Abstract
The three Gulf of Guinea oceanic islands, Príncipe, São Tomé, and Annobón, have always remained isolated from the African continent and correspond to the westernmost peaks of the Cameroon Volcanic Line, while the island of Bioko, part of the same ridge, was connected to the mainland during glaciations. Despite the small area of the oceanic islands, their relief and remoteness have enabled the evolution of remarkable ecological and biological diversity. Concerning diurnal Lepidoptera, 91 species and subspecies are known from the oceanic islands: 46 from Príncipe, 64 from São Tomé, and 8 from Annobón; and 35 are endemic: 17 to Príncipe, 23 to São Tomé, and only 5 shared among islands. Further species have been reported in error, either due to misidentification or to mislabelling. A revised checklist of the species and subspecies of the Gulf of Guinea oceanic islands Papilionoidea is presented as is a summary of their taxonomy, distribution, and ecology.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
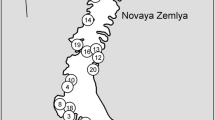
The Gulf of Guinea Oceanic Islands (GGOI), Príncipe, São Tomé, and Annobón (=Pagalu), represent the Cameroon Volcanic Line’s westernmost peaks; they have never been connected to one another nor to the mainland. Bioko (=Fernando Poo), on the other hand, is a continental island that was connected to mainland Africa during glaciations, most recently, ca. 11,000 years (Lambert and Chappel 2001). Annobón is part of Equatorial Guinea, and with ca. 17 km2, it is located ca. 180 km SSW from São Tomé and 340 km W from Gabon (Jones and Tye 2006). The maximum altitude of this small island is ca. 600 m (Quioveo peak) and its biodiversity (Exell 1944, 1956, 1973; Heras et al. 2002), Papilionoidea included, is much lower than that of the other GGOI. Príncipe is the smaller of the Democratic Republic of São Tomé e Príncipe main islands. With ca. 140 km2, it is ca. 210 km SSW off Bioko and 140 km NNE of ST with a maximum altitude ca. 935 m (Pico Príncipe). It was, however, much larger historically and extended to the SSW (Jones and Tye 2006), including the plantless Tinhosas Islets. The Tinhosas are currently a breeding colony of sea birds, a Ramsar site, from which no Papilionoidea are known. São Tomé is ca. 860 km2, and lies almost 150 km SW of Príncipe, 180 km NNE of Annobón and ca. 255 NW of Gabon, with a maximum altitude (Pico São Tomé) ca. 2024 m. the Equator crosses Rolas Islet, which is just South of São Tomé.
The predominant natural ecosystem type of the GGOI was described as rainforest (Exell 1944, 1973) or tropical moist broadleaf forest (Gascoigne 2004), which is stratified into lowland (0–800 m), montane (800–1400 m), and mist forest (1400–2024 m), the last category being absent from Príncipe. In Annobón the three types appear compressed in its small altitudinal range as in the lower São Tomé southern peaks (Ogonovszky 2003). Due to rain shadow, NW São Tomé has a dry forest, which is rich in endemic butterfly species.
In relation to diurnal Lepidoptera, the skippers (Hesperiidae) were formerly placed in the superfamily Hesperioidea, independent from the butterflies, but Heikkilä et al. (2012), supported by morphological and genetic data, demonstrated that they are part of the same evolutionary group, the superfamily Papilionoidea. Their diversity and endemism in São Tomé and Príncipe has been the focus of several notable studies (Pyrcz 1992a; Mendes and Bivar-de-Sousa 2012a) and a further contribution is currently in preparation (Mendes et al. in prep.). Here we provide an updated checklist of the Papilionoidea of the GGOI and review previously incorrectly cited taxa.
The Papilionoidea of the Gulf of Guinea Oceanic Islands: Data Sources
The first references to the Papilionoidea of Annobón were given by Aurivillius (1910), who reported Borbo fatuellus (sub Baoris), and Kheil (1910) who noted Leptotes pirithous (sub Syntarucus), A. zetes (as monotypic) and Telchinia pharsalus (sub Acraea). Aurivillius (1928) and Bacelar (1948) recorded Euchrysops osiris (sub Cupido). D’Abrera (1980) described Acraea zetes annobona (type locality), which also occurs in São Tomé and Príncipe, being endemic to the three islands. Viejo (1984) added Melanitis leda, Danaus chrysippus, Hypolimnas misippus and Telchinia pharsalus (sub Acraea) to the list of species of these islands but some of the samples were reported only from “Spanish Guinea”. Olano and Marcos (1993) report the species assigned by Viejo (1984) but consider D. chrysippus as restricted to Rio Muni and Bioko while Euchrysops osiris is assigned to the mainland only. Acraea zetes annobona is not recognized by Olano and Marcos (1993) despite Annobón being its type locality. These authors also question the presence in the islands of Sevenia boisduvalli insularis (today considered a São Tomé endemic) and of Dixeia piscicollis reported by D’Abrera (1980) from Equatorial Guinea but later considered as a São Tomé endemic (D’Abrera 1997). These are the only data known on the Papilionoidea of the most remote and less diverse GGOI. An even lower diversity is known from the remote Saint Helena that, while not part of the Cameroon Volcanic Line, lies eastwards of the Mid-Atlantic chain (Ashmole and Ashmole 2000): Lampides boeticus, Danaus chrysippus, Vanessa cardui and Hypolimnas misippus, all widely ranged migratory species.
The scientific knowledge of butterfly and skipper diversity of the GGOI has increased intermittently since Cramer (1775–1776). It (almost) stabilized during the eighteenth century (1850–1870) but suddenly increased from the turn of the nineteenth until the twentieth century with the contributions of Aurivillius, Joicey and Talbot, Sharpe and Snellen. After another hiatus, several additional phases of discovery followed, firstly due to the work of Amélia Bacelar (Bacelar 1948, 1958) in the mid-twentieth century, and then, toward the end of the century the contributions of Libert (2004, 2011), Pyrcz (1991a–c) and our work in São Tomé and Príncipe (Mendes et al. 1988, 2018; Mendes and Bivar-de-Sousa 2006, 2012a, b).
Some large collections, which are mostly currently housed in the Museu Nacional de História Natural e da Ciência (MUHNAC), in Lisbon, Portugal, were studied by Mendes and Bivar-de-Sousa 2006, 2012a, b; Mendes et al. 2018, but the revised checklist (Appendix) takes into account bibliographic records that have been reviewed and confirmed by newly collected data. The bibliographic data considered for assembling this checklist originated from the following references: Cramer (1775–76—who reports the first species for São Tomé and for the GGOI: Acraea medea), Snellen (1873), Sharpe (1893), Holland (1896), Aurivillius (1898, 1910), Rothschild and Jordan (1900), Eltringham (1912), Le Cerf (1924), Joicey and Talbot (1926, 1927), Hawker-Smith (1928), Riley (1928), Evans (1937), Bacelar (1948), Someren (1971a, b, 1972, 1974, 1975), Pinhey (1972), D’Abrera (1980, 1997, 2004, 2009), Plantrou (1983), Hancock (1984), Henning (1988), Pyrcz (1991a, b, c, 1992a, b), Canu (1994), Wojtusiak and Pyrcz (1995, 1997), Pierre et al. (2002), Hecq (2003a, b), Turlin (2005a, b, 2007a, b, c), Anonymous (2007), Bonfim and Carvalho (2009), Koçak and Kemal (2009), Williams (2008, 2015), Velzen et al. (2009), Libert (2011), Oremans (2012), Pierre and Bernaud (2013, 2014), Collins and Larsen (2013), Wikipedia (2014), Collins (2015) and Awanao et al. (2018).
