAs (lncRNA) and Cysteinyl-aspartate-specific proteases (CASPs) showed a high-level concentration in nervous systems in patients with chronic pain. Mitochondria also was found to be involved in chronic pain through reactive oxygen species (ROS). Blocking ATP pathways in mitochondria showed positive results. However, the absolute mechanism is not well understood, multiple theories have recommended many drugs with promising effects.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- BDNF:
-
Brain-derived neurotrophic factor
- Caspase-6:
-
Cysteine-aspartic acid protease-6
- CNS:
-
Central nervous system
- CSF-1:
-
Colony-stimulating factor-1
- CX:
-
Connexin
- DAMPs:
-
Damage-associated molecular patterns
- GABA:
-
Gamma-aminobutyric acid
- JAK-STAT:
-
Janus kinase/signal transducer and activator of the transcription
- lncRNA:
-
Long non-coding RNAs
- MMPs:
-
Matrix metalloproteinases
- NMDA:
-
N-methyl-d-aspartate
- PRR:
-
Pattern-recognition receptors
- RAGE:
-
Advanced glycation end products
- TNF:
-
Tumor necrosis factors
- TRP:
-
Transient receptor potential channels
References
Fornasari D. Pain mechanisms in patients with chronic pain. Clin Drug Investig. 2012;32(Suppl 1):45–52.
Rapo-Pylkkö S, Haanpää M, Liira H. A one-year follow-up study of chronic pain in community-dwelling older adults with and without neuropathic pain. BMC Geriatr. 2017;17(1):152.
Huguet A, Miró J. The severity of chronic pediatric pain: an epidemiological study. J Pain. 2008;9(3):226–36.
Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet (London, England). 2017;390(10100):1211–59.
Mills SEE, Nicolson KP, Smith BH. Chronic pain: a review of its epidemiology and associated factors in population-based studies. Br J Anaesth. 2019;123(2):e273–83.
Pergolizzi J, Ahlbeck K, Aldington D, Alon E, Coluzzi F, Dahan A, et al. The development of chronic pain: physiological CHANGE necessitates a multidisciplinary approach to treatment. Curr Med Res Opin. 2013;29(9):1127–35.
Helme RD, Gibson S, Khalil Z. Neural pathways in chronic pain. Med J Aust. 1990;153(7):400–6.
Nicholson B. Differential diagnosis: nociceptive and neuropathic pain. Am J Manag Care. 2006;12(9 Suppl):S256–62.
Lallemend F, Ernfors P. Molecular interactions underlying the specification of sensory neurons. Trends Neurosci. 2012;35(6):373–81.
Mantyh PW. Cancer pain and its impact on diagnosis, survival and quality of life. Nat Rev Neurosci. 2006;7(10):797–809.
Schaible HG, Richter F, Ebersberger A, Boettger MK, Vanegas H, Natura G, et al. Joint pain. Exp Brain Res. 2009;196(1):153–62.
Costigan M, Scholz J, Woolf CJ. Neuropathic pain: a maladaptive response of the nervous system to damage. Annu Rev Neurosci. 2009;32:1–32.
Zhang N, Oppenheim JJ. Crosstalk between chemokines and neuronal receptors bridges immune and nervous systems. J Leukoc Biol. 2005;78(6):1210–4.
Kato J, Agalave NM, Svensson CI. Pattern recognition receptors in chronic pain: mechanisms and therapeutic implications. Eur J Pharmacol. 2016;788:261–73.
Edwards RR, Cahalan C, Mensing G, Smith M, Haythornthwaite JA. Pain, catastrophizing, and depression in the rheumatic diseases. Nat Rev Rheumatol. 2011;7(4):216–24.
Smart KM, Blake C, Staines A, Doody C. Self-reported pain severity, quality of life, disability, anxiety and depression in patients classified with ‘nociceptive’, ‘peripheral neuropathic’ and ‘central sensitisation’ pain. The discriminant validity of mechanisms-based classifications of low back (±leg) pain. Man Ther. 2012;17(2):119–25.
Sandkühler J. Models and mechanisms of hyperalgesia and allodynia. Physiol Rev. 2009;89(2):707–58.
Pedersen JL, Andersen OK, Arendt-Nielsen L, Kehlet H. Hyperalgesia and temporal summation of pain after heat injury in man. Pain. 1998;74(2–3):189–97.
Pertovaara A, Almeida A. Chapter 13: Descending inhibitory systems. In: Handbook of clinical neurology, vol. 81. Amsterdam: Elsevier; 2006. p. 179–92.
