Abstract
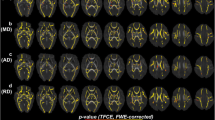
Prematurity and preterm stressors severely affect the development of infants born before 37 weeks of gestation, with increasing effects seen at earlier gestations. Although preterm mortality rates have declined due to the advances in neonatal care, disability rates, especially in middle-income settings, continue to grow. With the advances in MRI imaging technology, there has been a focus on safely imaging the preterm brain to better understand its development and discover the brain regions and networks affected by prematurity. Such studies aim to support interventions and improve the neurodevelopment of preterm infants and deliver accurate prognoses. Few studies, however, have focused on the fully developed brain of preterm born infants, especially in extremely preterm subjects. To assess the long-term effect of prematurity on the adult brain, myelin related biomarkers such as myelin water fraction and g-ratio are measured for a cohort of 19-year-old extremely preterm subjects. Using multi-modal imaging techniques that combine T2 relaxometry and neurite density information, the results show that specific regions of the brain associated with white matter injuries due to preterm birth, such as the Posterior Limb of the Internal Capsule and Corpus Callosum, are still less myelinated in adulthood. Such findings might imply reduced connectivity in the adult preterm brain and explain the poor cognitive outcome.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Young, J.M., et al.: Longitudinal study of white matter development and outcomes in children born very preterm. Cereb. Cortex 27(8), 4094–4105 (2017)
Ball, G., et al.: The influence of preterm birth on the developing thalamocortical connectome. Cortex 49(6), 1711–1721 (2013)
Cardoso, M.J., et al.: Geodesic information flows: spatially-variant graphs and their application to segmentation and fusion. IEEE Trans. Med. Imaging 34(9), 1976–1988 (2015)
Dingwall, N., et al.: T2 relaxometry in the extremely-preterm brain at adolescence. Magn. Reson. Imaging 34(4), 508–514 (2016)
Glass, H.C., et al.: Outcomes for extremely premature infants. Anesth. Analg. 120(6), 1337 (2015)
Gozdas, E., Parikh, N.A., Merhar, S.L., Tkach, J.A., He, L., Holland, S.K.: Altered functional network connectivity in preterm infants: antecedents of cognitive and motor impairments? Brain Struct. Funct. 223(8), 3665–3680 (2018). https://doi.org/10.1007/s00429-018-1707-0
Irzan, H., Molteni, E., Hütel, M., Ourselin, S., Marlow, N., Melbourne, A.: White matter analysis of the extremely preterm born adult brain. Neuroimage 237, 118112 (2021)
Jeurissen, B., Tournier, J.-D., Dhollander, T., Connelly, A., Sijbers, J.: Multi-tissue constrained spherical deconvolution for improved analysis of multi-shell diffusion mri data. Neuroimage 103, 411–426 (2014)
MacKay, A., et al.: Insights into brain microstructure from the T2 distribution (2006)
Malhotra, A., et al.: Detection and assessment of brain injury in the growth-restricted fetus and neonate. Pediatr. Res. 82(2), 184–193 (2017)
Melbourne, A., et al.: Multi-modal measurement of the myelin-to-axon diameter g-ratio in preterm-born neonates and adult controls. In: Golland, P., Hata, N., Barillot, C., Hornegger, J., Howe, R. (eds.) MICCAI 2014. LNCS, vol. 8674, pp. 268–275. Springer, Cham (2014). https://doi.org/10.1007/978-3-319-10470-6_34
Melbourne, A., et al.: Longitudinal development in the preterm thalamus and posterior white matter: MRI correlations between diffusion weighted imaging and T2 relaxometry. Hum. Brain Mapp. 37(7), 2479–2492 (2016)
Narberhaus, A., et al.: Neural substrates of visual paired associates in young adults with a history of very preterm birth: alterations in fronto-parieto-occipital networks and caudate nucleus. Neuroimage 47(4), 1884–1893 (2009)
World Health Organization, The Partnership for Maternal, Newborn & Child Health, and Save the Children. Born too soon: the global action report on preterm birth (2012)
Smith, R.E., Tournier, J.D., Calamante, F., Connelly, A.: Sift2: enabling dense quantitative assessment of brain white matter connectivity using streamlines tractography. Neuroimage 119, 338–351 (2015)
Tournier, J.D., Calamante, F., Connelly, A.: Robust determination of the fibre orientation distribution in diffusion MRI: non-negativity constrained super-resolved spherical deconvolution. Neuroimage 35(4), 1459–1472 (2007)
Wang, S., et al.: Quantitative assessment of myelination patterns in preterm neonates using T2-weighted MRI. Sci. Rep. 9(1), 1–12 (2019)
Zhang, H., Schneider, T., Wheeler-Kingshott, C.A., Alexander, D.C.: Noddi: practical in vivo neurite orientation dispersion and density imaging of the human brain. Neuroimage 61(4), 1000–1016 (2012)
Acknowledgements
This work is supported by the EPSRC-funded UCL Centre for Doctoral Training in Medical Imaging (EP/L016478/1), the Department of Health NIHR-funded Biomedical Research Centre at University College London Hospitals and Medical Research Council (MR/N024869/1).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this paper
Cite this paper
Laureano, B., Irzan, H., Ourselin, S., Marlow, N., Melbourne, A. (2021). Myelination of Preterm Brain Networks at Adolescence. In: Sudre, C.H., et al. Uncertainty for Safe Utilization of Machine Learning in Medical Imaging, and Perinatal Imaging, Placental and Preterm Image Analysis. UNSURE PIPPI 2021 2021. Lecture Notes in Computer Science(), vol 12959. Springer, Cham. https://doi.org/10.1007/978-3-030-87735-4_17
Download citation
DOI: https://doi.org/10.1007/978-3-030-87735-4_17
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-87734-7
Online ISBN: 978-3-030-87735-4
eBook Packages: Computer ScienceComputer Science (R0)