Specimens deposited in the Museu Bocage (MB), a precursor institution of present-day MUHNAC, were also partially studied by the authors. However, all of MB specimens were destroyed in the fire that engulfed the collections, library, and associated structures on March 28, 1978. Among the destroyed collections were Sharpe (1893) São Tomé type specimens, as had been recorded by Fernandes (1958), the holotype of Pyrrhiades bocagei (sub Rhopalocampta), three specimens of Leptotes sanctithomae (as Catochrysops sancti-thomae), comprising holotype and allotype, as well as the holotypes of Acraea niobe, Telchinia insularis (sub Acraea) and Telchinia newtoni (sub Acraea).
Most of the information concerning newly identified and re-examined specimens is based on the analysis of specimens from São Tomé and Príncipe collected by the zoological expeditions held in 1954–1955 by the Centro de Zoologia (CZ) of the Junta de Investigações do Ultramar and a mission to São Tomé Island in 1984 by MB and the Faculty of Sciences of the Lisbon University (Mendes et al. 1988). These studies were followed by Mendes and Bivar-de-Sousa (2006, 2012a, b) and Mendes et al. (2018) contributions, all based on newly collected specimens between 2004 and 2019. Some of these specimens were collected during a 2015 California Academy of Sciences funded expedition, to which the senior author was invited by Dr. Robert (Bob) Drewes to participate. Some other material was studied, namely that available in the collections of the Centro de Investigação Agronómica e Tecnológica de São Tomé e Príncipe (CIAT) in Potó, São Tomé Island. This collection was established by members of the CZ before the country gained independence in 1975. Data available from the private collections of the second co-author of this study, as well as those of António Figueira (1924–2017; now housed in the collections of the Museu de História Natural e da Ciência da Universidade do Porto, Porto, Portugal) and Carlos da Silva were also used. Information from local collaborators, especially those of Rato Cabinda, are also included in this revised checklist.
Incorrectly Recorded Taxa
Despite most of the known references being scientifically trustworthy, several reported taxa are based on misidentified or mislabelled specimens of species and subspecies that are mostly typical from savanna and Sahel-biotopes (see Appendix):
Hesperiidae
Sarangesa phidyle, Spialia diomus, S. spio, Gomalia elma, Borbo gemella and Pelopidas mathias were recorded for the islands but do not occur there (Appendix). Evans (1937) and Chiba (2009) consider Coeliades hanno as present in São Tomé, but the only brown Coeliades confirmed in São Tomé and Príncipe is the morphologically similar C. forestan.
Papilionidae
Papilio dardanus sulfurea is a Bioko endemic whose type locality was wrongly considered to be Príncipe (Palisot de Beauvois 1805–1821). Its occurrence on Príncipe Island was also recorded by Canu (1994) and by Koçak and Kemal (2009—sub princeps), but all the recent authors consider it a Bioko endemic. Graphium angolanus baronis, G. latreillianus theorini and G. ridleyanus, which are large and unmistakable taxa occurring in continental Africa have also been recorded from São Tomé based on some specimens in the collections of the Natural History Museum (NHM) of London (Smith and Vane-Wright 2001); however, the location of these specimens is likely mislabelled, as no other records of these taxa exist for the island.
Pieridae
Larsen (2005) considers Colotis doubledayi, noted for instance by Ackery et al. (1995) and D’Abrera (1997) as occurring in São Tomé, as a misidentification. Berger (1981) is the only record of Belenois gidica for São Tomé. The single reference of Appias phaola to the island of São Tomé (Bacelar 1948) refers to three males and one female that were subsequently destroyed during the MB fire. These likely represented records of A. epaphia aequatorialis, later described from the island (Mendes and Bivar-de-Sousa 2006). Leptosia medusa, reported also by Bacelar (1948, four males, one female) from São Tomé corresponds to a misidentified L. alcesta (Mendes and Bivar-de-Sousa 2012a, b). All the São Tomé records of species of the genus Mylothris, except those of M. rembina, are putative misidentifications and were partially rectified: M. asphodelus by Bacelar (1948); M. bernice by Sharpe (1893); M. nubila by Schultze (1917); M. poppea by Viejo (1984); M. rhodope and M. spica by Berger (1979); and M. sulphurea by Pyrcz (1992a, who stresses the muddled knowledge of the genus in São Tomé).
Lycaenidae
Hypolycaena philippus, Anthene amarah, Azanus moriqua, Azanus ubaldus, Leptotes brevidentatus, L. jeannelli, and Zizula hylax were recorded for the islands but do not occur there (Appendix). Liptena evanescens f. xanthis reported by Stempffer et al. (1974) from São Tomé either represents a mislabeled specimen or a locally extirpated population. Libert (2004) reports the presence of Hypomyrina fournieri either in São Tomé or in Príncipe (material not seen, occurrence not mapped), but the species is not mentioned as occurring in the GGOI by Ackery et al. (1995) nor by D’Abrera (2009). Sharpe (1893) reports Rubropelates aruma in São Tomé, but this record may represent a non-established population or a misidentified specimen of Deudorix lorisona. In addition, Sharpe (1893) registers Leptotes pulchra in São Tomé but does not consider the quite common L. pirithous.
Nymphalidae
Bicyclus dorothea concolor, B. funebris and B. martius sanaos were recorded for the islands but do not occur there (Appendix). Furthermore, B. italus was mapped in São Tomé by Condamin (1973), but all these records may have been based on a misidentified B. medontias, a similar forest species whose presence on the island was unknown until recently (Larsen 2005). Precis hierta, Precis orithya, Salamis anacardii (currently a species of Protogoniomorpha), Byblia ilithyia and Hamanumida daedalus are certainly not present in São Tomé and Príncipe. Neptis serena is listed by Koçak and Kemal (2009) for São Tomé and Príncipe, but these authors do not report the endemic Neptis larseni from Príncipe; indeed, N. serena is only assigned to Príncipe when that endemic was described (Wojtusiak and Pyrcz 1997). Pyrcz (1992a) reports an undetermined red Cymothoe that Canu (1994) assigned to São Tomé, but was unable to find the specimens later; several species of the C. sangaris group are known from Central and West Africa and thus this information is insufficient to consider its presence in the GGOI. The same must be stated about the references of several “acraea” species (currently Acraea and Telchinia) reported by Bacelar (1958), Mendes and Bivar-de-Sousa (2012a, b), Pierre and Bernaud (1999, 2009a, b), Pierre et al. (2002), Pyrcz (1992a), Snellen (1873) and Viejo (1984), namely Acraea pseudegina, Telchinia e. encedon, T. esebria, T. pentapolis, and T. vesperalis. Telchinia jodutta was reported from São Tomé and Príncipe by several authors—first reference by Aurivillius (1910)—though Oremans (2012) describes its insular vicariant, A. severina, with one subspecies in São Tomé and another in Príncipe. Bacelar (1958) refers A. eponina latifasciata but she did not have access to new specimens, while Pyrcz (1992a) questioned its presence in São Tomé and Príncipe and Pierre et al. (2002) did not report the species as occurring in the GGOI despite its huge African range. Bacelar (1948) reports Acraea monteironis from São Tomé based upon one male whose identification we could not confirm—if the specimen was in MB it was certainly destroyed in 1978; this species, described from Angola, was never recorded again from the island. Viejo (1984), based in Bacelar (1948), was the only subsequent author who considers Acraea monteironis to occur in São Tomé, without providing further comments. According to Bacelar (1948), the reference of Aurivillius (1928, Figs, 57d–e) of an A. esebria form shall represent T. severina instead, a taxon that has been until recently considered a synonym of T. esebria (Eltringham 1912; Ackery et al. 1995; Williams 2008; Pierre and Bernaud 2014). This is, however, certainly not true in the GGOI—it may, indeed, belong to T. s. severina synonymy in the case of its reference to São Tomé. Reports of Phalanta phalantha aethiopica in the GGOI must represent the morphologically similar forest dweller P. eurytis, known from Príncipe, São Tomé, and Rio Muni (Viejo 1984).