Chen G, Zhang Y-Q, Qadri YJ, Serhan CN, Ji R-R. Microglia in pain: detrimental and protective roles in pathogenesis and resolution of pain. Neuron. 2018;100(6):1292–311.
Ji R-R, Berta T, Nedergaard M. Glia and pain: is chronic pain a gliopathy? Pain. 2013;154:S10–28.
Shi Y, Shu J, Liang Z, Yuan S, Tang SJ. EXPRESS: oligodendrocytes in HIV-associated pain pathogenesis. Mol Pain. 2016;12:1744806916656845.
Berta T, Park C-K, Xu Z-Z, Xie R-G, Liu T, Lü N, et al. Extracellular caspase-6 drives murine inflammatory pain via microglial TNF-α secretion. J Clin Invest. 2014;124(3):1173–86.
Abbadie C, Bhangoo S, De Koninck Y, Malcangio M, Melik-Parsadaniantz S, White FA. Chemokines and pain mechanisms. Brain Res Rev. 2009;60(1):125–34.
Guan Z, Kuhn JA, Wang X, Colquitt B, Solorzano C, Vaman S, et al. Injured sensory neuron-derived CSF1 induces microglial proliferation and DAP12-dependent pain. Nat Neurosci. 2016;19(1):94–101.
Ji R-R, Xu Z-Z, Gao Y-J. Emerging targets in neuroinflammation-driven chronic pain. Nat Rev Drug Discov. 2014;13(7):533–48.
Raghavendra V, Tanga F, DeLeo JA. Inhibition of microglial activation attenuates the development but not existing hypersensitivity in a rat model of neuropathy. J Pharmacol Exp Ther. 2003;306(2):624–30.
Echeverry S, Shi XQ, Yang M, Huang H, Wu Y, Lorenzo LE, et al. Spinal microglia are required for long-term maintenance of neuropathic pain. Pain. 2017;158(9):1792–801.
Sweitzer S, Schubert P, DeLeo J. Propentofylline, a glial modulating agent, exhibits antiallodynic properties in a rat model of neuropathic pain. J Pharmacol Exp Ther. 2001;297(3):1210–7.
Walters ET. Neuroinflammatory contributions to pain after SCI: roles for central glial mechanisms and nociceptor-mediated host defense. Exp Neurol. 2014;258:48–61.
Ellis A, Wieseler J, Favret J, Johnson KW, Rice KC, Maier SF, et al. Systemic administration of propentofylline, ibudilast, and (+)-naltrexone each reverses mechanical allodynia in a novel rat model of central neuropathic pain. J Pain. 2014;15(4):407–21.
Binshtok AM, Wang H, Zimmermann K, Amaya F, Vardeh D, Shi L, et al. Nociceptors are interleukin-1β sensors. J Neurosci. 2008;28(52):14062–73.
Takeda M, Kitagawa J, Takahashi M, Matsumoto S. Activation of interleukin-1β receptor suppresses the voltage-gated potassium currents in the small-diameter trigeminal ganglion neurons following peripheral inflammation. Pain. 2008;139(3):594–602.
Gustafson-Vickers SL, Van Lu B, Lai AY, Todd KG, Ballanyi K, Smith PA. Long-term actions of interleukin-1β on delay and tonic firing neurons in rat superficial dorsal horn and their relevance to central sensitization. Mol Pain. 2008;4:63.
Kawasaki Y, Zhang L, Cheng J-K, Ji R-R. Cytokine mechanisms of central sensitization: distinct and overlapping role of interleukin-1β, interleukin-6, and tumor necrosis factor-α in regulating synaptic and neuronal activity in the superficial spinal cord. J Neurosci. 2008;28(20):5189–94.
Zhang R-X, Li A, Liu B, Wang L, Ren K, Zhang H, et al. IL-1ra alleviates inflammatory hyperalgesia through preventing phosphorylation of NMDA receptor NR-1 subunit in rats. Pain. 2008;135(3):232–9.
Milligan ED, O’Connor KA, Nguyen KT, Armstrong CB, Twining C, Gaykema RP, et al. Intrathecal HIV-1 envelope glycoprotein gp120 induces enhanced pain states mediated by spinal cord proinflammatory cytokines. J Neurosci. 2001;21(8):2808–19.
Gabay E, Wolf G, Shavit Y, Yirmiya R, Tal M. Chronic blockade of interleukin-1 (IL-1) prevents and attenuates neuropathic pain behavior and spontaneous ectopic neuronal activity following nerve injury. Eur J Pain. 2011;15(3):242–8.
Chen G, Park C-K, Xie R-G, Berta T, Nedergaard M, Ji R-R. Connexin-43 induces chemokine release from spinal cord astrocytes to maintain late-phase neuropathic pain in mice. Brain. 2014;137(8):2193–209.