Composition, Diversity, and Endemism of the GGOI Papilionoidea
Isolation, geological age, area, catching-area, relief, climate, and diversity of biotopes are fundamental to the potential biodiversity and endemism of an island (Whittaker et al. 2017). Among the families of Papilionoidea known in Sub-Saharan Africa, only Riodinidae has not been recorded in the GGOI. Most of the subfamilies assigned from the region occur both in Príncipe and in São Tomé (the scant diversity of Annobón was already discussed). The currently known number of species (in parentheses) per family and respective subfamily is as follows: Hesperiidae (nine): Coeliadinae (two), Pyrginae (one), and Hesperiinae (six); Papilionidae (four): Papilioninae (four); Pieridae (12): Coliadinae (five) and Pierinae (seven); Lycaenidae (21): Miletinae (one), Aphnaeinae (five) and Polyomatinae (15); and Nymphalidae (45): Libytheinae (one), Danainae (one), Satyrinae (two), Charaxinae (eight), Nymphalinae (10), Cyrestinae (one), Biblidinae (two), Limenetidinae (four) and Heliconiinae (16). There are no Papilionoidea genera that are endemic from the GGOI. The Nymphalidae is the most diverse family (ca. 45% of the Papilionoidea) and the Heliconiinae (ca. 35%) its most diverse subfamily.
According to Gascoigne (1995), the Lepidoptera endemicity for São Tomé is 38.3% (47 taxa: 11 endemic species, 7 endemic subspecies) and 21.4% for Príncipe (42 taxa: six endemic species, three endemic subspecies). These estimates, based on Pyrcz (1991a, b), did not include the Hesperiidae, lumped the São Tomé and Príncipe species of the genus Neptis to a single species (N. eltringhami), and considered Leptotes terrenus and Chilades sanctithomae as independent taxa.
Currently, based on the specimens we have examined and bibliographic references, 91 species and subspecies are considered to occur in the GGOI: 46 are known for Príncipe, 64 (though three in need of revision) for São Tomé, and eight for Annobón (Appendix). All the taxa known for Annobón also occur in São Tomé and Príncipe, with the exception of Telchinia pharsalus, identified as T. p. carmen, but putatively referring to a still undescribed Annobón subspecies (Kheil 1910; Viejo 1984). The subspecies Acraea zetes annobona is the single taxon that is endemic to the three islands. The species that occur both in São Tomé and Príncipe are mostly those with the largest distributions. Regarding endemics (Fig. 13.1), 17 taxa are endemic for Príncipe (almost 37%), 23 for São Tomé (ca. 36%), and five are endemic to more than one island. These estimates approach those of Pyrcz (1991a, b), especially after considering that he did not consider skippers.
Some endemic Papilionoidea of the Gulf of Guinea oceanic islands. R: recto (dorsal), V: verso (ventral): (1) Pyrrhiades bocagei, ♂R; (2) Ibid, V. (3.) Papilio nerminae, ♂R; (4) Graphium leonidas santamarthae, ♂R; (5) Dixeia piscicollis, ♂R; (6) Deudorix (Virachola) odana chalybeate, ♂R; (7) Charaxes antiques, ♂R; (8) Ibid, V; (9) Pseudacraea gamae, ♀R; (10) Acraea zetes annobona, ♂R; (11) Ibid, V; (12) Acraea medea, ♂R; (13) Ibid, V. Photo credits: (1–3, 5, 10–13) António Bivar-de-Sousa and Luís Mendes, (4, 6–9) Carlos da Silva. Photos not to scale
Conservation
Modifications of the natural environment associated with human expansion and climatic change have impacted the diversity of butterflies and skippers around the globe. Activities with detrimental impacts range from deforestation, fires, charcoal production, introduction of invasive weeds or of animal pests, use of chemicals, water, air and soil pollution, expanse of cultivated fields and of monocultures (agriculture or forestry) and human expansion in its strict sense. The considerable taxonomic knowledge and data available for lepidopteran distributions and ecological requirements make them important indicators for environmental change and for monitoring the health of ecosystems (Parmesan 2019).
In the GGOI, population sizes of species with high sensitivity to ecological changes may be important to monitor, especially under current climate change. Some of the GGOI species are very common while others seem to be restricted to small areas or associated to specific biotopes (Mendes et al. in prep.). As noted elsewhere, most of these more rare and poorly distributed taxa are endemics and strictly associated with natural biotopes, although some exceptions are remarkable: we frequently found images of the São Tomé endemic Acraea niobe pollinating cultivated introduced coffee plants Coffea sp. (Rubiaceae) or, as firstly noted Pierre et al. (2002), feeding on the nectar of the introduced Mexican sunflower, Tithonia diversifolia (Asteraceae). These observations suggest that some endemic species may be resilient to some ecological changes.
The observation that some species are known from only a small number of individuals may be the consequence of two fundamental and unrelated mechanisms. First, some species are considered rare when they occur in quite localized areas, they fly during a short period, or they are restricted to particular biotopes, and thus challenging to locate. However, these species with patchy distributions may be geographically widespread and common in other parts of their range. Second, insect abundance is known to fluctuate from year to year according to annual ecological conditions and in the GGOI some areas are quite complicated to access. Thus, temporal and geographic biases in survey effort may partly explain why some taxa appear to be rare. However, some species may be considered threatened because they are truly rare and at real risk of extirpation or extinction due to environmental pressures. We note that these concepts apply to both endemic and non-endemic species.
One good example of a rare endemic species is Dixeia piscicollis, which is restricted to the São Tomé dry forest and known from a small number of specimens. However, at the end of the dry season, locally known as gravana, it may be quite abundant, even becoming the dominant or the only butterfly in the area—although it is restricted to this area. Some endemic Charaxes restricted to the São Tomé or Príncipe highland forests also fit this description. Likewise, the insular subspecies of Graphium leonidas are both rare and limited to precise biotopes and seem to fly during a short period only.
A certain number of threatened species in São Tomé and Príncipe were reported by Gascoigne (1995). However, this list included taxa that even if rare, may not be threatened, as is the case of Pyrrhiades bocagei (sub Coeliades) which is noted as vulnerable, and despite being present on both islands, is reported only for São Tomé. Both Graphium leonidas subspecies are listed as endangered; Leptotes terrenus and Chilades sanctithomae, Pseudacraea gamae are “undetermined,” while Epamera bellina maris and Charaxes defulvata are said to be extinct, despite Pyrcz (1992a) recording live specimens of both. The threatened community of Lagoa Azul in São Tomé is discussed, with special attention on Coeliades bocagei, the “endemic Charaxes” (species not discriminated), and Neptis eltringhami that occur there. Dixeia piscicollis was also considered to be threatened though it is common along the northwestern dry forest.
The statuses of species considered endemic or almost endemic are in dire need of new data to update assessments, as several of them may be threatened or even extinct. This is the case for Andronymus thomasi, treated as a subspecies from Andronymus neander by Gascoigne (1995) and possibly others. Iolaus bellina maris and Charaxes defulvata if not extinct, likely occur only in very small numbers.
Potential Future Discoveries and Research
Given the current knowledge of the GGOI Papilionoidea, the intermittent increase in the number of recognized species over time, and the considerable bias of knowledge toward São Tomé and Príncipe when compared with Annobón, several areas of future research are needed. Fieldwork in Annobón is essential, as the known taxonomic diversity is likely incomplete. São Tomé and Príncipe have been more extensively surveyed, but large areas of their most pristine forest, where endemics are expected to have evolved, have been little explored due to the difficult access, high rainfall, and dense vegetation. As such, new species for the islands (and even for science) are expected to be found, and populations of some of the species considered as (almost) extinct may be rediscovered. Information on the range and abundance of each species is essential to establish their conservation status. This is especially urgent for the rare island endemics. Although some information already exists about the activity periods of caterpillars and imagoes, more data are needed to allow the implementation of effective protection measures. For instance, the morphology of the caterpillar life stage is unknown for most of the tropical species, particularly the endemics. Likewise, knowledge of caterpillar host-plants is also incomplete. In the non-endemic taxa, the caterpillar food-plants may be different from those of populations in mainland Africa, while for some of the insular endemics they simply remain unknown. Genetic studies on GGOI Papilionoidea are virtually inexistent. They are fundamental to taxonomical and phylogeographic studies and are key to investigating the biogeographic history of this unique island fauna.
References
Ackery PR, Smith CR, Vane-Wright RI (1995) Carcasson’s African butterflies. An annotated atalogue of the Papilionoidea and Hesperioidea of the Afrotropical region. Commonwealth Scientific and Industrial Research Organisation, Victoria, pp i–xii. + 803
Anonymous (2007) National report on the status of biodiversity in S. Tomé and Príncipe. Ministry for Natural Resources and the Environment – Directorate General for Environment, República Democrática de S. Tomé e Príncipe, p 109
Ashmole P, Ashmole M (2000) St Helena and W Ascension islands: a natural history. A. Nelson, England, p 475
Aurivillius C (1898) Rhopalocera Aethiopica. Die Tagfalter des äthiopischen Faunengebietes. Eine systematisch -geographische Studie. Kongliga Svenska Vetenskaps-Akademiens Handlinge 31(5):11–561
Aurivillius C (1910) Schmeterlinge gesammelt in Westafrika von Leonardo Fea in den Jahren 1897–1902. Annali del Museo Civico di Storia Naturale di Genova (Ser 3) 4:494–530
Aurivillius C (1928) Les macrolépidoptères de la faune ethiopiènne. 13. (Diurnes ethiopiens). In: Seitz A (ed) Les macrolépidoptères du Globe. Fritz Lehmann/E. Le Moult, Stuttgart and Paris, p 615. + 80 pl
Awanao K, Ozawa H, Yago M, Nishino Y (2018) Catalogue of the Shingeru Eda insect collection, the university museum, the University of Tokyo. Part I. Lepidoptera, Rhopalocera. Available via the University of Tokyo. http://umdb.um.u-tokyo.ac.jp/DDoubutu/eda/en/index.php. Accessed 1 June 2018
Bacelar A (1948) Lepidópteros de África principalmente das colónias portuguesas (Colecção do Museu Bocage). Arquivos do Museu Bocage 19:165–207
Bacelar A (1958) Entomofauna da Guiné Portuguesa e das ilhas de S. Tomé e do Príncipe. V. Lepidoptera (Rhopalocera). Conferência Internacional dos Africanistas Ocidentais 4:47–60
Berger LA (1979) Espèces peu connues et description de deux nouvelles sous-espèces de Mylothris (Lepidoptera Pieridae). Revue de Zoologie Africaine 93(1):1–9
Berger LA (1981) Les papillons du Zaïre. Présidence de la République, Kinshasa, p 543
Bonfim F, Carvalho S (2009) Fourth national report on the biodiversity. Ministry of Natural Resources and Environment – directorate general of the environment, República Democrática de S. Tomé e Príncipe, p 93
Canu JG (1994) Souvenirs entomologiques d’Afrique et des iles voisines (Lepidoptera, Rhopalocera). Lambillionea 94(3):311–320
Chiba H (2009) A revision of the subfamily Coeliadinae (Lepidoptera: Hesperiidae). Bulletin of the Kitakyushu Museum of Natural History and Human History (A - Natural History) 7:1–102
Collins S (2015) Part 43. The ABRI collection II. Unusual butterflies. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 4. + 32 pl
Collins SC, Larsen TB (2013) A new species in the Afrotropical genus Artitropa from São Tomé and Príncipe (Lepidoptera: Hesperiidae: Hesperiinae (incerta sedis)). Metamorphosis 24:20–24
Condamin M (1973) Monographie du genre Bicyclus (Lepidoptera Satyridae). Mémoires de l’Institut Fondamental d’Afrique Noire 88:1–324
Cramer P (1775–76) De uitlandsche kapellee voorkomende in de drie waereld-deelen Asia, Africa en America. S. J. Baalde and Barthelemy Wild, Amsterdam and Utrecht, i–xxx. + 155 pp + 96 pl
D’Abrera B (1980) Butterflies of the Afrotropical region. Lansdowne Publ., Melbourne, i–xx + 593 pp
D’Abrera B (1997) Butterflies of the Afrotropical region. Part I. Papilionidae, Pieride, Acraeidae, Danaidae & Satyridae. New and Revised Edition. Hill House Publ., Melbourne, i-xxiv + 1–257 pp
D’Abrera B (2004) Butterflies of the Afrotropical region. Part II. Nymphalidae, Libytheidae. New and Revised Edition. Hill House Publ., Melbourne, pp i–xli. + 258–530
D’Abrera B (2009) Butterflies of the Afrotropical region. Part III. Lycaenidae, Riodinidae. New and Revised Edition. Hill House Publ, Melbourne, pp i–xxvi. + 531–876
Palisot de Beauvois AMFJ (1805–21) Insectes recueillis en Afrique et en Amérique, dans les royaumes d'Oware et de Benin, a Saint-Domingue et dans les Etats-Unis, pendant les années 1786–1797. Levrault, Schoell et Cie, Paris, 276. + 90 pl
Eltringham H (1912) A monograph of the African species of the genus Acraea fab., with a supplement on those of the oriental region. Transactions of the Entomological Society of London 1912:1–413
Evans WH (1937) A catalogue of the African Hesperiidae indicating the classification and nomenclature adopted in the British museum. British museum, natural history, London, p 212 + 30 pl