Wang H, Cao Y, Chiang C-Y, Dostrovsky JO, Sessle BJ. The gap junction blocker carbenoxolone attenuates nociceptive behavior and medullary dorsal horn central sensitization induced by partial infraorbital nerve transection in rats. Pain. 2014;155(2):429–35.
Ji R-R, Xu Z-Z, Wang X, Lo EH. Matrix metalloprotease regulation of neuropathic pain. Trends Pharmacol Sci. 2009;30(7):336–40.
Kawasaki Y, Xu Z-Z, Wang X, Park JY, Zhuang Z-Y, Tan P-H, et al. Distinct roles of matrix metalloproteases in the early- and late-phase development of neuropathic pain. Nat Med. 2008;14(3):331–6.
Diatchenko L, Nackley AG, Tchivileva IE, Shabalina SA, Maixner W. Genetic architecture of human pain perception. Trends Genet. 2007;23(12):605–13.
Liu H, Yao YM, Yu Y, Dong N, Yin HN, Sheng ZY. Role of Janus kinase/signal transducer and activator of transcription pathway in regulation of expression and inflammation-promoting activity of high mobility group box protein 1 in rat peritoneal macrophages. Shock. 2007;27(1):55–60.
Salaffi F, Giacobazzi G, Di Carlo M. Chronic pain in inflammatory arthritis: mechanisms, metrology, and emerging targets—a focus on the JAK-STAT pathway. Pain Res Manag. 2018;2018:8564215.
Taylor RC, Cullen SP, Martin SJ. Apoptosis: controlled demolition at the cellular level. Nat Rev Mol Cell Biol. 2008;9(3):231–41.
Berta T, Lee JE, Park CK. Unconventional role of caspase-6 in spinal microglia activation and chronic pain. Mediat Inflamm. 2017;2017:9383184.
Li Z, Li X, Chen X, Li S, Ho IHT, Liu X, et al. Emerging roles of long non-coding RNAs in neuropathic pain. Cell Prolif. 2019;52(1):e12528.
Wu W, Ji X, Zhao Y. Emerging roles of long non-coding RNAs in chronic neuropathic pain. Front Neurosci. 2019;13:1097.
Li C, Lei Y, Tian Y, Xu S, Shen X, Wu H, et al. The etiological contribution of GABAergic plasticity to the pathogenesis of neuropathic pain. Mol Pain. 2019;15:1744806919847366.
von Hehn CA, Baron R, Woolf CJ. Deconstructing the neuropathic pain phenotype to reveal neural mechanisms. Neuron. 2012;73(4):638–52.
Ulmann L, Hatcher JP, Hughes JP, Chaumont S, Green PJ, Conquet F, et al. Up-regulation of P2X4 receptors in spinal microglia after peripheral nerve injury mediates BDNF release and neuropathic pain. J Neurosci. 2008;28(44):11263–8.
Coull JA, Beggs S, Boudreau D, Boivin D, Tsuda M, Inoue K, et al. BDNF from microglia causes the shift in neuronal anion gradient underlying neuropathic pain. Nature. 2005;438(7070):1017–21.
Inoue K. Purinergic signaling in microglia in the pathogenesis of neuropathic pain. Proc Jpn Acad Ser B Phys Biol Sci. 2017;93(4):174–82.
Sui BD, Xu TQ, Liu JW, Wei W, Zheng CX, Guo BL, et al. Understanding the role of mitochondria in the pathogenesis of chronic pain. Postgrad Med J. 2013;89(1058):709–14.
Kim HY, Chung JM, Chung K. Increased production of mitochondrial superoxide in the spinal cord induces pain behaviors in mice: the effect of mitochondrial electron transport complex inhibitors. Neurosci Lett. 2008;447(1):87–91.
Joseph EK, Levine JD. Mitochondrial electron transport in models of neuropathic and inflammatory pain. Pain. 2006;121(1–2):105–14.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Abbas, K.S., Allam, A.ES., Galluccio, F., El OUMRI, A.A., AlKharabsheh, A., Salti, A. (2022). Pathophysiology of Chronic Pain. In: de Castro, J., El Miedany, Y. (eds) Advances in Chronic and Neuropathic Pain. Contemporary Rheumatology. Springer, Cham. https://doi.org/10.1007/978-3-031-10687-3_3
Download citation
DOI: https://doi.org/10.1007/978-3-031-10687-3_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-10686-6
Online ISBN: 978-3-031-10687-3
eBook Packages: MedicineMedicine (R0)