Exell AW (1944) Catalogue of the vascular plants of S. Tomé (with Principe and Annobon). British museum (natural history), London, p 428
Exell AW (1956) Supplement to the catalogue of the vascular plants of S. Tomé (with Principe and Annobón). British museum (natural history), London, p 58
Exell AW (1973) Angiosperms of the islands of the Gulf of Guinea (Fernando Pó, Príncipe, S. Tomé and Annobón). Bulletin of the British museum (natural history). Botany 4:325–411
Fernandes JA (1958) Tipos entomológicos do Museu Bocage. Lista dos tipos das colecções entomológicas do Museu e Laboratório Zoológico e Antropológico da Faculdade de Ciências de Lisboa (1ª nota). Revista Portuguesa de Zoologia e Biologia Geral 1(4):293–302
Gascoigne A (1995) A Lista Vermelha de Animais Ameaçados de São Tomé e Príncipe. ECOFAC (Conservation et Utilisation Rattionelle des Ecosystèmes Forestiers en Afrique Centrale) – Componente de São Tomé e Príncipe, São Tomé, p 15
Gascoigne A (2004) São Tomé, Príncipe, and Annobón Moist Lowland Forests. In: Burgess N, Hales JDA, Underwoof E et al (eds) Terrestrial ecoregions of Africa and Madagascar: a conservation assessment. Island Press, Washington DC, pp 236–238
Hancock DL (1984) The Princeps nireus group of swallowtails (Lepidoptera: Papilionidae). Systematics, phylogeny and biogeography. Arnoldia Zimbabwe 9(12):181–215
Hawker-Smith W (1928) New forms of Lycaenidae in the Musée du Congo Belge at Tervueren. Revue de Zoologie et Botanique Africaine 16(2):214–216
Hecq J (2003a) Part 16. Nymphalidae VII. Pseudacraea. In: Bauer E, Franckebach T (eds) Butterflies of the world. Goecke & Evers, Keltem, pp 1–3. + 12 pl
Hecq J (2003b) Le genre Pseudacraea Westwood, 1850. Nymphalidae, Limenitinae, Limenitini. Supplement 7. In: Bauer E, Franckebach T (eds) Butterflies of the world. Goecke & Evers, Keltem, pp 1–12
Heikkilä M, Kaila L, Mutanen M et al (2012) Cretaceous origin and repeated tertiary diversification of the redefined butterflies. Proceedings of the Royal Society of London B 279:1093–1099
Henning SF (1988) The Charaxinae butterflies of Africa. Aloe Books, Frandsen, p 457
Heras P, Infante M, Ondó CO et al (2002) Vegetacíon de la isla de Annobón (República de Guinea Ecuatorial). Estudios del Museo de Ciencias Naturales de Álava 17:115–123
Holland WJ (1896) A preliminary revision and synonymic catalogue of the Hesperiidae of Africa and adjacent islands, with description of some apparently new species. Proceedings of the Zoological Society 207 pp + pl I–V
Joicey JT, Talbot G (1926) New forms of Lepidoptera from the island of São Tomé, West Africa. The Entomologist 59:220–226
Joicey JT, Talbot G (1927) Four new butterflies from the island of St. Príncipe. The Entomologist 60:12–16
Jones PJ, Tye A (2006) The birds of São Tomé and Príncipe, with Annobón: islands of the Gulf of Guinea. British Ornithologists Union, Oxford (United Kingdom), p 172
Kheil NM (1910) Catálogo sistemático de la fauna de las posesiones españolas del Golfo de Guinea (Lepidópteros). Memorias de la Sociedad Española de Historia Natural 1:545–549
Koçak AÖ, Kemal M (2009) Third report on the temporary results of the lepidopteran list of Africa continent based upon the info-system of the Cesa. Cesa Publications on African Lepidoptera (Royal Museum for Central Africa) 25:1–2810
Lambert K, Chappel J (2001) Sea level change through the last glacial cycle. Science 292:679–686
Larsen TB (2005) Butterflies of West Africa. Text volume. Apollo Books, Stenstrup, p 595
Le Cerf F (1924) Lépidoptères nouveaux de la collection du Muséum (Rhopalocera). Bulletin du Muséum National d’Histoire Naturelle 30:137–139
Libert M (2004) Révision des Deudorix africains (Lepidoptera, Lycaenidae). Ed. Lambillionea, Tervuren, p 201. + 16 pl
Libert M (2011) Note sur le genre Leptotes Scudder, 1876 (Lepidoptera, Lycaenidae). Lambillionea 111:53–66
Mendes LF, Bivar-de-Sousa A (2006) Notes and descriptions of Afrotropical Appias butterflies (Lepidoptera: Pieridae). Boletín de la Sociedad Entomológica Aragonesa 39:151–160
Mendes LF, Bivar-de-Sousa A (2012a) Riqueza de espécies e endemicidade das borboletas diurnas (Lepidoptera: Rhopalocera) em São Tomé e Príncipe. Actas do Colóquio Internacional São Tomé e Príncipe numa perspectiva interdisciplinar, diacrónica e sincrónica. Instituto Universitário de Lisboa, Centro de Estudos Africanos, Instituto de Investigação Científica Tropical, Lisbon, pp 653–663
Mendes LF, Bivar-de-Sousa A (2012b) New account on the butterflies (Lepidoptera: Rhopalocera) from São Tomé e Príncipe. Boletín de la Sociedad Entomológica Aragonesa 51:157–186
Mendes LF, Rocha-Pité MT, Serrano ARM et al. (1988) Sobre a fauna terrestre e ribeirinha da República Democrática de São Tomé e Príncipe. A Missão Zoológica do Departamento de Zoologia e Antropologia da Faculdade de Ciências de Lisboa e do Museu Nacional de História Natural (Museu Bocage) à República Democrática de São Tomé e Príncipe (13/VI–7/VII/1984). Nota preliminar. Arquivos do Museu Bocage Volume especial:1–16
Mendes LF, Bivar-de-Sousa A, Vasconcelos S (2018) New data on the butterflies of São Tomé e Príncipe: description of a new subspecies from Príncipe, notes, and reference to two faunistic novelties from São Tomé (Lepidoptera, Papilionoidea). Shilap 46(181):65–74
Mendes LF, Bivar-de-Sousa A, Melo M et al (in prep.) Butterflies and skippers (Lepidoptera: Papilionoidea) of São Tomé e Príncipe/Borboletas diurnas (Lepidoptera: Papilionoidea) de São Tomé e Príncipe. Arte e Ciência, Porto
Ogonovszky M (2003) Endémisme et phytogéographie des plantes de São Tomé et Príncipe. Université Libre de Bruxelles, Brussels, p 100. + 127 pp annexes
Olano I, Marcos JM (1993) Lepidópteros Papilionoidea de Guinea Ecuatorial y sus islas. Estudios del Museo de Ciencias Naturales de Álava 8:137–169
Oremans P (2012) Contribution à l’étude de la biodiversité des îles du Golfe de Guinée: description d’une nouvelle espèce et d’une nouvelle sous-espèce de Sao Tome et Principe (Lepidoptera, Nymphalidae). Saturnafrica 11:21–23
Parmesan C (2019) Chapter 24. Butterflies as bioindicators for climate change effects. In: Boggs CL, Watt WB, Ehrlich PR (eds) Butterflies: ecology and evolution taking flight. University of Chicago Press, Chicago, pp 541–560
Pierre J, Bernaud D (1999) Révision de la super-espèce Acraea (pentapolis): A. (p.) polis n. sp. et premiers états de A. (pentapolis) et A. vesperalis (Lepidoptera, Nymphalidae). Bulletin de la Société Entomologique de France 104(1):5–14
Pierre J, Bernaud D (2009a) Part 31. Nymphalidae XVI. Acraea subgenus Actinote. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 5. + 19 pl
Pierre J, Bernaud D (2009b) Supplement 16. Les Acraeinae Fabricius, 1807 de l’Ancien Monde (Lepidoptera, Nymphalidae). Le genre Acraea Fabricius, 180, sous-genre Actinote. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 19
Pierre J, Bernaud D (2013) Part 39. Nymphalidae XXIII. Acraea subgenus Acraea. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 8. + 28 pl
Pierre J, Bernaud D (2014) Supplement 24. Le genre Acraea Fabricius, 1807: Liste systématique, synonymique et liste des noms infrasubspécifiques. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 30
Pierre J, Bernaud D, Oremans P (2002) Les Acraea de São Tomé (Lepidoptera, Nymphalidae). Bulletin de la Société Entomologique de France 107(3):285–294
Pinhey E (1972) A new species of Dixeia Talbot (Lepidoptera: Pieridae). Arnoldia 5(25):1–5
Plantrou J (1983) Systématique, biogéographie et évolution des Charaxes africains (Lepidoptera, Nymphalidae). Publications du Laboratoire de Zoologie de l’École Normale Supérieure 25:1–457
Pyrcz T (1991a) Account on two expeditions to Sao Tome and Principe islands January-March 1989 and July-September 1990 including a description of new subspecies of Acraea pharsalus Ward and Bematites alcinoe Felder. Lambillionea 91(4):362–367
Pyrcz T (1991b) Description of a new subspecies of Acraea pharsalus Ward from Príncipe Island (Lepidoptera, Acraeidae). Lambillionea 91(4):368–370
Pyrcz T (1991c) Description of a new subspecies of Bematites alcinoe Felder from Príncipe Island (Lepidoptera, Acraeidae). Lambillionea 91(4):370–374
Pyrcz T (1992a) Provisional check-list of the butterflies of São Tomé and Príncipe islands. Lambillionea 92(1):46–52
Pyrcz T (1992b) Rain forests of São Tomé e Príncipe: butterflies and conservation. Tropical Lepidoptera 3(2):95–100
Riley ND (1928) A list of Hesperiidae collected in the island of St. Thome by T. A. Barnes. The Entomologist 61:54–65
Rothschild A, Jordan K (1900) A monograph of Charaxes and allied Prionopterous genera. Novitates Zoologicae 7:281–524
Schultze A (1917) Lepidoptera. Teil I. Wissenschaftliche Ergebnisse der zweiten deutschen Zentral-Afrika-Expedition, 1910–1911, unter Führung Adolf Friedrichs. Klinkhardt & Biermann, Leipzig, pp 511–596. + 3 pl
Sharpe E (1893) Description of new species of butterflies from the island of St. Thomas, West Africa. Proceedings of the Zoological Society of London 1893:553–558
Smith CR, Vane-Wright RI (2001) A review of the Afrotropical species of the genus Graphium (Lepidoptera: Rhopalocera: Papilionidae). Bulletin of the Natural History Museum of London (Entomology) 70(2):503–719
Snellen PCT (1873) Lepidoptera van het Prinzen eiland (Ilha-do-Príncipe). Tijdschrift voor Entomologie 16:71–74
Someren VGL (1971a) Revisional notes on African Charaxes. Part VI. Bulletin of the British Museum (Natural History) (Entomology) 25(5):197–250
Someren VGL (1971b) Revisional notes on African Charaxes. Part VII. Bulletin of the British Museum (Natural History) (Entomology) 26(4):181–226
Someren VGL (1972) Revisional notes on African Charaxes. Part VIII. Bulletin of the British Museum (Natural History) (Entomology) 27(4):215–264
Someren VGL (1974) Revisional notes on African Charaxes. Part X. Bulletin of the British Museum (Natural History) (Entomology) 29(8):415–487
Someren VGL (1975) Revisional notes on African Charaxes, Palla and Euxanthe. Part X. Bulletin of the British Museum (Natural History) (Entomology) 32(3):65–136
Stempffer H, Bennett NH, May SJ (1974) A revision of some groups of Liptena Westwood (Lepidoptera, Lycaenidae). Bulletin of the British Museum (Natural History) (Entomology) 13:107–181
Turlin B (2005a) Part 22. Nymphalidae X.Charaxes I. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 7. + 35 pl
Turlin B (2005b) Supplement 10. Les Charaxes de la faune afrotropicale. Partie I. Nymphalidae: Charaxinae: Charaxini. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 11
Turlin B (2007a) Part 25. Nymphalidae XII. Charaxes II. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 8. + 23 pl
Turlin B (2007b) Supplement 12. Les Charaxes de la faune afrotropicale. Partie II. Nymphalidae: Charaxinae: Charaxini. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 8
Turlin B (2007c) Part 28. Nymphalidae XII. Charaxes III. In: Bauer E, Franckenbach T (eds) Butterflies of the world. Goecke & Evers, Keltem, p 8. + 32 pl
Velzen R, Larsen TB, Bakker FT (2009) A new hidden species of the Cymothoe caenis -complex (Lepidoptera: Nymphalidae) from Western Africa. Zootaxa 2197:53–63
Viejo JL (1984) Contribución al conocimiento de las mariposas del Golfo de Guinea. Eos 60:335–369
Whittaker RJ, Fernández-Palacios JM, Matthews TJ, Borregaard MK, Triantis KA (2017) Island biogeography: taking the long view of nature’s laboratories. Science 357(6354):eaam8326
Wikipedia (2014) List of butterflies of São Tomé and Príncipe. Available via Wikipedia. https://en.wikipedia.org/wiki/List_of_butterflies_of_S%C3%A3o_Tom%C3%A9_and_Pr%C3%ADncipe. Accessed 31 Aug 2015
Williams MC (2008) Butterflies and skippers of the Afrotropical region (Papilionoidea and Hesperioidea). An encyclopedia. 7th edition. Available via https://atelim.com/butterflies-and-skippers-of-the-afrotropical-region.html. Accessed 21 Oct 2021
Williams MC (2015) Classification of the Afrotropical butterflies to generic level. Metamorphosis 26:102–108
Wojtusiak J, Pyrcz TW (1995) Observations on the postembryonic development and comments on the systematic status of the butterfly Kamilla cymodoce Cramer, 1777 (Lepidoptera: Nymphalidae). Lambillionea 95(2):116–120
Wojtusiak J, Pyrcz TW (1997) Taxonomy of three butterfly species from the islands of São Tomé and Príncipe in the Gulf of Guinea: Neptis larseni n. sp., Charaxes thomasius n. stat., Papilio furvus n. stat. (Lepidoptera: Papilionoidea). Lambillionea 97(1):53–63
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Appendix
Appendix
Commented checklist of Papilionoidea recorded from the Gulf of Guinea oceanic islands. Names of the 91 species and subspecies considered correctly assigned to Príncipe (P), São Tomé (ST) and Annobón (A) are numbered. Taxa considered incorrectly assigned to the GGOI, as justified in the text, are not numbered. K&K (2009): Koçak and Kemal; M&BS: Mendes and Bivar-de-Sousa. Endemic taxa (E), if shared by P and ST and/or A, respectively as E-1, E-2 and E-3 according to their topotypical island. Samples examined by the authors are marked in the SS column (studied specimens) with a ●; 17 species are known only from reliable bibliographic references. Preferred habitats (H): A—Humid lowland forest; B—Humid highland forest; C—Dry forest, forest margins, and somewhat degraded biotopes; D—Several types of forest; U—Ubiquitous or almost ubiquitous; ?—Doubtful
Families and species/subspecies | First reference | SS | P | ST | A | H |
|---|---|---|---|---|---|---|
Fam. HESPERIIDAE | ||||||
1. Pyrrhiades bocagei (Sharpe, 1893) | Sharpe (1893) | ● | E | E-2 | D | |
2. Coeliades forestan (Stoll, 1784) | Sharpe (1893) | ● | X | X | C, D | |
Coeliades hanno (Plötz, 1879) | Riley (1928) | |||||
3. Tagiades flesus (Fabricius, 1871) | Sharpe (1893) | ● | X | X | A, C | |
Sarangesa phydile (Walker, 1870) | K&K (2009) | |||||
Spialia diomus (Hopffer, 1855) | K& K (2009) | |||||
Spialia spio (Linnaeus, 1764) | K&K (2009) | |||||
Gomalia elma (Trimen, 1862) | K&K (2009) | |||||
4. Andronymus thomasi Riley, 1928 | Riley (1928) | ● | E | E-2 | A | |
5. Artitropa principetome Collins and Larsen 2013 | (Collins and Larsen 2013) | E-1 | E | A | ||
Pelopidas mathias (Fabricius, 1798) | K&K (2009) | |||||
6. Borbo borbonica (Boisduval, 1833) | Aurivillius (1910) | ● | X | X | C | |
7. Borbo detecta (Trimen, 1893) | Riley (1928) | ● | X | X | C | |
8. Borbo f. fatuellus (Hopffer, 1855) | Aurivillius (1910) | ● | X | X | X | C |
Borbo gemella (Mabille, 1884) | K&K (2009) | |||||
9. Afrogegenes letterstedti (Wallengren, 1857) | Sharpe (1893) | ● | X | X | C | |
Fam. PAPILIONIDAE | ||||||
10. Papilio nerminae Koçak, 1983 | Sharpe (1893) | ● | E | A, B | ||
Papilio dardanus sulfurea Palisot de Beauvois 1806 | Palisot de Beauvois (1806) | |||||
11. Papilio d. demodocus Esper, 1798 | Snellen (1873) | ● | X | X | U | |
Graphium angolanus baronis (Ungemach, 1932) | Smith and Vane-Wright (2001) | |||||
Graphium latreillianus theorini (Aurivillius, 1831) | Smith and Vane-Wright (2001) | |||||
12. Graphium leonidas santamarthae Joicey and Talbot, 1927 | Joicey and Talbot, (1927) | ● | E | A, C | ||
13. Graphium leonidas thomasius Le Cerf 1924 | Le Cerf (1924) | ● | E | A, C | ||
Graphium ridleyanus (White, 1843) | Smith and Vane-Wright (2001) | |||||
Fam. PIERIDAE | ||||||
14. Catopsilia florella (Faricius, 1775) | Bacelar (1958) | ● | X | X | U | |
15. Eurema b. brigitta (Stoll, 1780) | K&K (2009) | ● | X | X | C | |
16. Eurema hecabe solifera (Butler, 1875) | Snellen (1882) | ● | X | C | ||
17. Eurema floricola leonis (Butler, 1886) | Snellen (1882) | ● | X | D | ||
18. Eurema senegalensis (Boisuval, 1836) | Sharpe (1893) | ● | X | X | A | |
Colotis doubledayi (Hopffer, 1872) | Ackery et al. (1995) | |||||
Belenois gidica Godart, 1819 | Berger (1981) | |||||
19. Belenois c. creona (Stoll, 1780) | M&BS (2012) | ● | X | C | ||
20. Dixeia piscicollis Pinhey, 1972 | Pinhey (1972) | ● | E | C | ||
21. Appias epaphia aequatorialis Mendes & Bivar-de-Sousa, 2006 | M&BS (2006) | ● | E | C | ||
22. Appias epaphia piresi Mendes & Bivar-de-Sousa, 2006 | M&BS (2006) | ● | E | C | ||
Appias phaola (Doubleday, 1847) | Bacelar (1948) | |||||
23. Leptosia a. alcesta (Stoll, 1781) | Snellen (1873) | ● | X | A | ||
Leptosia medusa (Cramer, 1777) | Bacelar (1948) | |||||
24. Leptosia n. nupta (Butler, 1873) | Sharpe (1873) | ● | X | X | A, B | |
Mylothris asphodelus Butler, 1888 | Bacelar (1948) | |||||
Mylothris bernice (Hewitson, 1862) | Sharpe (1893) | |||||
Mylothris nubila (Möschler, 1884) | Schutze (1917) | |||||
Mylothris popea (Cramer, 1777) | Viejo (1984) | |||||
25. Mylothris rembina (Plötz, 1880) | Schutze (1917) | ● | X | X | A | |
Mylothris rhodope (Fabricius, 1775) | Berger (1979) | |||||
Mylothris spica (Möschler, 1884) | K&K (2009) | |||||
Mylothris sulphurea (Aurivillius, 1895) | K&K (2009) | |||||
Fam. LYCAENIDAE | ||||||
26. Spalgis l. lemolea Druce, 1890 | Pyrcz (1992) | ● | X | A, C | ||
Liptena evanescens xanthis (Holland, 1890) | Stempffer (1974) | |||||
27. Iolaus (Epamera) bellina maris (Riley, 1928) | Riley (1928) | E | A? | |||
Hypomyrina fournieri Gabriel, 1939 | Libert (2004) | |||||
Hypolycaena phillippus (Fabricius, 1793) | K&K (2009) | |||||
28. Deudorix (Virachola) l. lorisona (Hewitson, 1862) | Hawker-Smith (1928) | ● | X | X? | A | |
29. Deudorix (Virachola) a. antalus (Hopffer, 1855) | Pyrcz (1992) | ● | X | X | C | |
30. Deudorix (Virachola) caliginosa Lathy,1903 | Libert (2004) | X | C? | |||
31. Deudorix (Virachola) odana chalybeata (Joicey and Talbot, 1926) | Joicey and Talbot (1926) | ● | E | E-2 | A | |
Rubropelates a. aruma (Hewitson, 1873) | Sharpe (1893) | |||||
Anthene amarah (Guérin-Méneville,1849) | K&K (2009) | |||||
32. Anthene l. lunulata (Trimen, 1894) | Viejo (1984) | ● | X | X? | A, B | |
33. Anthene prínceps (Butler, 1876) | Pyrcz (1992) | ● | X | X | A, B | |
34. Pseudonacaduba s. sichela (Wallengren, 1857) | Mendes et al. (ad.Prep.) | ● | X | C | ||
35. Lampides boeticus (Linnaeus, 1767) | Bacelar (1958) | ● | X | X | D | |
36. Cacyreus lingeus (Stoll, 1782) | Joicey and Talbot (1926) | ● | X | X | A, C | |
37. Leptotes p. pirithous (Linnaeus, 1767) | Kheil (1910) | ● | X | X | X | C, D |
Leptotes brevidentatus (Tite, 1958) | K&K (2009) | |||||
Leptotes jeanneli (Stempffer, 1935) | K&K (2009) | |||||
Leptotes pulchra (Murray, 1874) | Sharpe (1893) | |||||
38. Leptotes pyrczi Libert, 2011 | Libert (2011) | ● | E | A | ||
39. Leptotes sanctithomae Sharpe, 1893 (= L. terrenus (Joicey and Talbot, 1926)) | Sharpe (1893) | E | B? | |||
40. Zizeeria knysna (Trimen, 1862) | Joicey and Talbot (1926) | ● | X | X | A, C | |
41. Zizina otis antanossa (Mabille, 1877) | Pyrcz (1992) | ● | X | X | A, C | |
Zizula hylax (Fabricius, 1775) | K&K (2009) | |||||
42. Azanus mirzá (Plötz, 1880) | Pyrcz (1992) | ● | X | C, D | ||
Azanus moriqua (Wallengren, 1857) | K&K (2009) | |||||
Azanus ubaldus (Stoll, 1782) | K&K (2009) | |||||
43. Eicochrysops hippocrates (Fabricius, 1793) | Pyrcz (1992) | ● | X | X | D | |
44. Euchrysops malathana (Boisduval, 1833) | Aurivillius (1928) | ● | X | X | C, D | |
45. Euchrysops cf. osiris (Hopffer, 1855) | Aurivillius (1928) | ● | X | X? | X | C, D |
46. Chilades trochylus (Freyer, 1844) | K& K (2009) | ● | X | D | ||
Fam. NYMPHALIDAE | ||||||
47. Libythea l. labdaca Westwood, 1851 | Sharpe (1893) | ● | X | X | A, C | |
48. Danaus c. chrysippus (Linnaeus, 1758) | Snellen (1873) | ● | X | X | X | U |
49. Melanitis leda (Linnaeus, 1758) | Sharpe (1893) | ● | X | X | X | C, D |
Bicyclus dorothea concolor Condamin and Fox, 1964 | Condamin and Fox (1964) | |||||
Bicyclus funebris (Guérin-Méneville, 1844) | Condamin (1973) | |||||
Bicyclus italus (Hewitson, 1865) | Condamin (1973) | |||||
50. Bicyclus medontias (Hewitson, 1873) | Larsen (2005) | X | B? | |||
Bicyclus martius sanaos (Hewitson, 1866) | Condamin (1973) | |||||
51. Bicyclus vulgaris (Butler, 1868) | Aurivillius (1910) | ● | X | C | ||
52. Charaxes defulvata (Joicey and Talbot, 1926) | Joicey and Talbot (1926) | E | A? | |||
53. Charaxes c. candiope (Godart, 1824) | Plantrou (1983) | X | X | A, C | ||
54. Charaxes thomasius Staudinger, 1886 | Staudinger (1886) | ● | E | B | ||
55. Charaxes lemosi (Joicey and Talbot, 1927) | Aurivillius (1910) as C.lucretius | ● | E | B | ||
56. Charaxes odysseus Staudinger 1892 | Staudinger (1892) | E | A?, B | |||
57. Charaxes antiquus Joicey and Talbot 1926 | Joicey and Talbot (1926) | E | B | |||
58. Charaxes barnsi Joicey and Talbot, 1927 | Joicey and Talbot (1927) | E | A | |||
59. Charaxes monteiri Staudinger 1886 | Staudinger (1886) | E | B | |||
60. Vanessa cardui (Linnaeus, 1758) | Pyrcz (1992) | ● | X | X | C | |
Precis hierta crebrene (Trimen, 1870) | K&K (2009) | |||||
Precis orythia madagascariensis (Guenée, 1865) | K&K (2009) | |||||
61. Precis pelarga (Fabricius, 1775) | Aurivillius (1910) | ● | X | X | B, C | |
62. Precis s. sinuta Plötz, 1880 | Sharpe (1893) | ● | X | C, D | ||
63. Hypolimnas a. anthedon (Doubleday, 1845) | Sharpe (1893) | ● | X | X | C, D | |
64. Hypolimnas misippus (Linnaeus, 1764) | Snellen (1873) | ● | X | X | X | C |
65. Hypolimnas m. monteironis (Druce, 1874) | Pyrcz (1992) as H. salmacis | ● | X | A, B | ||
66. Hypolimnas salmacis thomensis Aurivillius, 1910 | Aurivillius (1910) | ● | E | A, B | ||
Protogoniomorpha anacardi (Linnaeus, 1758) | K&K (2009) | |||||
67. Junonia cymodoce lugens (Schultze, 1912) | Bacelar (1958) | ● | X | A | ||
68. Junonia o. oenone (Linnaeus, 1758) | Bacelar (1958) | ● | X | X | C, D | |
69. Junonia t. terea (Drury, 1773) | Aurivillius (1910) | ● | X | A, B | ||
70. Cyrestis c. camillus (Fabricius, 1781) | Bacelar (1958) | ● | X | A | ||
Byblia ilithyia (Drury, 1773) | K&K (2009) | |||||
71. Sevenia amulia principensis Mendes & Bivar-de-Sousa, 2018 n.stat. | Bacelar (1958)—no ssp | ● | E | A, B | ||
72. Sevenia boisduvali insularis (Joicey & Talbot 1926) | Sharpe (1893) | ● | E | B | ||
73. Pseudacraea gamae | Joicey and Talbo (1926) | E | A | |||
Neptis serena Overlaet, 1955 | K&K (2009) | |||||
74. Neptis eltringhami Joicey and Talbot 1926 | Joicey and Talbot (1926) | E | A? | |||
75. Neptis larseni Wojtuziak and Pyrcz, 1997 | Pyrcz (1991) as N. eltringhami | ● | E | A, C | ||
76. Cymothoe caenis (Drury, 1773) | van Velzen et al. (2009) | X | A? | |||
Cymothoe sp. (“sangaris-group”) | Pyrcz (1992)—after Canu | |||||
Hamanumida daedalus (Fabricius, 1775) | K&K (2009) | |||||
77. Acraea n. neobule Doubleday, 1847 | K&K (2009) | ● | X | C, D | ||
78. Acraea q. quirina (Fabricius, 1781) | Aurivillius (1910) | ● | X | X | A | |
79. Acraea zetes annobona D’Abrera, 1980 | Snellen (1873)—no ssp | ● | E | E | E-3 | A, C |
80. Acraea e. egina (Cramer, 1775) | M&BS (2012) | ● | X | A, B | ||
81. Acraea medea (Craner, 1775) | Cramer (1775) | ● | E | A, B | ||
82. Acraea niobe Sharpe, 1893 | Sharpe (1893) | ● | E | A, B | ||
Acraea pseudegina Westwood, 1852 | Pyrcz (1992) | |||||
83. Acraea alcinoe racaji Pyrcz, 1991 | Snellen (1873) as A. esebria | E | A, B | |||
84. Telchinia alciope (Hewitson, 1852) | Pierre (1985) | X | X | A | ||
Telchinia esebria (Hewitson, 1861) | Snellen (1873) | |||||
Telchinia encedon (Linnaeus, 1758) | K&K (2009) | |||||
85. Telchinia insularis (Sharpe 1893) | Sharpe (1893) | ● | E | A | ||
Telchinia j. jodutta (Fabricius, 1793) | Aurivillius (1910) | |||||
86. Telchinia lycoa (Godart, 1819) | Aurivillius (1910) | ● | X | A, C | ||
Telchinia p. pentapolis (Ward, 1871) | Pierre and Bernaud (1999) | |||||
87. Telchinia pharsalus carmen (Pyrcz, 1991) | Aurivillius (1910)—no ssp | ● | E | X? | A, B | |
Telchinia serena (Fabricius, 1775) | Snellen (1873) as A. manjaca | |||||
88. Telchinia severina severina (Ouremans, 2012) | Berger (1986) as A. jodutta | ● | E | A, B | ||
89. Telchinia severina terreirovelhoensis (Ouremans, 2012) | Aurivillius (1910) as A. jodutta | ● | E | A, B | ||
Telchinia vesperalis (Grose-Smith, 1890) | Pyrcz (1992) | |||||
90. Telchinia newtoni (Sharpe 1893) | Sharpe (1893) | ● | E | |||
Acraea monteironis Butler, 1874 | Bacelar (1958) | |||||
91. Phalanta e. eurytis (Doubleday, 1847) | Pyrcz (1992) | X | X | A | ||
Phalanta phalantha (Drury, 1773) | K&K (2009) | |||||
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2022 The Author(s)
About this chapter
Cite this chapter
Mendes, L.F., Bivar-de-Sousa, A. (2022). Butterflies and Skippers (Lepidoptera: Papilionoidea) of the Gulf of Guinea Oceanic Islands. In: Ceríaco, L.M.P., de Lima, R.F., Melo, M., Bell, R.C. (eds) Biodiversity of the Gulf of Guinea Oceanic Islands. Springer, Cham. https://doi.org/10.1007/978-3-031-06153-0_13
Download citation
DOI: https://doi.org/10.1007/978-3-031-06153-0_13
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-06152-3
Online ISBN: 978-3-031-06153-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)